
Khen ngợi LIFESPAN
“Trong cuốn sách sâu sắc và kích thích suy nghĩ này, đặt ra những câu hỏi về cách chúng ta già đi,”
và liệu con người có thể vượt qua sự suy tàn và thoái hóa, Sinclair vật lộn
với một số câu hỏi cơ bản nhất xung quanh khoa học về quá trình lão hóa.
Kết quả là một cuốn sách tinh tế và thú vị xứng đáng được đọc rộng rãi và
sâu sắc
—Siddhartha Mukherjee, người đoạt giải Pulitzer và là tác giả sách bán chạy nhất của New York Times
tác giả bán chạy nhất
"Nếu bạn từng tự hỏi chúng ta già đi như thế nào, liệu chúng ta có thể làm chậm hoặc thậm chí đảo ngược quá trình lão hóa hay không,"
Có thể sống khỏe mạnh hơn 100 năm, thì cuốn sách mới của David Sinclair, "Tuổi thọ" . . .
Tôi sẽ hướng dẫn bạn qua khoa học và các chiến lược thực tiễn để thực hiện điều đó.
"Thời gian khỏe mạnh của bạn bằng với tuổi thọ của bạn, và làm cho tuổi thọ của bạn dài và đầy sức sống."
—Mark Hyman, MD Giám đốc, Trung tâm Chức năng Cleveland Clinic
Y học và tác giả sách bán chạy số 1 của New York Times
"Đây là cuốn sách đầy tầm nhìn nhất về sự già đi mà tôi từng đọc. Hãy nắm bắt cơ hội—"
"và bắt lấy cuốn sách này!"
—Dean Ornish, MD, người sáng lập và tổng thống, Nghiên cứu Y học Phòng ngừa
Viện và tác giả sách bán chạy của New York Times của UnDo It!
"Trong cuốn sách Lifespan, David Sinclair một cách suôn sẻ cho chúng ta biết bí mật mà mọi người đều muốn biết."
Biết: cách sống lâu hơn và lão hóa chậm hơn. Sinclair thuyết phục chúng ta rằng không chỉ...
Có thể sống vượt qua một trăm năm, thì khó tránh khỏi rằng chúng ta sẽ có thể.
Một ngày nào đó hãy làm như vậy. Nếu bạn là người muốn biết cách đánh bại sự lão hóa,
"Sự sống còn là một cuốn sách cần đọc."
—William W. Li, MD, tác giả sách bán chạy nhất của New York Times, Eat to Beat
Bệnh
“Sáng suốt, truyền cảm hứng và đầy thông tin. [Sinclair] đã chuyển ngữ một khối lượng lớn”
chi tiết phân tử thành một chương trình mà chúng ta có thể sử dụng để sống lâu hơn và khỏe mạnh hơn.
Đối với bất kỳ ai quan tâm đến việc hiểu quá trình lão hóa, sống lâu hơn, và
"Tránh xa các bệnh tật của lão hóa, đây là cuốn sách nên đọc."
—Dale Bredesen, MD, tác giả sách bán chạy của New York Times với cuốn The End of
Bệnh Alzheimer
“Một cuốn sách đầy tầm nhìn từ một trong những nhà khoa học về tuổi thọ tài ba nhất của chúng ta”
thời gian. Tuổi thọ trao quyền cho chúng ta thay đổi sức khỏe của mình hôm nay trong khi tiết lộ một
"tiềm năng tương lai khi chúng ta sống trẻ lâu hơn."
—Sara Gottfried, MD, tác giả ăn khách của New York Times của cuốn The Hormone
Chữa trị
"Sẵn sàng để tâm trí bạn bị chấn động. Bạn đang cầm trong tay món quý giá"
kết quả của hàng thập kỷ làm việc, như được chia sẻ bởi Tiến sĩ David Sinclair, ngôi sao sáng trong lĩnh vực lão hóa
và tuổi thọ con người.
—Dave Asprey, nhà sáng lập và CEO của Bulletproof và New York Times
tác giả bán chạy nhất của Cuộc sống không khổ sở
"Hãy tưởng tượng một thế giới trong đó chúng ta có thể sống đủ lâu để gặp gỡ không chỉ riêng chúng ta"
cháu chắt, nhưng là chắt của chúng ta. Đây là tầm nhìn của Sinclair cho
tương lai của nhân loại, một tầm nhìn hướng tới khoa học, thiên nhiên, lịch sử, và thậm chí
chính trị để lập luận rằng điều này là có thể sống khỏe mạnh đến cả trăm tuổi.
“Thời gian sống đang dẫn đầu một cách mạnh mẽ.”
—Jason Fung, MD, tác giả của The Diabetes Code và The Obesity Code
"Trong cuốn sách Lifespan, Tiến sĩ David Sinclair . . . cung cấp cho chúng ta những công cụ hàng ngày mà chúng ta cần."
Bạn có thể sử dụng để ngăn chặn cái mà anh ấy hiện gọi là "căn bệnh của sự lão hóa."... Bạn nợ điều đó cho
bạn và những người thân yêu của bạn hãy đọc và làm theo lời khuyên của ông ấy, như tôi đã làm trong suốt thời gian qua.
15 năm!
—Steven R. Gundry, MD, tác giả bán chạy nhất của New York Times với cuốn sách The
Nghịch lý tuổi thọ và giám đốc y khoa của Hội Tim mạch Quốc tế
Viện Phổi
"Tuổi thọ vượt qua mọi điều chúng ta biết về sự lão hóa và tuổi thọ."
sự kết hợp giữa công trình khoa học xuất sắc, tư duy tiên phong và ước mơ về một
cuộc sống dài hơn, khỏe mạnh hơn và hạnh phúc hơn. Tuổi thọ mang đến một tầm nhìn cho tương lai của chúng ta và
l roadmap về cách để đến đó, kết hợp các đột phá khoa học và sự đơn giản
“Những thay đổi lối sống không chỉ giúp chúng ta cảm thấy trẻ hơn, mà còn thực sự giúp trẻ hóa.”
—Naomi Whittel, tác giả sách bán chạy của New York Times với tác phẩm Glow15
“David Sinclair tài ba trình bày một tầm nhìn táo bạo về tương lai trong đó”
Nhân loại có khả năng làm chậm hoặc đảo ngược quá trình lão hóa và sống trẻ hơn, khỏe mạnh hơn.
sống lâu hơn
—Victor J. Dzau, MD, chủ tịch Viện Hàn lâm Y học Hoa Kỳ
và Giám đốc điều hành của Trung tâm Y tế Đại học Duke
"Có rất ít cuốn sách khiến tôi suy nghĩ về khoa học theo một"
cách tiếp cận cơ bản và mới. Cuốn sách của David Sinclair đã làm điều đó cho tôi về lão hóa. Đây là một
"Cuốn sách mà ai cũng phải đọc khi trưởng thành."
—Leroy Hood, Tiến sĩ, giáo sư tại Viện Công nghệ California,
nhà phát minh, doanh nhân, thành viên của cả ba Học viện Quốc gia Hoa Kỳ, và
đồng tác giả của Bộ quy tắc các quy tắc
Trong "Lifespan", sức mạnh đầy đủ của [Sinclair's] lạc quan, hài hước và giọng nói nhẹ nhàng.
tài năng hùng biện như một nhà khoa học kể chuyện xuất hiện. Tôi hy vọng chúng ta có David.
Sinclair với chúng tôi và tiến hành khoa học cũng như viết sách trong 500 năm nữa.
khoảng một thế kỷ nữa.
—David Ewing Duncan, nhà báo đoạt giải, tác giả bán chạy, và
người điều hành Arc Fusion
“Tuổi thọ mang lại cho chúng ta hy vọng về một cuộc sống phi thường. Như bác sĩ xuất sắc David.”
Sinclair giải thích, lão hóa là một căn bệnh, và căn bệnh đó có thể được điều trị. Điều này thật sự mở mang tầm mắt.
Cuốn sách đưa bạn đến những mặt trận của những đột phá đáng kinh ngạc. Hãy tận hưởng tác phẩm này.
kiệt tác!
—Peter H. Diamandis, MD, tác giả sách bán chạy nhất của New York Times
Sự phong phú và Táo bạo
“Mô tả khoa học thực sự sẽ đặt câu hỏi về nền tảng của mọi thứ chúng ta”
"giả định về cuộc sống và xã hội của chúng ta."
—Salman Khan, người sáng lập Khan Academy
“David là một người tiên phong sẵn sàng thay đổi cách mà chúng ta suy nghĩ và hiểu.”
lão hóa
—Stephanie Lederman, Giám đốc điều hành của Liên đoàn Người cao tuổi Hoa Kỳ
Nghiên cứu (AFAR), New York
"Thông điệp và ưu tiên quan trọng nhất của thời đại chúng ta. Trong nhiều năm tới,"
"Nhân loại sẽ suy ngẫm về cuốn sách này với sự kinh ngạc và tôn trọng. Hãy đọc nó... Cuộc đời của bạn."
phụ thuộc vào điều đó.
—Marc Hodosh, cựu chủ sở hữu và đồng sáng lập TEDMED
Một cuộc trình diễn xuất sắc. Cuốn sách của Sinclair, và công trình cả đời của ông, đứng ngang hàng với nhân loại.
các đóng góp lớn nhất để giúp nâng cao niềm vui và hạnh phúc trong cuộc sống, xếp hạng
với các công trình của Jenner, Pasteur, Salk, Locke, Gandhi và Edison. A
kiệt tác.
—Martine Rothblatt, người sáng lập, Chủ tịch Hội đồng quản trị và Giám đốc điều hành của
"United Therapeutics và người sáng lập SiriusXM Đài Phát Thanh Vệ Tinh"
"Chạm chân lên mặt trăng đã thay đổi nhân loại. Trong Lifespan, Sinclair nói về"
bước cuối cùng cho nhân loại sẽ biến đổi cuộc sống của chúng ta vượt xa bất cứ điều gì chúng ta đã biết.
chưa từng có thể tưởng tượng. Tác giả táo bạo, khoa học sâu sắc, và chúng ta
Tương lai đã đến.
—Henry Markram, tiến sĩ, giáo sư tại EPFL, Thụy Sĩ, giám đốc của
Dự án Blue Brain và người sáng lập các tạp chí mở Frontiers
" Một cuốn sách trí thức và hấp dẫn với những cái nhìn kích thích về những điều nhất"
"vấn đề quan trọng về tương lai của bạn và mọi người."
—Andrew Scott, Tiến sĩ, giáo sư kinh tế tại Trường Kinh doanh London
Trường và tác giả của cuốn sách Cuộc sống 100 năm
Cảm ơn bạn đã tải xuống
cái này Simon & Schuster
sách điện tử
Nhận một sách điện tử MIỄN PHÍ khi bạn tham gia danh sách gửi thư của chúng tôi. Thêm vào đó, nhận thông tin cập nhật về những điều mới.
các bản phát hành, giao dịch, sách đọc được khuyên dùng và nhiều hơn nữa từ Simon & Schuster.
Nhấp vào bên dưới để đăng ký và xem các điều khoản và điều kiện.
NHẤN VÀO ĐÂY ĐỂ ĐĂNG KÝ
Bạn đã là người đăng ký? Vui lòng cung cấp email của bạn một lần nữa để chúng tôi có thể đăng ký ebook này.
và gửi cho bạn nhiều hơn những gì bạn thích đọc. Bạn sẽ tiếp tục nhận được
các ưu đãi độc quyền trong hộp thư của bạn.

Gửi bà nội Vera của tôi, người đã dạy tôi nhìn thế giới theo cách mà nó có thể.
là
Đến mẹ tôi, Diana, người đã chăm sóc cho con cái nhiều hơn cho bản thân.
Đến vợ tôi, Sandra, nền tảng vững chắc của tôi.
Và đến những đứa chắt chắt của tôi; Tôi rất mong được gặp gỡ.
bạn

HÀNH RỪNG. Trong thế giới hoang dã và kỳ diệu của tộc Garigal, thác nước và nước mặn
các cửa sông gió thổi qua những mỏm đá sa thạch cổ xưa, dưới những tán cây tối tăm của những chỗ bị cháy.
các cây máu, angophora và bạch đàn gai mà những chú kookaburra, currawong và walabies ăn
nhà
GIỚI THIỆU
LỜI CẦU NGUYỆN CỦA MỘT BÀ NGOẠI
Tôi lớn lên ở rìa của rừng. Về nghĩa bóng, sân sau của tôi là một...
rừng trăm mẫu. Về mặt nghĩa đen, nó lớn hơn nhiều so với điều đó. Nó tiếp tục như
đến mức mắt trẻ tuổi của tôi có thể nhìn thấy, và tôi chưa bao giờ cảm thấy chán khi khám phá nó. Tôi sẽ
leo núi và leo núi, dừng lại để nghiên cứu những con chim, những loài côn trùng, những loài bò sát. Tôi đã kéo
những thứ tách rời. Tôi xoa bụi giữa các ngón tay. Tôi nghe những âm thanh của...
hoang dã và cố gắng kết nối chúng với nguồn gốc của chúng.
Và tôi đã chơi. Tôi đã làm kiếm từ que và pháo đài từ đá. Tôi đã leo cây.
và đu trên các nhành cây, để chân qua những vách đá dốc và nhảy xuống
o của những điều mà có lẽ tôi không nên nhảy vào. Tôi hình dung mình như một
nhà du hành vũ trụ trên một hành tinh xa xôi. Tôi đã giả vờ là một thợ săn trong safari. Tôi nâng cao chiếc của mình
Giọng nói cho động vật như thể chúng là một khán giả tại nhà hát opera.
“Coooeey!” tôi sẽ gọi, có nghĩa là “Đến đây” trong ngôn ngữ của
Người Garigal, những cư dân bản địa.
Tôi không phải là người đặc biệt trong bất kỳ điều gì này, tất nhiên. Có rất nhiều đứa trẻ trong đó.
các vùng ngoại ô phía bắc của Sydney, những người chia sẻ niềm đam mê phiêu lưu và khám phá của tôi
và trí tưởng tượng. Chúng ta mong đợi điều này ở trẻ em. Chúng ta muốn chúng chơi theo cách này.
Cho đến khi, tất nhiên, họ “quá lớn” cho những thứ như vậy. Khi đó, chúng ta lại muốn họ.
đi học. Sau đó, chúng tôi muốn họ đi làm. Tìm một người bạn đời. Tiết kiệm tiền.
Mua một ngôi nhà.
Bởi vì, bạn biết đấy, thời gian đang trôi.
Bà của tôi là người đầu tiên nói với tôi rằng không nhất thiết phải như vậy.
Theo cách đó. Hoặc, tôi đoán, cô ấy không nói với tôi nhiều như là chỉ cho tôi thấy.
Cô đã lớn lên ở Hungary, nơi cô đã trải qua những mùa hè Bohemian.
bơi trong làn nước mát của hồ Balaton và đi bộ đường dài ở những ngọn núi của nó
bờ biển phía bắc tại một khu nghỉ dưỡng phục vụ cho các diễn viên, họa sĩ và nhà thơ.
các tháng mùa đông, cô ấy đã giúp quản lý một khách sạn ở Buda Hills trước khi người Đức Quốc xã đến.
đã tiếp quản và chuyển đổi nó thành bộ chỉ huy trung tâm của Schutzsta el, hoặc
"SS." in Vietnamese is typically translated as "SS." (the abbreviation remains the same). If you need a specific context, please provide more details.
Mười năm sau chiến tranh, trong những ngày đầu của sự chiếm đóng của Liên Xô,
Cộng sản bắt đầu đóng cửa biên giới. Khi mẹ của cô ấy cố gắng vượt qua.
Cô ấy đã bị bắt, giam giữ và bị kết án hai năm tù.
và đã qua đời ngay sau đó. Trong cuộc nổi dậy Hungary năm 1956, tôi
Bà ngoại đã viết và phát hành các bản tin chống Cộng sản trên đường phố.
của Budapest. Sau khi cuộc cách mạng bị đàn áp, người Soviet bắt đầu bắt giữ hàng chục
của hàng ngàn những người bất đồng chính kiến, và bà đã di cư sang Úc cùng với con trai của bà, cha tôi,
lý do là đây là nơi xa nhất họ có thể tới từ châu Âu.
Cô ấy không bao giờ đặt chân tới châu Âu nữa, nhưng cô ấy đã mang theo mọi thứ của Bohemia.
cô ấy. Tôi đã được nghe nói, cô là một trong những người phụ nữ đầu tiên mặc bikini tại
Nước Úc và bị đuổi khỏi bãi biển Bondi vì điều đó. Cô đã sống ở đây nhiều năm.
New Guinea—mà ngay cả ngày nay vẫn là một trong những nơi gồ ghề nhất trên thế giới.
hành tinh của chúng ta—một mình cô ấy.
Mặc dù dòng máu của cô ấy là người Do Thái Ashkenazi và cô đã được nuôi dạy một cách...
"Lutheran, bà ngoại tôi là một người rất thế tục. Chúng tôi có tương đương của"
Lời cầu nguyện của Chúa là bài thơ “Bây Giờ Chúng Ta” của tác giả người Anh Alan Alexander Milne.
Là sáu, kết thúc:
Nhưng bây giờ tôi sáu tuổi.
Tôi thông minh như người thông minh.
Vậy tôi nghĩ tôi sẽ được sáu tuổi bây giờ.
mãi mãi và mãi mãi
Cô ấy đã đọc bài thơ đó cho anh trai và tôi nghe đi nghe lại nhiều lần. Cô ấy nói, "sáu".
đó là độ tuổi tuyệt vời nhất, và cô ấy đã nỗ lực hết mình để sống cuộc đời với tinh thần và
sự ngưỡng mộ của một đứa trẻ ở độ tuổi đó.
Ngay cả khi chúng tôi còn rất trẻ, bà tôi không muốn chúng tôi gọi bà là...
“bà ngoại.” Cô cũng không thích thuật ngữ tiếng Hungary “nagymama,” hoặc bất kỳ từ nào khác.
các thuật ngữ thân mật ấm áp khác như “bubbie,” “bà,” và “nana.”
Đối với chúng tôi, những cậu bé, và mọi người khác, cô ấy đơn giản chỉ là Vera.
Vera đã dạy tôi lái xe, lạng lách và nghiêng ngả qua tất cả các làn đường.
“nhảy múa” theo bất kỳ bản nhạc nào trên đài radio của xe. Cô ấy bảo tôi hãy tận hưởng điều đó.
tuổi trẻ, để thưởng thức cảm giác được trẻ. Người lớn, cô ấy nói, luôn làm hỏng mọi thứ.
"Đừng lớn lên, cô ấy nói. Đừng bao giờ lớn lên."
Khi vào những năm 60 và 70 của bà, bà vẫn là người mà chúng ta gọi là "trẻ trung trong tâm hồn."
uống rượu vang với bạn bè và gia đình, ăn món ngon, kể những câu chuyện thú vị
giúp đỡ người nghèo, người ốm, và những người kém may mắn, giả vờ chỉ huy các buổi hòa nhạc
cười khuya đến tận đêm. Theo tiêu chuẩn của hầu hết mọi người, đó là dấu hiệu của một
"Cuộc sống sống đẹp."
Nhưng đúng vậy, thời gian đang trôi.
Vào giữa những năm 80 tuổi, Vera chỉ còn là một cái bóng của chính mình, và thập kỷ cuối cùng của bà.
Cuộc sống thật khó chịu khi chứng kiến. Cô ấy yếu ớt và ốm đau. Cô vẫn còn đủ sự khôn ngoan để
nhấn mạnh rằng tôi phải kết hôn với vị hôn thê của mình, Sandra, nhưng đến lúc đó âm nhạc không còn mang lại niềm vui cho cô ấy nữa và cô ấy
hầu như không ra khỏi ghế; sự năng động từng định hình cô ấy đã biến mất.
Cuối cùng, cô ấy đã từ bỏ hy vọng. “Chỉ đơn giản là vậy thôi,” cô ấy nói với tôi.
Cô ấy đã qua đời ở tuổi 92. Và, theo cách mà chúng ta đã được dạy để suy nghĩ về điều đó.
những điều này, cô ấy đã có một cuộc sống dài và tốt đẹp. Nhưng càng nghĩ về điều đó,
Càng ngày tôi càng tin rằng người mà cô ấy thực sự là đã chết rồi.
nhiều năm vào thời điểm đó.
Việc già đi có thể có vẻ là một sự kiện xa vời, nhưng mỗi chúng ta sẽ trải qua điều đó.
cuối đời. Sau khi chúng ta thở hơi thở cuối cùng, các tế bào của chúng ta sẽ kêu gào đòi oxy, chất độc.
sẽ tích lũy, năng lượng hóa học sẽ bị cạn kiệt, và cấu trúc tế bào sẽ
vỡ vụn. Vài phút sau, tất cả kiến thức, trí tuệ và kỷ niệm
những thứ mà chúng tôi trân quý, và tất cả tiềm năng tương lai của chúng ta, sẽ bị xóa sổ một cách không thể đảo ngược.
Tôi đã học được điều này một cách trực tiếp khi mẹ tôi, Diana, qua đời. Cha tôi, người mà tôi...
Anh trai và tôi đã ở đó. Đó là một cái chết nhanh chóng, may mắn, do sự tích tụ gây ra.
của dịch trong phổi còn lại của cô ấy. Chúng tôi vừa mới cười đùa cùng nhau về việc
điếu văn tôi đã viết trên chuyến đi từ Hoa Kỳ đến Úc, và sau đó
đột ngột cô ấy quằn quại trên giường, thở hổn hển tìm kiếm không khí mà không thể làm thoả mãn được cô.
"Nhu cầu oxy của cơ thể, nhìn chúng ta với sự tuyệt vọng trong ánh mắt."
Tôi nghiêng người vào và thì thầm vào tai cô ấy rằng cô là người mẹ tuyệt vời nhất mà tôi có thể có.
mong muốn. Chỉ trong vài phút, các nơ-ron của cô ấy đang chết đi, xóa đi không chỉ sự
kỷ niệm của những lời cuối cùng tôi dành cho cô ấy nhưng tất cả những kỷ niệm của cô ấy. Tôi biết một số người
"chết yên bình. Nhưng đó không phải là những gì xảy ra với mẹ tôi. Trong những khoảnh khắc đó"
Cô ấy đã biến đổi từ người đã nuôi dưỡng tôi thành một người co giật.
đám tế bào nghẹt thở, chiến đấu vì những dấu vết cuối cùng của năng lượng đang được tạo ra tại
cấp độ nguyên tử của sự tồn tại của cô ấy.
Mọi điều tôi có thể nghĩ là “Không ai nói cho bạn biết cái cảm giác chết đi như thế nào. Tại sao không…”
Có ai nói với bạn không?
Có rất ít người đã nghiên cứu cái chết một cách sâu sắc như Thế chiến II.
nhà làm phim tài liệu Claude Lanzmann. Và đánh giá của ông—thật vậy, sự
Cảnh báo—đang gây rùng mình. “Mỗi cái chết đều bạo lực,” ông nói vào năm 2010. “Không có...
cái chết tự nhiên, khác với hình ảnh chúng ta thường vẽ về người cha qua đời một cách yên tĩnh trong
"giấc ngủ của anh, được bao quanh bởi những người thân yêu. Tôi không tin vào điều đó."
Ngay cả khi chúng không nhận ra bạo lực của nó, trẻ em cũng đến để hiểu điều đó.
bi kịch của cái chết đến một cách bất ngờ khi còn quá trẻ. Đến tuổi bốn hoặc năm, họ
biết rằng cái chết xảy ra và là không thể đảo ngược. Đó là một suy nghĩ chấn động đối với họ, một
cơn ác mộng có thật

“MỘT CUỘC SỐNG TỐT ĐẸP, DÀI LÂU.” Bà của tôi “Vera” đã che chở cho người Do Thái trong Thế chiến II, sống trong điều kiện sơ sài.
New Guinea, và đã bị đuổi khỏi bãi biển Bondi vì mặc bikini. Cuộc đời của cô kết thúc là
khó nhìn. “Đây chỉ là cách mọi chuyện diễn ra,” cô ấy nói. Nhưng người mà cô thực sự là đã chết.
nhiều năm vào lúc đó.
Trước tiên, vì nó mang lại sự bình yên, hầu hết trẻ em thích nghĩ rằng có
các nhóm người nhất định được bảo vệ khỏi cái chết: cha mẹ, giáo viên và
chúng. Giữa 5 và 7 tuổi, tuy nhiên, tất cả trẻ em bắt đầu hiểu rõ về điều này.
Tính phổ quát của cái chết. Mỗi thành viên trong gia đình sẽ chết. Mỗi thú cưng. Mỗi cây cối.
Mọi thứ họ yêu. Cả chính họ nữa. Tôi nhớ lần đầu tiên học điều này. Tôi có thể.
cũng rất nhớ con trai lớn của chúng tôi, Alex, đã học nó.
"Ba, con sẽ không luôn có ba ở đây sao?"
“Thật buồn, không có,” tôi nói.
Alex đã khóc trong vài ngày, rồi ngừng lại, và không bao giờ hỏi tôi về điều đó.
Một lần nữa. Và tôi cũng chưa bao giờ nhắc đến nó nữa.
Nó không mất nhiều thời gian để suy nghĩ bi thảm bị chôn vùi sâu trong những ngóc ngách của
tiềm thức của chúng ta. Khi được hỏi liệu chúng có lo lắng về cái chết hay không, trẻ em thường nói
rằng họ không nghĩ về nó. Nếu được hỏi họ nghĩ gì về điều đó, họ sẽ nói rằng đó là
không phải mối bận tâm vì nó chỉ xảy ra trong tương lai xa, khi họ già đi.
Đó là quan điểm mà hầu hết chúng ta duy trì cho đến giữa tuổi năm mươi. Cái chết chỉ đơn giản là
Quá buồn và tê liệt để suy nghĩ về mỗi ngày. Thường thì, chúng ta nhận ra điều đó quá muộn. Khi mà
Khi nó gõ cửa, và chúng ta không chuẩn bị, điều đó có thể là thảm khốc.
Đối với Robin Marantz Henig, một cây bút của New York Times, “cay đắng”.
"Sự thật" về cái chết đến muộn trong cuộc đời, sau khi bà trở thành ông bà.
"Dưới tất cả những khoảnh khắc tuyệt vời mà bạn may mắn được chia sẻ và"
"Hãy tận hưởng," cô ấy viết, "cuộc sống của cháu bạn sẽ là một chuỗi dài những ngày sinh nhật."
"không sống để thấy."
Cần có sự can đảm để suy nghĩ một cách có ý thức về sự sống chết của những người thân yêu trước.
Nó thực sự xảy ra. Cần có nhiều can đảm hơn nữa để suy ngẫm sâu sắc về chính mình.
Đó là diễn viên hài và diễn viên Robin Williams người đầu tiên yêu cầu điều này.
sự can đảm từ tôi qua hình ảnh của John Keating, người thầy và anh hùng trong
bộ phim Xã Hội Những Poets Chết, người thách thức các học sinh tuổi teen của mình nhìn thẳng vào
gương mặt của những cậu bé đã khuất từ lâu trong bức ảnh phai nhạt. 4
“Chúng không khác bạn nhiều lắm, phải không?” Keating nói. “Bất khả chiến bại,”
Như bạn cảm thấy. . . . Đôi mắt của họ đầy hy vọng . . . Nhưng các quý ông, bạn thấy đấy, những cái này
"Các chàng trai bây giờ đang bón phân cho hoa đào."
Keating khuyến khích các cậu bé đến gần hơn để lắng nghe một thông điệp từ.
mộ. Đứng phía sau họ, trong một giọng nói tĩnh lặng, ma quái, anh thì thầm, “Carpe.
"Hãy nắm bắt từng khoảnh khắc. Hãy tận dụng ngày hôm nay, các chàng trai. Hãy làm cho cuộc sống của các bạn trở nên phi thường."
Cảnh đó đã có ảnh hưởng vô cùng lớn đến tôi. Có lẽ tôi sẽ không...
có động lực để trở thành giáo sư tại Harvard nếu không vì điều đó
Bộ phim. Ở độ tuổi 20, tôi cuối cùng đã nghe ai đó nói về điều mà tôi
"Bà ngoại đã dạy tôi từ khi còn nhỏ: Hãy làm phần của mình để nhân loại tồn tại."
Tốt nhất có thể. Đừng lãng phí một khoảnh khắc nào. Hãy ôm lấy tuổi trẻ của bạn; giữ chặt nó lại.
Cố gắng hết sức có thể. Chiến đấu vì điều đó. Chiến đấu vì điều đó. Đừng bao giờ ngừng chiến đấu vì điều đó.
Nhưng thay vì chiến đấu cho tuổi trẻ, chúng ta chiến đấu cho cuộc sống. Hoặc, cụ thể hơn, chúng ta...
đấu tranh chống lại cái chết.
Như một loài, chúng ta đang sống lâu hơn bao giờ hết. Nhưng không khỏe mạnh hơn. Không.
trong thế kỷ qua, chúng ta đã có thêm những năm tháng, nhưng không có thêm
cuộc sống—không phải là cuộc sống đáng sống. 5
Và vì vậy, hầu hết chúng ta, khi nghĩ về việc sống đến 100 tuổi, vẫn nghĩ rằng "Chúa".
"Cấm," vì chúng ta đã thấy những thập kỷ cuối cùng trông như thế nào, và đối với hầu hết.
Mọi người, hầu hết thời gian, họ không trông hấp dẫn chút nào. Máy thở và thuốc.
cocktail. Xương hông gãy và tã. Hóa trị và xạ trị. Phẫu thuật sau.
phẫu thuật sau phẫu thuật. Và hóa đơn bệnh viện; ôi trời ơi, những hóa đơn bệnh viện.
Chúng tôi đang chết dần và đau đớn. Người dân ở các nước giàu thường chi tiêu một cách...
một thập kỷ hoặc hơn chịu đựng bệnh tật sau những căn bệnh ở những giai đoạn cuối của cuộc sống.
nghĩ rằng điều này là bình thường. Khi tuổi thọ tiếp tục tăng ở các quốc gia nghèo hơn, điều này sẽ
trở thành số phận của hàng tỷ người khác. Những thành công của chúng ta trong việc kéo dài sự sống,
nhà phẫu thuật và bác sĩ Atul Gawande đã lưu ý, có tác dụng “làm
"tử vong trải nghiệm y tế."
Nhưng nếu điều đó không cần phải như vậy? Nếu chúng ta có thể trẻ hơn?
lâu hơn? Không phải là nhiều năm hơn mà là hàng thập kỷ. Điều gì sẽ xảy ra nếu những năm cuối cùng đó không trông giống như...
thật sự khác biệt đến mức nào so với những năm trước đó? Và nếu như, bằng
"Cứu lấy bản thân, chúng ta cũng có thể cứu thế giới?"
Có thể chúng ta sẽ không bao giờ có thể là sáu lần nữa—nhưng còn hai mươi sáu hoặc ba mươi sáu thì sao?
"Liệu có thể chơi như những đứa trẻ, sâu hơn vào cuộc sống của chúng ta, mà không lo lắng không?"
Về việc chuyển sang những điều mà người lớn phải làm sớm như vậy? Thì sao nếu tất cả điều đó...
Những điều chúng ta cần nén lại trong những năm thanh thiếu niên không cần phải như vậy.
nén lại sau al? Thì sao nếu chúng ta không bị căng thẳng trong độ tuổi 20? Thì sao nếu chúng ta...
không cảm thấy ở tuổi trung niên trong độ tuổi 30 và 40? Liệu có thể, ở tuổi 50, chúng ta muốn
"để tái phát minh bản thân và không thể nghĩ ra một lý do nào tại sao chúng ta không nên?"
Điều gì sẽ xảy ra nếu, ở độ tuổi 60, chúng ta không phải lo lắng về việc để lại di sản mà bắt đầu?
Một? Thì sao nếu chúng ta không phải lo lắng rằng thời gian đang trôi qua? Và nếu như tôi...
"Tôi đã nói với bạn rằng sớm thôi—thực ra là rất sớm, chúng ta sẽ không?"
Vâng, đó là điều tôi đang nói với bạn.
Tôi thật may mắn vì sau ba mươi năm tìm kiếm sự thật về con người.
Sinh học, tôi thấy mình ở trong một vị trí độc đáo. Nếu bạn đến thăm tôi ở Boston,
Bạn sẽ thấy tôi thường xuyên ở trong phòng thí nghiệm của mình tại Trường Y Harvard.
nơi tôi là giáo sư tại Khoa Di truyền và là đồng giám đốc của Paul
Trung tâm F. Glenn về Cơ chế Sinh học của Lão hóa. Tôi cũng điều hành một phòng thí nghiệm chị em tại
trường đại học của tôi, Đại học New South Wales ở Sydney. Trong các phòng thí nghiệm của tôi, các đội
của những sinh viên xuất sắc và tiến sĩ đã cả tăng tốc và đảo ngược lão hóa trong mô hình
các sinh vật và đã chịu trách nhiệm cho một số nghiên cứu được trích dẫn nhiều nhất trong lĩnh vực này
eld, đã được công bố trong một số tạp chí khoa học hàng đầu thế giới. Tôi cũng là một
người đồng sáng lập một tạp chí, Aging, cung cấp không gian cho các nhà khoa học khác để công bố bài viết.
Nghiên cứu của họ về một trong những câu hỏi thách thức và thú vị nhất của thời đại chúng ta.
và là một đồng sáng lập của Học viện Nghiên cứu Sức khỏe và Tuổi thọ, một nhóm các
hai mươi nhà nghiên cứu hàng đầu về lão hóa trên toàn thế giới.
Trong quá trình cố gắng áp dụng những phát hiện của mình vào thực tiễn, tôi đã giúp bắt đầu một số dự án.
các công ty công nghệ sinh học và ngồi ở vị trí chủ tịch của các hội đồng cố vấn khoa học
của nhiều công ty khác. Những công ty này làm việc với hàng trăm học giả hàng đầu trong
các lĩnh vực khoa học từ nguồn gốc của sự sống đến hệ gen học đến dược phẩm. 7 Tôi
Tôi tất nhiên biết về những phát hiện của phòng thí nghiệm của mình nhiều năm trước khi chúng được thực hiện.
công khai, nhưng qua những mối liên hệ này, tôi cũng nhận thức được nhiều điều khác
những khám phá chuyển mình trước thời gian, đôi khi trước cả một thập kỷ.
Các trang tiếp theo sẽ là thẻ backstage của bạn và chỗ ngồi hàng đầu của bạn.
Đã nhận được sự công nhận tương đương với tước hiệp sĩ ở Úc và đảm nhận
Vai trò của một đại sứ, tôi đã dành khá nhiều thời gian để tóm tắt.
các nhà lãnh đạo chính trị và kinh doanh trên thế giới về những cách mà chúng ta
Sự hiểu biết về lão hóa đang thay đổi - và điều đó có nghĩa gì cho nhân loại trong tương lai.
tiến tới. 8
Tôi đã áp dụng nhiều phát hiện khoa học của mình vào cuộc sống của chính mình, như nhiều người khác cũng vậy.
các thành viên trong gia đình, bạn bè, và đồng nghiệp. Kết quả—mà, nên được
được ghi nhận, hoàn toàn là giai thoại - đang khích lệ. Tôi hiện tại 50 tuổi, và tôi cảm thấy như một
Nhóc. Vợ và bọn trẻ của tôi sẽ nói với bạn rằng tôi cũng hành xử như một đứa trẻ.
Điều đó bao gồm cả việc tò mò, thuật ngữ của Úc chỉ người thích xía vào chuyện người khác.
có phần thắc mắc quá nhiều, có lẽ được bắt nguồn từ những con quạ currawong thường hay đấm
qua các nắp nhôm của những chai sữa được giao đến nhà chúng ta và uống cái đó
Vắt sữa từ chúng. Những người bạn cũ ở trường trung học vẫn thích trêu chọc tôi về việc này.
Mỗi khi họ đến nhà bố mẹ tôi, họ đều thấy tôi đang kéo.
một cái gì đó riêng biệt: kén của một con bướm cưng, nơi trú ngụ cuộn lại của một con nhện, một cái cũ
máy tính, công cụ của cha tôi, một chiếc xe hơi. Tôi đã trở nên khá giỏi trong việc đó. Tôi chỉ không được quá
Giỏi trong việc lắp ráp những thứ này lại với nhau.
Tôi không thể chịu đựng được việc không biết một thứ gì đó hoạt động ra sao hoặc nó đến từ đâu.
Vẫn không thể - nhưng ít nhất bây giờ tôi được trả tiền cho điều đó.
Ngôi nhà thời thơ ấu của tôi nằm chênh vênh trên sườn núi đá. Phía dưới là một con sông mà
chảy vào cảng Sydney. Arthur Philip, thống đốc đầu tiên của New South
"Wales, đã khám phá những thung lũng này vào tháng 4 năm 1788, chỉ vài tháng sau khi ông ấy và"
Đội hạm đội đầu tiên của lính thủy đánh bộ, tù nhân và gia đình họ đã thành lập một thuộc địa trên...
"bờ biển của cái mà ông gọi là 'bến cảng đẹp nhất và rộng lớn nhất trong vũ trụ.'"
Người có trách nhiệm lớn nhất khiến ông ấy có mặt ở đó là nhà thực vật học Sir Joseph.
"Các ngân hàng, những người đã đi thuyền dọc theo bờ biển Australia mười tám năm trước"
Thuyền trưởng James Cook trong "hành trình vòng quanh thế giới." 9
Sau khi trở về London với hàng trăm mẫu thực vật để gây ấn tượng với ông ấy
Các đồng nghiệp, các ngân hàng đã vận động Vua George III để bắt đầu một thuộc địa hình sự của Anh trên.
lục địa, địa điểm tốt nhất cho điều này, theo ông lập luận, không phải ngẫu nhiên, sẽ là một vịnh
được gọi là "Thực vật học" ở "Mũi Banks." Những người định cư của Đoàn Tàu Đầu Tiên nhanh chóng phát hiện rằng
Vịnh Botany, mặc dù có tên rất xuất sắc, nhưng không có nguồn nước nào.
đã cập bến cảng Sydney và phát hiện một trong những “rias” lớn nhất thế giới, một nơi rất
Một con đường thủy sâu có nhánh hình thành khi hệ thống sông Hawkesbury đã...
bị ngập lụt do mực nước biển dâng lên sau kỷ băng hà cuối cùng.
Lúc 10 tuổi, tôi đã khám phá ra thông qua việc khám phá rằng con sông
Trong vườn sau nhà tôi, chảy xuống Middle Harbor, một nhánh của cảng Sydney.
Nhưng tôi không thể chịu đựng được việc không biết con sông bắt nguồn từ đâu nữa. Tôi cần phải
biết cái gì đó ở đầu nguồn một con sông trông như thế nào.
Tôi đã theo nó ngược dòng, rẽ trái lần đầu tiên nó phân nhánh và rẽ phải lần sau.
đi qua và ra khỏi một vài ngoại ô. Đến lúc hoàng hôn, tôi đã cách xa hàng miles.
nhà, ở phía bên kia ngọn núi cuối cùng trên đường chân trời. Tôi đã phải nhờ một người lạ cho phép tôi
Gọi mẹ tôi để cầu xin bà ấy đến đón tôi. Một vài lần sau đó, tôi đã thử.
Tìm kiếm dòng chảy phía thượng nguồn, nhưng không bao giờ gần đến nguồn. Như Juan.
Ponce de León, nhà thám hiểm người Tây Ban Nha của Florida, nổi tiếng với cuộc tìm kiếm thần thoại của ông.
Để tìm kiếm Suối Nguồn Trẻ, tôi đã thất bại. 11
Kể từ khi tôi còn nhớ, tôi đã muốn hiểu tại sao chúng ta lại già đi.
Nhưng việc tìm nguồn gốc của một quá trình sinh học phức tạp giống như việc tìm kiếm.
mùa xuân ở nguồn một con sông: không dễ dàng.
Trên hành trình của mình, tôi đã đi qua lại và có những ngày tôi muốn.
từ bỏ. Nhưng tôi đã kiên trì. Trên con đường đó, tôi đã thấy nhiều nhánh sông, nhưng
Tôi cũng đã tìm thấy cái mà có thể là nguồn gốc. Trong các trang tiếp theo, tôi sẽ trình bày một cái mới.
ý tưởng về lý do tại sao sự lão hóa phát triển và cách nó phù hợp với những gì tôi gọi là Thông tin
Lý thuyết về Lão hóa. Tôi cũng sẽ cho bạn biết lý do tại sao tôi đã coi lão hóa là một căn bệnh—
bệnh phổ biến nhất—một căn bệnh không chỉ có thể mà còn nên được điều trị tích cực
được điều trị. Đó là phần I.
Trong phần II, tôi sẽ giới thiệu cho bạn những bước có thể thực hiện ngay bây giờ—và
các liệu pháp mới đang được phát triển - có thể làm chậm, ngừng lại hoặc đảo ngược quá trình lão hóa, mang lại
một sự chấm dứt cho sự lão hóa như chúng ta biết đến.
Và vâng, tôi hoàn toàn nhận thức được ý nghĩa của cụm từ "đưa đến sự kết thúc".
“lão hóa như chúng ta biết,” vậy nên, trong phần III, tôi sẽ thừa nhận nhiều tương lai có thể xảy ra.
Những hành động này có thể tạo ra và đề xuất một con đường cho một tương lai mà chúng ta có thể nhìn tới.
về một thế giới mà cách chúng ta có thể đạt được tuổi thọ cao hơn là
thông qua một khoảng thời gian sức khỏe ngày càng tăng, phần đời của chúng ta sống không có bệnh tật
hoặc khuyết tật.
Có rất nhiều người sẽ nói với bạn rằng đó chỉ là một câu chuyện cổ tích—gần giống như cái.
Các tác phẩm của H. G. Wells hơn những tác phẩm của C. R. Darwin. Một số trong số đó rất thông minh.
Một vài người thậm chí là những người hiểu khá rõ sinh học con người và những người mà tôi
tôn trọng
Những người đó sẽ nói với bạn rằng lối sống hiện đại của chúng ta đã nguyền rủa chúng ta với
thời gian sống ngắn lại. Họ sẽ nói bạn khó có khả năng sống đến 100 tuổi và rằng
Con của bạn cũng không có khả năng đạt mốc thế kỷ. Chúng sẽ nói rằng chúng đã
nhìn vào khoa học của mọi thứ và đã thực hiện các dự đoán, và nó chắc chắn không có vẻ gì
Có khả năng rằng các cháu của bạn sẽ không đến được sinh nhật lần thứ 100 của chúng. Và
Họ sẽ nói rằng nếu bạn đạt đến 100 tuổi, có lẽ bạn sẽ không đến đó một cách khỏe mạnh.
Bạn chắc chắn sẽ không ở đó lâu đâu. Và nếu họ cho phép bạn thì mọi người sẽ sống.
dài hơn, họ sẽ nói với bạn rằng đó là điều tồi tệ nhất cho hành tinh này. Con người là
kẻ thù!
Họ có bằng chứng tốt cho tất cả điều này - toàn bộ lịch sử của nhân loại, trong.
Chắc chắn rồi, từng chút một, qua hàng thiên niên kỷ, chúng ta đã thêm năm tháng vào.
"Cuộc sống trung bình của con người, họ sẽ nói. Hầu hết chúng ta không sống qua tuổi 40, và rồi chúng ta lại làm được."
Hầu hết chúng ta không đến được 50, và rồi chúng ta đã đến. Hầu hết chúng ta không đến được 60, và...
sau đó chúng tôi đã làm. 12 Nói chung, những mức tăng này trong tuổi thọ đến khi ngày càng nhiều người trong chúng ta
có được quyền truy cập vào nguồn thực phẩm ổn định và nước sạch. Và chủ yếu là mức trung bình đã
đẩy lên từ dưới; số ca tử vong trong thời kỳ sơ sinh và trẻ em giảm, và
Tuổi thọ trung bình đã tăng. Đây là toán học đơn giản về tỷ lệ tử vong của con người.
Nhưng mặc dù trung bình vẫn tăng lên, giới hạn thì không. Miễn là chúng ta đã
đã ghi chép lịch sử, chúng ta đã biết đến những người đã đạt đến tuổi 100
năm và những người có thể đã sống thêm vài năm nữa. Nhưng rất ít người đạt được
Gần như không ai đạt được 115.
Hành tinh của chúng ta đã là nhà của hơn 100 tỷ người cho đến nay. Chúng ta biết
của chỉ một người, Jeanne Calment ở Pháp, người được cho là đã sống qua tuổi 120.
Hầu hết các nhà khoa học tin rằng bà đã qua đời vào năm 1997 ở tuổi 122, mặc dù cũng có những ý kiến khác.
có thể con gái của cô ấy đã thay thế cô để tránh trả thuế. 13 Dù có hay không
Cô ấy thực sự đã đạt đến độ tuổi đó, thật sự không quan trọng; những người khác đã đến gần trong một...
vài năm ở độ tuổi đó nhưng hầu hết chúng tôi, chính xác là 95 phần trăm, đã chết trước khi đó
100. Vì vậy, điều đó chắc chắn có nghĩa khi mọi người nói rằng chúng ta có thể tiếp tục chạm.
ra khỏi mức trung bình, nhưng chúng tôi không có khả năng thay đổi giới hạn. Họ nói là dễ dàng để
kéo dài tuổi thọ tối đa của chuột hoặc chó, nhưng chúng ta, con người, thì khác.
Chúng ta đang sống quá lâu rồi.
Họ sai.
Cũng có sự khác biệt giữa việc kéo dài cuộc sống và kéo dài sự sống động. Chúng ta
có khả năng cả hai, nhưng chỉ đơn giản là giữ cho mọi người sống—hàng thập kỷ sau khi họ đã sống
trở nên bị chi phối bởi nỗi đau, bệnh tật, sự yếu đuối và bất động—không phải là một đức tính.
Sức sống kéo dài - có nghĩa là không chỉ là thêm nhiều năm sống mà còn là nhiều năm sống tích cực hơn.
Những người khỏe mạnh và hạnh phúc đang đến. Nó sẽ đến sớm hơn hầu hết mọi người.
mong đợi. Khi những đứa trẻ sinh ra hôm nay đến tuổi trung niên,
Jeanne Calment có thể không thậm chí nằm trong danh sách 100 người già nhất thế giới.
thời gian. Và đến đầu thế kỷ tiếp theo, một người 122 tuổi vào ngày của anh ấy
Hoặc có thể nói rằng cái chết của cô ấy đã sống một cuộc đời đầy đủ, mặc dù không đặc biệt dài.
Một trăm hai mươi năm có thể không phải là một trường hợp ngoại lệ mà là một kỳ vọng, vì vậy
nhiều đến nỗi chúng ta thậm chí sẽ không gọi nó là sự sống lâu, mà chúng ta sẽ đơn giản gọi nó là “cuộc sống,” và chúng ta
sẽ nhìn lại với nỗi buồn về khoảng thời gian trong lịch sử của chúng ta mà không như vậy.
Giới hạn tối đa là gì? Tôi không nghĩ có giới hạn nào. Nhiều đồng nghiệp của tôi
Đồng ý. Không có quy luật sinh học nào nói rằng chúng ta phải lão hóa. Những người nói như vậy không biết họ đang nói về điều gì. Chúng ta có thể vẫn còn cách xa điều đó.
từ một thế giới mà cái chết là điều hiếm hoi, nhưng chúng ta không xa việc đẩy nó đi mãi mãi
xa hơn vào tương lai.
Tất cả điều này trên thực tế là không thể tránh khỏi. Sự kéo dài tuổi thọ khỏe mạnh là điều có thể nhìn thấy. Vâng,
Toàn bộ lịch sử của nhân loại gợi ý điều ngược lại. Nhưng khoa học về tuổi thọ
"Sự kéo dài trong thế kỷ này nói rằng những ngõ cụt trước đây là nghèo nàn."
hướng dẫn
Cần có tư duy cấp tiến để bắt đầu tiếp cận điều này sẽ có nghĩa là gì cho
loài của chúng ta. Không có gì trong hàng tỷ năm tiến hóa của chúng ta đã chuẩn bị cho điều này.
Điều này, đó là lý do tại sao nó dễ dàng, và thậm chí hấp dẫn, để tin rằng nó đơn giản không thể.
được hoàn thành.
Nhưng đó là điều mà mọi người đã nghĩ về quyền con người, cho đến khi
"Khoảnh khắc ai đó đã làm điều đó."
Hôm nay, anh em nhà Wright đã trở lại xưởng làm việc của họ và đạt được thành công.
sở hữu những chiếc glider của họ xuống những đụn cát ở Kitty Hawk. Thế giới sắp sửa
thay đổi.
Và cũng giống như đã xảy ra trong những ngày trước 17 tháng 12 năm 1903,
Phần lớn nhân loại không chú ý. Thật sự không có bối cảnh nào để
Xây dựng ý tưởng về ánh sáng có kiểm soát, được cung cấp sức mạnh vào thời điểm đó, vì vậy ý tưởng là.
huyền bí, ma thuật, chất liệu của tiểu thuyết giả tưởng.
Rồi: lifto. Và mọi thứ không còn như xưa nữa.
Chúng ta đang ở một điểm khác của bước ngoặt lịch sử. Những gì trước đây dường như
Ma thuật sẽ trở thành hiện thực. Đây là thời điểm mà nhân loại sẽ định nghĩa lại điều gì là.
có thể; một thời điểm kết thúc điều không thể tránh khỏi.
Thật vậy, đây là thời điểm chúng ta sẽ định nghĩa lại ý nghĩa của việc trở thành con người.
Đây không chỉ là sự khởi đầu của một cuộc cách mạng, mà là sự khởi đầu của một cuộc tiến hóa.
PHẦN I
NHỮNG GÌ CHÚNG TÔI BIẾT
QUÁ KHỨ
MỘT
Sống nguyên thủy
HÃY TƯỞNG TƯỢNG MỘT HÀNH TINH CÓ KÍCH THƯỚC TƯƠI ĐƯỢC VỚI HÀNH TINH CỦA CHÚNG TA, CÁCH SAO CỦA NÓ TƯƠI ĐƯỢC, ĐANG XOAY.
quanh trục của nó nhanh hơn một chút, sao cho một ngày kéo dài khoảng hai mươi giờ. Nó được bao phủ
với một đại dương nông đầy nước mặn và không có lục địa nào đáng nói—chỉ có một số
các chuỗi đảo đen basalt thưa thớt nổi lên trên đường nước.
không khí không có cùng hỗn hợp khí như của chúng ta. Nó là một môi trường ẩm ướt và độc hại.
màng chắn của nitơ, metan và carbon dioxide.
Không có oxy. Không có sự sống.
Bởi vì hành tinh này, hành tinh của chúng ta cách đây 4 tỷ năm, là một nơi tàn nhẫn.
nơi khắc nghiệt. Nóng bức và núi lửa. Điện khí. Dữ dội.
Nhưng điều đó sắp thay đổi. Nước đang tích tụ bên cạnh các lỗ thông hơi nhiệt độ ấm mà
rác một trong những hòn đảo lớn hơn. Các phân tử hữu cơ phủ lên tất cả các bề mặt, có
cưỡi trên lưng của các thiên thạch và sao chổi. Ngồi trên đá núi lửa khô cằn,
các phân tử này sẽ chỉ còn là các phân tử, nhưng khi hòa tan trong những bể nước ấm
nước, qua các chu kỳ ướt và khô ở rìa các hồ, một đặc biệt
Hóa học diễn ra. Khi các axit nucleic tập trung, chúng phát triển thành
polimere, cách mà tinh thể muối hình thành khi một vũng nước bên bờ biển bốc hơi. Những cái này là
các phân tử RNA đầu tiên của thế giới, là tiền thân của DNA. Khi cái ao
vật liệu di truyền nguyên thủy được bao bọc bởi axit béo để hình thành
bong bóng xà phòng siêu nhỏ—màng tế bào đầu tiên. 2
Chẳng bao lâu, có thể chỉ một tuần, trước khi những cái ao nông bị bao phủ.
với một lớp bọt vàng của hàng triệu triệu tế bào tiền thân được lấp đầy bởi các sợi ngắn của
axit nucleic, mà hôm nay chúng ta gọi là gen.
Hầu hết các protocel đều được tái chế, nhưng một số sống sót và bắt đầu tiến hóa.
"các con đường chuyển hóa nguyên thủy, cho đến khi ARN bắt đầu sao chép chính nó."
Điểm này đánh dấu nguồn gốc của sự sống. Giờ đây, khi sự sống đã hình thành - dưới dạng xà phòng axit béo.
các bọt khí chứa vật liệu di truyền—chúng bắt đầu cạnh tranh để giành sự thống trị.
Không có đủ tài nguyên để chia sẻ. Chúc cho kẻ xấu nhất giành chiến thắng.
Ngày này qua ngày khác, những sinh vật siêu nhỏ, mong manh bắt đầu tiến hóa thành
các hình thức tiên tiến hơn, lan tỏa vào các con sông và hồ.
Một mối đe dọa mới xuất hiện: một mùa khô dài. Mức độ của lớp bùn...
mực nước hồ đã giảm vài feet trong mùa khô, nhưng các hồ đã
Luôn luôn dồn lên lại khi những cơn mưa quay trở lại. Nhưng năm nay, nhờ vào sự bất thường.
hoạt động núi lửa mạnh mẽ ở phía bên kia của hành tinh, những cơn mưa hàng năm không
rơi như họ thường làm và những đám mây trôi qua. Các hồ hoàn toàn khô cạn.
Điều còn lại là một lớp vỏ dày, màu vàng phủ lên các đáy hồ. Đó là một hệ sinh thái.
được xác định không phải bởi sự lên xuống hàng năm của nước mà bởi một cách tàn nhẫn
cuộc đấu tranh sinh tồn. Và hơn thế nữa: đó là một cuộc đấu tranh cho tương lai—bởi vì
Các sinh vật sống sót sẽ trở thành tổ tiên của mọi sinh vật sống trong tương lai.
vi khuẩn, nấm, thực vật và động vật.
Trong đám tế bào đang hấp hối này, mỗi tế bào đều cố gắng và vật lộn để sống sót.
tối thiểu nhất về dinh dưỡng và độ ẩm, từng cái đều làm những gì có thể để
trả lời câu đố nguyên thủy để tái tạo, có một loài duy nhất. Hãy gọi nó là Magna.
siêu tồn tại.
Nó không trông khác biệt nhiều so với các sinh vật khác trong ngày, nhưng M.
superstes có một lợi thế rõ ràng: nó đã phát triển một cơ chế di truyền để sinh tồn.
Sẽ có nhiều bước tiến hóa phức tạp hơn trong những kỷ nguyên sắp tới.
"Các thay đổi cực đoan đến mức toàn bộ nhánh sự sống sẽ xuất hiện. Những thay đổi này—"
sản phẩm của đột biến, chèn, tái sắp xếp gen và ngang
chuyển giao gen từ loài này sang loài khác—sẽ tạo ra các sinh vật có
đối xứng hai bên, thị giác lập thể, và thậm chí là ý thức.
So với những điều khác, bước tiến hóa ban đầu này trông có vẻ khá đơn giản.
Đó là một mạch. Một mạch gen.
Mạch bắt đầu với gen A, một người quản lý ngăn cản tế bào sinh sản.
khi thời điểm khó khăn. Điều này rất quan trọng, vì trên hành tinh Trái Đất sơ khai, hầu hết các thời điểm đều là
khó. Mạch này cũng có một gen B, mã hóa cho một protein “im lặng”.
Protein im lặng này tắt gen A khi mọi thứ tốt đẹp, để tế bào có thể tạo ra.
bản sao của chính nó khi và chỉ khi nó và con cái của nó có khả năng sống sót.
Các gen đó không phải là mới. Tất cả đời sống trong hồ đều có hai gen này. Nhưng
Điều làm cho M. superstes trở nên độc đáo là gen ức chế B đã đột biến để mang lại cho nó.
một chức năng thứ hai: nó giúp sửa chữa DNA. Khi DNA của tế bào bị đứt,
Protein tắt mã bởi gen B di chuyển từ gen A để giúp đỡ với DNA.
sửa chữa, làm cho gen A hoạt động. Điều này tạm thời ngừng toàn bộ tình dục và sinh sản.
cho đến khi việc sửa chữa DNA hoàn tất.
Điều này có lý, bởi vì trong khi DNA bị hỏng, tình dục và sinh sản thì vẫn tiếp tục.
những điều cuối cùng mà một sinh vật nên làm. Ở các sinh vật đa bào trong tương lai,
Ví dụ, tế bào mà không ngừng lại khi vượt qua một vết gãy DNA sẽ gần như
chắc chắn mất đi vật chất di truyền. Điều này xảy ra vì DNA bị kéo ra trước khi tế bào
phân chia từ chỉ một vị trí gắn kết trên DNA, kéo theo phần còn lại của
DNA với nó. Nếu DNA bị gãy, một phần của nhiễm sắc thể sẽ bị mất hoặc
Bị lặp lại. Các tế bào sẽ có khả năng chết hoặc nhân lên một cách không kiểm soát thành khối u.
Với một loại bộ giảm âm gen mới có khả năng sửa chữa DNA, M. superstes sở hữu một
Cạnh. Nó co cụm lại khi DNA của nó bị tổn thương, sau đó hồi phục. Nó được siêu chuẩn bị.
để sinh tồn.
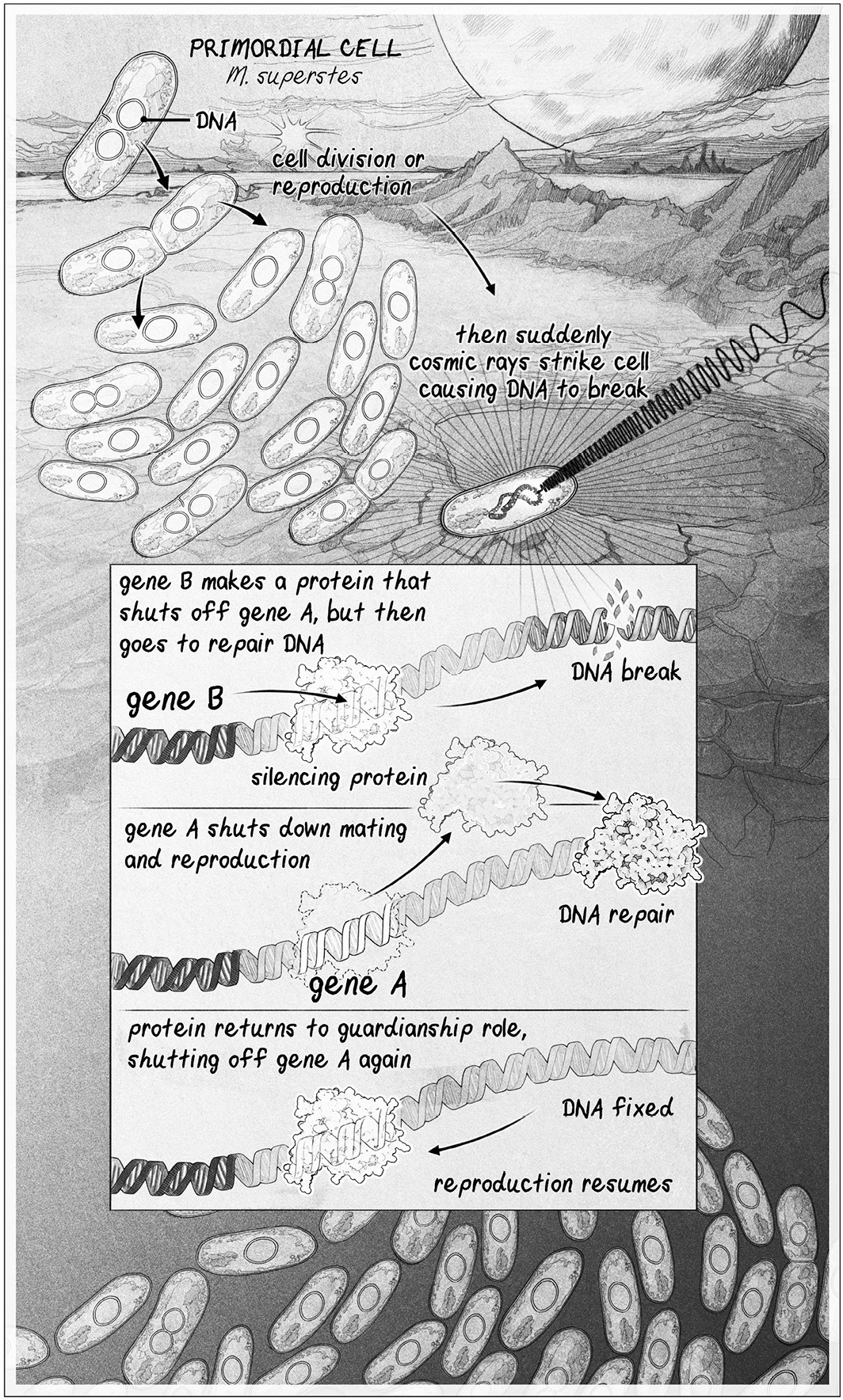
SỰ TIẾN HÓA CỦA LÃO HÓA. Một mạch gen 4 tỷ năm tuổi trong các dạng sống đầu tiên sẽ có
đã chuyển sang sinh sản trong khi DNA đang được sửa chữa, cung cấp lợi thế sống sót. Gen A
biến đổi và gene B tạo ra một protein giúp chuyển gene A khi an toàn.
tái sản xuất. Khi DNA bị đứt, protein được tạo ra bởi gen B sẽ rời đi để sửa chữa DNA.
Kết quả là, gen A được kích hoạt để ngừng sinh sản cho đến khi quá trình sửa chữa hoàn tất. Chúng ta đã thừa hưởng
một phiên bản nâng cao của mạch sinh tồn này.
Và điều đó thì tốt, vì bây giờ lại đến một cuộc tấn công nữa vào cuộc sống. Mạnh mẽ.
các tia vũ trụ từ một vụ bùng phát mặt trời xa xôi đang chiếu xuống Trái Đất, xé toạc những
DNA của tất cả các vi sinh vật trong những hồ đang chết. Phần lớn trong số chúng tiếp tục tồn tại.
chia sẻ như thể không có điều gì xảy ra, không biết rằng bộ gen của họ đã bị
Bị hỏng và việc tái tạo sẽ giết chúng. Số lượng DNA không đồng đều là
chia sẻ giữa các tế bào của mẹ và con gái, làm cho cả hai đều bị hỏng.
Cuối cùng, nỗ lực này là vô vọng. Các tế bào sẽ chết, và không còn gì nữa.
Không có gì, nghĩa là, nhưng M. superstes. Bởi vì khi các tia gây ra sự tàn phá, M.
superstes làm điều gì đó khác thường: nhờ vào sự di chuyển của protein B ra xa
từ gen A để giúp sửa chữa các đứt gãy DNA, gen A được kích hoạt và tế bào
dừng hầu như mọi thứ họ đang làm, chuyển hướng năng lượng hạn chế của họ vào
xây dựng lại DNA đã bị đứt. Nhờ vào sự coi thường của nó đối với cổ xưa
"Cần thiết để tái sản xuất, M. superstes đã tồn tại."
Khi thời kỳ khô hạn mới nhất kết thúc và các hồ nước hồi phục, M. superstes tỉnh dậy.
Bây giờ nó có thể tái sản xuất. Lại và lại nó làm như vậy. Nhân lên. Di chuyển đến mới.
hệ sinh thái. Tiến hóa. Tạo ra những thế hệ con cháu mới qua các thế hệ.
Họ là Adam và Eva của chúng ta.
Giống như Adam và Eva, chúng ta không biết liệu M. superstes có từng tồn tại hay không. Nhưng tôi...
Nghiên cứu trong suốt hai mươi lăm năm qua cho thấy rằng mọi sinh vật sống mà chúng ta thấy
Xung quanh chúng ta hôm nay là sản phẩm của một người sống sót vĩ đại, hoặc ít nhất là một nguyên thủy.
Sinh vật rất giống nó. Hồ sơ hóa thạch trong gen của chúng ta đi một chặng đường dài để
chứng minh rằng mọi sinh vật sống chia sẻ hành tinh này với chúng ta vẫn mang theo điều này
mạch di truyền sinh tồn cổ xưa, với hình thức cơ bản gần như giống nhau. Nó vẫn hiện hữu trong
mỗi loại cây. Nó có ở mọi loại nấm. Nó có ở mọi loài động vật.
Nó ở trong chúng ta.
Tôi đề xuất lý do mà mạch gen này được bảo tồn là vì nó khá đơn giản.
và giải pháp thanh thoát cho những thử thách đôi khi thô bạo và đôi khi
thế giới phong phú hơn để đảm bảo sự sống còn của các sinh vật mang nó. Nó
là, về bản chất, một bộ dụng cụ sinh tồn nguyên thủy chuyển hướng năng lượng đến khu vực lớn nhất.
Cần, xing những gì tồn tại trong thời điểm mà những căng thẳng của thế giới đang cấu kết để
Gây ra sự tàn phá trên bộ gen, trong khi cho phép sinh sản chỉ khi nhiều hơn.
Thời điểm thuận lợi sẽ chiếm ưu thế.
Và nó đơn giản và mạnh mẽ đến mức không chỉ đảm bảo sự tiếp tục của cuộc sống.
sự tồn tại trên hành tinh, nó đảm bảo rằng mạch tồn tại hóa học của Trái Đất đã
được truyền từ cha mẹ đến thế hệ sau, biến đổi và cải thiện dần dần, giúp đỡ
Cuộc sống tiếp tục hàng tỷ năm, bất kể vũ trụ mang đến điều gì, và trong
nhiều trường hợp cho phép cuộc sống của cá nhân tiếp tục lâu hơn nhiều so với họ
thực tế và cần thiết phải.
Cơ thể con người, mặc dù còn xa sự hoàn hảo và vẫn đang tiến hóa, mang theo một
phiên bản nâng cao của mạch sinh tồn cho phép nó tồn tại hàng thập kỷ sau đây
thời kỳ sinh sản. Trong khi thật thú vị để suy nghĩ về lý do tại sao chúng ta có tuổi thọ dài.
Nhu cầu cho ông bà giáo dục bộ tộc là một điều thu hút.
thuyết—với sự hỗn loạn tồn tại ở cấp độ phân tử, thật kỳ diệu khi chúng ta
sống sót trong ba mươi giây, chứ chưa nói đến việc đạt đến tuổi sinh sản, chứ chưa nói đến việc đạt đến
80 nhiều hơn là không.
Nhưng chúng tôi làm được. Kỳ diệu thay, chúng tôi làm được. Kỳ tích thay, chúng tôi làm được. Bởi vì chúng tôi là thế hệ kế thừa.
của một dòng dõi rất dài của những người sống sót vĩ đại. Vậy nên, chúng tôi là những người sống sót vĩ đại.
Nhưng có một sự đánh đổi. Đối với mạch này bên trong chúng ta, hậu duệ của một chuỗi
"Các đột biến ở tổ tiên xa xôi nhất của chúng ta cũng là lý do chúng ta lão hóa."
Và đúng rồi, mạo từ xác định số ít đó là chính xác: đó là lý do.
MỌI THỨ ĐỀU CÓ LÝ DO.
Nếu bạn cảm thấy bất ngờ trước quan niệm rằng có một nguyên nhân duy nhất gây lão hóa, bạn...
không phải cô đơn. Nếu bạn chưa bao giờ suy nghĩ về lý do tại sao chúng ta lão hóa, thì đó là
hoàn toàn bình thường, cũng vậy. Nhiều nhà sinh học cũng không nghĩ nhiều về điều đó.
Ngay cả các nhà lão học, những bác sĩ chuyên về lão hóa, thường không hỏi tại sao chúng ta lão hóa.
—họ chỉ đơn giản cố gắng điều trị các hậu quả.
Điều này không phải là một cận thị chỉ dành cho sự lão hóa. Ngay từ cuối những năm 1960, cho...
Ví dụ, cuộc chiến chống lại ung thư là một cuộc chiến chống lại các triệu chứng của nó. Không có.
Giải thích thống nhất về lý do tại sao ung thư xảy ra, vì vậy các bác sĩ đã loại bỏ các khối u một cách tốt nhất.
Họ có thể và đã dành rất nhiều thời gian để bảo bệnh nhân sắp xếp công việc của họ.
Ung thư là “chỉ là cách nó diễn ra,” vì đó là những gì chúng ta nói khi chúng ta không thể.
giải thích điều gì đó.
Sau đó, vào những năm 1970, các gen gây ra ung thư khi bị đột biến đã được phát hiện bởi
các nhà sinh học phân tử Peter Vogt và Peter Duesberg. Những người được gọi là
Các gen ung thư đã thay đổi hoàn toàn mô hình nghiên cứu ung thư. Dược phẩm
các nhà phát triển giờ đây có mục tiêu để theo đuổi: các protein gây khối u được mã hóa bởi
các gen, chẳng hạn như BRAF, HER2 và BCR-ABL. Bằng cách phát minh ra các hóa chất mà
cụ thể nhằm chặn các protein gây ung thư, chúng tôi có thể cuối cùng bắt đầu di chuyển
ra khỏi việc sử dụng bức xạ và các tác nhân hóa trị độc hại để tấn công ung thư
tại nguồn gốc di truyền của chúng, trong khi để các tế bào bình thường không bị ảnh hưởng. Chúng tôi chắc chắn
không chữa được tất cả các loại ung thư trong nhiều thập kỷ kể từ đó, nhưng chúng tôi không còn
tin rằng không thể làm như vậy.
Thật vậy, trong số ngày càng nhiều nhà nghiên cứu ung thư, sự lạc quan
tràn đầy. Và sự hy vọng đó là trung tâm của điều mà có thể nói là nhất
phần đáng nhớ trong bài phát biểu Tình trạng Liên bang cuối cùng của Tổng thống Barack Obama
2016.
“Cho những người thân yêu mà chúng ta đã mất, cho gia đình mà chúng ta vẫn có thể cứu, hãy cùng nhau hành động.”
"Ông Obama nói: 'Mỹ, đất nước chữa trị ung thư một lần và mãi mãi.'"
trong phòng họp của Hạ viện và kêu gọi một “chiến dịch đột phá chống ung thư.”
Khi ông đặt cựu Phó Tổng thống Joe Biden—người có con trai Beau đã qua đời do
ung thư não một năm trước - đảm nhận nỗ lực, ngay cả một số đảng viên Dân chủ
Các kẻ thù chính trị kiên quyết đã gặp khó khăn trong việc ngăn nước mắt rơi.
Trong những ngày và tuần tiếp theo, nhiều chuyên gia ung thư đã nhận thấy rằng điều này sẽ...
cần nhiều hơn một năm còn lại của chính quyền Obama-Biden để
Chấm dứt ung thư. Rất ít trong số những chuyên gia đó, tuy nhiên, đã nói rằng nó hoàn toàn không thể.
Đã xong. Và đó là vì, trong khoảng vài thập kỷ ngắn ngủi, chúng ta đã hoàn toàn
"đã thay đổi cách chúng ta nghĩ về ung thư. Chúng ta không còn chấp nhận nó nữa."
sự không thể tránh khỏi như một phần của điều kiện con người.
Một trong những đột phá hứa hẹn nhất trong thập kỷ qua là
"Liệu pháp điểm kiểm soát miễn dịch, hay đơn giản là “miễn dịch trị liệu.” Tế bào T miễn dịch"
liên tục và tuần tra cơ thể của chúng ta, tìm kiếm các tế bào nổi loạn để xác định và tiêu diệt trước khi
Chúng có thể nhân lên thành một khối u. Nếu không nhờ có các tế bào T, chúng ta sẽ đều phát triển thành ung thư.
Trong độ tuổi đôi mươi của chúng ta. Nhưng các tế bào ung thư bất lương phát triển những cách để đánh lừa các tế bào T phát hiện ung thư.
"điều đó để họ có thể tiếp tục sinh sôi một cách hạnh phúc. Mới nhất và hiệu quả nhất"
Các liệu pháp miễn dịch gắn vào các protein trên bề mặt của tế bào ung thư.
tương đương với việc lấy lớp áo choàng vô hình của tế bào ung thư để tế bào T có thể nhận ra
và giết họ. Mặc dù chưa đến 10 phần trăm tổng số bệnh nhân ung thư hiện tại
"lợi ích từ liệu pháp miễn dịch, số lượng đó nên tăng lên nhờ vào"
Hàng trăm cuộc thử nghiệm hiện đang diễn ra.
Chúng tôi tiếp tục phản đối một căn bệnh mà trước đây chúng tôi từng chấp nhận như số phận, đổ hàng triệu.
mỗi năm vào nghiên cứu, và nỗ lực đang đạt được thành quả. Tỷ lệ sống sót cho
Các loại ung thư chết người đang gia tăng một cách đột ngột. Cảm ơn sự kết hợp của một
Thuốc ức chế BRAF và liệu pháp miễn dịch, sự sống sót của di căn não do melanoma.
Một trong những loại ung thư chết người nhất, đã tăng 91% kể từ năm 2011.
Giữa năm 1991 và 2016, tổng số ca tử vong do ung thư ở Hoa Kỳ.
giảm 27 phần trăm và tiếp tục giảm. 3 Đó là một chiến thắng được đo bằng hàng triệu sinh mạng.
Nghiên cứu lão hóa ngày nay đang ở giai đoạn tương tự như nghiên cứu ung thư vào những năm 1960.
Chúng tôi có hiểu biết vững chắc về cách lão hóa diễn ra và những tác động của nó đối với chúng ta.
và một thỏa thuận mới nổi về những gì gây ra nó và những gì giữ cho nó không xảy ra.
Như nó trông có vẻ, việc điều trị lão hóa sẽ không quá khó, dễ hơn nhiều so với việc chữa trị.
ung thư
Cho đến nửa sau của thế kỷ hai mươi, nó được chấp nhận chung.
các sinh vật già đi và chết "vì lợi ích của loài"—một ý tưởng rằng
có từ thời Aristotle, nếu không muốn nói là còn xa hơn. Ý tưởng này có vẻ khá trực quan. Đây là
Giải thích được cung cấp bởi hầu hết mọi người ở các bữa tiệc. Nhưng nó hoàn toàn sai. Chúng tôi không làm.
không chết để nhường chỗ cho thế hệ tiếp theo.
Vào thập niên 1950, khái niệm "lựa chọn nhóm" trong tiến hóa đang dần bị lãng quên.
phong cách, gợi ý ba nhà sinh học tiến hóa, J. B. S. Haldane, Peter B.
Medawar và George C. Williams đã đề xuất một số ý tưởng quan trọng về lý do tại sao
Chúng ta già đi. Khi nói đến tuổi thọ, họ đồng ý rằng các cá nhân quan tâm đến
chính họ. Bị thúc đẩy bởi những gen vị kỷ của mình, họ tiếp tục tiến lên và cố gắng sinh sản để đạt được
dài và nhanh nhất có thể, miễn là nó không giết họ. (Trong một số trường hợp,
Tuy nhiên, họ thúc ép quá nhiều, như tổ tiên vĩ đại của tôi, Miklós Vitéz, một
Nhà biên kịch Hungary đã chứng minh cho cô dâu của mình, kém ông bốn mươi lăm tuổi, trong ngày cưới của họ.
đêm tân hôn.)
"Nếu gen của chúng ta không bao giờ muốn chết, tại sao chúng ta không sống mãi mãi? Bộ ba của"
Các nhà sinh vật học lập luận rằng chúng ta trải qua lão hóa vì các lực của chọn lọc tự nhiên.
Cần thiết để xây dựng một cơ thể khỏe mạnh có thể mạnh mẽ khi chúng ta 18 tuổi nhưng sẽ suy giảm.
nhanh chóng khi chúng tôi đạt 40, vì khi đó chúng tôi có thể đã nhân bản các gen sel sh của mình trong
biện pháp đủ để đảm bảo sự tồn tại của họ. Cuối cùng, các lực lượng tự nhiên
Lựa chọn đạt đến số không. Các gen tiếp tục tiến bước. Chúng ta thì không.
Medawar, người có sở thích với lời lẽ dài dòng, đã trình bày về một lý thuyết tinh vi.
được gọi là "đa hình đối kháng." Nói đơn giản, nó nói rằng các gen giúp chúng ta sinh sản
Khi chúng ta trẻ, không chỉ trở nên ít hữu ích hơn khi già đi, họ thực sự có thể...
trở lại cắn chúng ta khi chúng ta già.
Hai mươi năm sau, Thomas Kirkwood tại Đại học Newcastle đã hình thành ý tưởng về
câu hỏi về lý do tại sao chúng ta lão hóa liên quan đến nguồn lực có sẵn của một sinh vật. Được biết đến với tên gọi
Giả thuyết "Soma có thể tiêu hủy," nó dựa trên thực tế rằng luôn luôn có
tài nguyên hạn chế có sẵn cho các loài—năng lượng, dinh dưỡng, nước. Do đó, chúng
tiến hóa đến mức nằm ở đâu đó giữa hai lối sống rất khác biệt: giống nòi
"Sống nhanh và chết trẻ, hoặc sinh sản chậm và bảo vệ soma, hay cơ thể của bạn. Kirkwood"
lập luận rằng các sinh vật không thể sinh sản nhanh và duy trì một cơ thể khỏe mạnh, vững chắc—
Không đủ năng lượng để làm cả hai. Nói cách khác, trong lịch sử.
của cuộc sống, bất kỳ loài sinh vật nào có đột biến khiến nó sống nhanh và cố gắng
"để chết già sớm đã cạn kiệt tài nguyên và do đó đã bị xóa khỏi nguồn gen."
Lý thuyết của Kirkwood được minh họa tốt nhất bằng những tình huống hư cấu nhưng có thể là thực tế.
Ví dụ. Hãy tưởng tượng bạn là một loài gặm nhấm nhỏ có khả năng bị một con chim bắt được.
của con mồi. Vì lý do này, bạn sẽ cần phải truyền lại vật chất di truyền của mình một cách nhanh chóng,
cũng như cha mẹ của bạn và ông bà của họ trước đây. Các tổ hợp gen mà
sẽ cung cấp một cơ thể bền lâu hơn nếu không được làm giàu trong loài của bạn
bởi vì tổ tiên của bạn có thể đã không thoát khỏi sự săn mồi lâu dài (và bạn cũng sẽ không).
hoặc).
Bây giờ hãy tưởng tượng rằng bạn là một loài chim săn mồi ở đỉnh của chuỗi thức ăn.
Vì lý do này, gen của bạn—thực ra là gen của tổ tiên bạn—đã được hưởng lợi.
từ việc xây dựng một cơ thể chắc khỏe, lâu dài hơn có thể sinh sản trong nhiều thập kỷ. Nhưng trong
trở lại, họ chỉ có thể đủ khả năng nuôi chỉ một vài con chim non mỗi năm.
Giả thuyết của Kirkwood giải thích tại sao một con chuột sống được 3 năm trong khi một số loài chim...
có thể sống đến 100,5. Nó cũng giải thích một cách khá tinh tế lý do tại sao tắc kè hoa Mỹ.
Thằn lằn, Anolis carolinensis, đang tiến hóa một tuổi thọ dài hơn ngay khi chúng ta nói, đã tìm thấy
một vài thập kỷ trước trên những hòn đảo xa xôi của Nhật Bản mà không có kẻ săn mồi. 6
Những lý thuyết này phù hợp với các quan sát và được chấp nhận rộng rãi. Các cá nhân
Đừng sống mãi mãi vì chọn lọc tự nhiên không chọn sự bất tử.
thế giới mà một kế hoạch cơ thể hiện có hoạt động hoàn hảo để truyền đạt cơ thể
Chọn các gen sh. Và vì tất cả các loài đều có giới hạn về tài nguyên, chúng đã tiến hóa để...
cung cấp năng lượng có sẵn cho sinh sản hoặc cho tuổi thọ, nhưng không cho
cả hai. Điều đó đúng với M. superstes cũng như với tất cả các loài khác mà
đã từng sống trên hành tinh này.
Al, tức là, ngoại trừ một: Homo sapiens.
Tận dụng bộ não tương đối lớn và một nền văn minh phát triển để
vượt qua bàn tay không may mà tiến hóa đã ban tặng—các chi yếu ớt, nhạy cảm
đến lạnh, khứu giác kém, và đôi mắt chỉ nhìn thấy rõ vào ban ngày và trong
quang phổ nhìn thấy - loài rất đặc biệt này tiếp tục đổi mới. Nó có
đã tự cung cấp cho mình một nguồn thực phẩm, dinh dưỡng và nước dồi dào trong khi
giảm tử vong do bị săn mồi, tiếp xúc, bệnh truyền nhiễm và chiến tranh.
Điều này từng là giới hạn cho sự phát triển của nó thành một tuổi thọ dài hơn. Khi chúng được loại bỏ, một...
Vài triệu năm tiến hóa có thể gấp đôi tuổi thọ của nó, đưa nó lại gần hơn với.
tuổi thọ của một số loài khác ở đỉnh cao của chúng. Nhưng sẽ không phải chờ đợi.
Cái đó dài, không gần như vậy. Bởi vì loài này đang nỗ lực làm việc để phát minh.
thuốc và công nghệ để mang lại độ bền vững như một sản phẩm có tuổi thọ lâu hơn.
nghĩa đen và vượt qua những gì tiến hóa đã không cung cấp.
CHẾ ĐỘ KHỦNG HOẢNG
Wilbur và Orvil e Wright không bao giờ có thể chế tạo một máy bay nếu không có một
kiến thức về dòng khí và áp suất âm cũng như một đường hầm gió. Cũng không thể
Hoa Kỳ đã đặt người lên mặt trăng mà không có sự hiểu biết về
kim loại nóng chảy, sự cháy lỏng, máy tính, và một số mức độ tự tin rằng
Mặt trăng không được làm bằng phô mai xanh. 7
Tương tự, nếu chúng ta muốn đạt được tiến bộ thực sự trong nỗ lực giảm bớt sự...
Đau khổ đi kèm với tuổi tác, cái cần thiết là một lời giải thích thống nhất cho lý do tại sao.
Chúng ta lão hóa, không chỉ ở cấp độ tiến hóa mà còn ở cấp độ cơ bản.
Nhưng việc giải thích quá trình lão hóa ở mức độ cơ bản không phải là một nhiệm vụ dễ dàng. Nó sẽ phải
đáp ứng tất cả các luật đã biết của vật lý và tất cả các quy tắc của hóa học và nhất quán với
"nhiều thế kỷ quan sát sinh học. Nó sẽ cần trải qua những điều ít được hiểu nhất."
thế giới giữa kích thước của một phân tử và kích thước của một hạt cát, 8 và nó nên giải thích đồng thời sự sống đơn giản nhất và phức tạp nhất.
máy móc đã từng tồn tại.
Do đó, không có gì ngạc nhiên khi chưa bao giờ có một sự thống nhất.
Lý thuyết về sự lão hóa, ít nhất là không có một lý thuyết nào được chứng minh - mặc dù không phải vì thiếu sự cố gắng.
Một giả thuyết, được đề xuất độc lập bởi Peter Medawar và Leo Szilard,
là việc lão hóa do tổn thương DNA và mất mát di truyền gây ra.
thông tin. Khác với Medawar, người luôn là một nhà sinh vật học, người đã xây dựng một Giải Nobel
Sự nghiệp đoạt giải thưởng trong lĩnh vực miễn dịch học, Szilard đã đến để học sinh vật học ở một
theo cách vòng. Nhà đa tài và phát minh sinh ra ở Budapest sống một cuộc sống du mục.
cuộc sống không có công việc hoặc địa chỉ cố định, thích dành thời gian ở bên
các đồng nghiệp đã thỏa mãn những tò mò về tâm lý của ông về những câu hỏi lớn mà ông đang đối diện
nhân loại. Đầu sự nghiệp của mình, ông là một nhà vật lý hạt nhân tiên phong và một
nhà hợp tác sáng lập trong Dự án Manhattan, đưa vào kỷ nguyên của
chiến tranh hạt nhân. Kinh hoàng trước vô số sinh mạng mà công việc của ông đã góp phần kết thúc, ông
"xoay đầu óc đau khổ của anh ấy về việc làm cho cuộc sống dài tối đa."
Ý tưởng rằng sự tích lũy đột biến gây ra lão hóa đã được các nhà khoa học chấp nhận.
và công chúng giống nhau vào những năm 1950 và 1960, vào thời điểm khi những ảnh hưởng của
"Bức xạ lên DNA con người đang là mối quan tâm của rất nhiều người."
biết chắc chắn rằng bức xạ có thể gây ra đủ loại vấn đề trong chúng ta
Điều đó chỉ gây ra một tập hợp con của các dấu hiệu và triệu chứng mà chúng ta quan sát trong suốt.
lão hóa, 10 vì vậy nó không thể đóng vai trò như một lý thuyết phổ quát.
Vào năm 1963, nhà sinh học người Anh Leslie Orgel đã tham gia vào cuộc tranh luận với...
"Hypothesis về Thảm họa Lỗi," giả thuyết rằng những sai lầm xảy ra trong quá trình
Quá trình sao chép DNA dẫn đến các đột biến trong gen, bao gồm cả những gen cần thiết để
tạo ra các máy móc protein sao chép DNA. Quá trình này ngày càng gây gián đoạn
các quá trình đó, nhân lên trên chính chúng cho đến khi bộ gen của một người đã
đã bị sao chép sai và rơi vào quên lãng. 11
Vào khoảng thời gian mà Szilard đang tập trung vào bức xạ, Denham
Harman, một nhà hóa học tại Shel Oil, cũng đang suy nghĩ theo cách nguyên tử, mặc dù theo cách khác.
cách. Sau khi dành thời gian hoàn thành trường y tại Đại học Stanford, anh ấy
đã đưa ra "Lý thuyết Tự do gốc của Lão hóa," cho rằng lão hóa là do
các electron đơn lẻ bay xung quanh trong các tế bào, gây hại cho DNA thông qua
Sự oxy hóa, đặc biệt là ở ti thể, vì đó là nơi hầu hết các gốc tự do xuất hiện.
được tạo ra. Harman đã dành phần lớn cuộc đời để thử nghiệm lý thuyết.
Tôi đã có dịp gặp gỡ gia đình Harman vào năm 2013. Vợ anh ấy đã nói với tôi.
Giáo sư Harman đã sử dụng liều cao axit alpha-lipoic trong phần lớn thời gian.
của cuộc sống của anh ấy để làm dịu các gốc tự do. Xét rằng anh ấy đã làm việc không mệt mỏi với
Nghiên cứu thậm chí có thể kéo dài đến những năm 90, tôi đoán, ít nhất thì cũng chẳng hại gì.
Trong những năm 1970 và 1980, Harman và hàng trăm các nhà nghiên cứu khác
đã kiểm tra xem các chất chống oxy hóa có kéo dài tuổi thọ của động vật hay không. Kết quả
Tổng thể đều gây thất vọng. Mặc dù Harman có một số thành công trong việc tăng cường...
tuổi thọ trung bình của gặm nhấm, chẳng hạn như với phụ gia thực phẩm butylated
hydroxytoluene, không có loại nào cho thấy sự gia tăng trong thời gian sống tối đa. Nói cách khác,
một nhóm động vật nghiên cứu có thể sống lâu hơn vài tuần, trung bình, nhưng không ai trong số đó
Các loài động vật đang lập kỷ lục về tuổi thọ cá nhân. Khoa học kể từ đó...
đã chứng minh rằng những tác động tích cực đến sức khỏe có thể đạt được từ một chế độ ăn giàu chất chống oxy hóa
chế độ ăn uống có nhiều khả năng được gây ra bởi việc kích thích các cơ chế phòng thủ tự nhiên của cơ thể chống lại
lão hóa, bao gồm việc tăng cường sản xuất các enzyme của cơ thể làm tiêu diệt
các gốc tự do, không phải do hoạt động của chất chống oxy hóa tự nó.
Nếu thói quen cũ khó bỏ, thì ý tưởng về gốc tự do giống như heroin. Lý thuyết là
bị các nhà khoa học bác bỏ trong các phòng thí nghiệm của tôi hơn một thập kỷ trước
tuy nhiên nó vẫn được duy trì rộng rãi bởi những người cung cấp thuốc viên và đồ uống, những người tiếp tay cho một thị trường trị giá 3 đô la
ngành công nghiệp toàn cầu. 13 Với tất cả những quảng cáo đó, không có gì ngạc nhiên khi nhiều
Hơn 60% người tiêu dùng Mỹ vẫn tìm kiếm thực phẩm và đồ uống tốt cho sức khỏe.
nguồn của chất chống oxy hóa.
Các gốc tự do thực sự gây ra sự đột biến. Chắc chắn là như vậy. Bạn có thể tìm thấy sự đột biến.
trong sự phong phú, đặc biệt là trong các tế bào tiếp xúc với thế giới bên ngoài và trong
genomes ti thể của những cá thể già. Sự suy giảm ti thể chắc chắn
một dấu hiệu lão hóa và có thể dẫn đến suy chức năng cơ quan. Nhưng chỉ riêng các đột biến,
đặc biệt và đột biến trong bộ gen hạt nhân, xung đột với một sự gia tăng không ngừng.
số lượng bằng chứng ngược lại
Arlan Richardson và Holly Van Remmen đã dành khoảng một thập kỷ tại
Đại học Texas tại San Antonio kiểm tra xem việc tăng cường tổn thương gốc tự do có...
Các đột biến ở chuột dẫn đến sự lão hóa; nó đã không. Trong phòng thí nghiệm của tôi và những nơi khác, điều này đã được chứng minh.
thật ngạc nhiên là đơn giản để phục hồi chức năng của ty thể ở chuột già
ngụ ý rằng một phần lớn của quá trình lão hóa không phải do đột biến ở ti thể.
DNA, ít nhất không cho đến cuối đời. 17
Mặc dù cuộc thảo luận về vai trò của đột biến DNA hạt nhân trong quá trình lão hóa
tiếp tục, có một thực tế mâu thuẫn với tất cả những lý thuyết này, một cái mà khó khăn.
bác bỏ
Nghịch lý thay, chính Szilard, vào năm 1960, đã khởi xướng sự sụp đổ của lý thuyết của chính ông.
bằng cách tìm hiểu cách sao chép một tế bào người. 18 Sao chép cung cấp cho chúng ta câu trả lời cho
liệu rằng các đột biến có gây ra sự lão hóa hay không. Nếu các tế bào cũ thực sự đã mất đi những gen quan trọng
Thông tin này là nguyên nhân của sự lão hóa, chúng ta không nên có khả năng sao chép mới.
động vật từ những cá nhân già hơn. Các bản sao sẽ được sinh ra với tuổi già.
Đó là một sự hiểu lầm rằng động vật được nhân bản lão hóa sớm. Nó đã được phổ biến rộng rãi
"được duy trì trong phương tiện truyền thông và ngay cả trang web của Viện Y tế Quốc gia cũng nói"
Vậy, đúng là Dol y, con cừu được nhân bản đầu tiên, được tạo ra bởi Keith Campbell.
và Ian Wilmut tại Viện Roslin thuộc Đại học Edinburgh, đã sống
chỉ sống được nửa tuổi thọ bình thường và đã chết vì một bệnh phổi tiến triển. Nhưng rộng rãi
Phân tích di thể của cô ấy không cho thấy dấu hiệu của sự lão hóa sớm. 20 Trong khi đó, danh sách
các loài động vật đã được nhân bản và được chứng minh là sống khỏe mạnh, bình thường
Tuổi thọ hiện nay bao gồm dê, cừu, chuột và bò. 21
Vì thực tế rằng chuyển giao hạt nhân hoạt động trong nhân bản, chúng ta có thể nói rằng
mức độ tin cậy cao rằng lão hóa không phải do các đột biến trong DNA nhân.
Chắc chắn, có thể một số tế bào trong cơ thể không đột biến và đó là những tế bào.
Điều đó cuối cùng dẫn đến việc tạo ra những bản sao thành công, nhưng điều đó có vẻ rất không chắc xảy ra.
Giải thích đơn giản nhất là những con vật già giữ lại tất cả gen cần thiết.
thông tin để tạo ra một động vật hoàn toàn mới, khỏe mạnh và rằng các đột biến là
không phải là nguyên nhân chính gây lão hóa.22
Chắc chắn không có sự nhục nhã nào đối với những nhà nghiên cứu xuất sắc đó rằng các lý thuyết của họ
không chịu được thử thách của thời gian. Đó là điều xảy ra với hầu hết các ngành khoa học, và
có lẽ tất cả sẽ cuối cùng. Trong Cấu trúc của các cuộc cách mạng khoa học, Thomas
Kuhn đã lưu ý rằng phát hiện khoa học không bao giờ hoàn chỉnh; nó trải qua.
predictable stages of evolution. When a theory succeeds at explaining previously
unexplainable observations about the world, it becomes a tool that scientists can
use to discover even more.
Inevitably, however, new discoveries lead to new questions that are not
entirely answerable by the theory, and those questions beget more questions.
Soon the model enters crisis mode and begins to drift as scientists seek to adjust
it, as little as possible, to account for that which it cannot explain.
Crisis mode is always a fascinating time in science but one that is not for the
faint of heart, as doubts about the views of previous generations continue to
grow against the old guard’s protestations. But the chaos is ultimately replaced
by a paradigm shift, one in which a new consensus model emerges that can
explain more than the previous model.
That’s what happened about a decade ago, as the ideas of leading scientists inthe aging eld began to coalesce around a new model—one that suggested that
the reason so many bril iant people had struggled to identify a single cause of
aging was that there wasn’t one.
In this more nuanced view, aging and the diseases that come with it are the
result of multiple “hal marks” of aging:
• Genomic instability caused by DNA damage
• Attrition of the protective chromosomal endcaps, the telomeres
• Alterations to the epigenome that controls which genes are turned on and
off
• Loss of healthy protein maintenance, known as proteostasis
• Deregulated nutrient sensing caused by metabolic changes
• Mitochondrial dysfunction
• Accumulation of senescent zombielike cel s that inflame healthy cel s
• Exhaustion of stem cel s
• Altered intercel ular communication and the production of inflammatory
molecules
Researchers began to cautiously agree: address these hal marks, and you can
slow down aging. Slow down aging, and you can forestal disease. Forestal
disease, and you can push back death.
Take stem cel s, which have the potential to develop into many other kinds of
cel s: if we can keep these undi erentiated cel s from tiring out, they can
continue to generate al the di erentiated cel s necessary to heal damaged tissues
and battle al kinds of diseases.
Meanwhile, we’re improving the rates of acceptance of bone marrow
transplants, which are the most common form of stem cel therapy, and using
stem cel s for the treatment of arthritic joints, type 1 diabetes, loss of vision, and
neurodegenerative diseases such as Alzheimer’s and Parkinson’s. These stem
cel –based interventions are adding years to people’s lives.
Or take senescent cel s, which have reached the end of their ability to divide
but refuse to die, continuing to spit out panic signals that in ame surrounding
cel s: if we can kil o senescent cel s or keep them from accumulating in the rstplace, we can keep our tissues much healthier for longer.
The same can be said for combating telomere loss, the decline in proteostasis,
and al of the other hal marks. Each can be addressed one by one, a little at a
time, in ways that can help us extend human healthspans.
Over the past quarter century, researchers have increasingly homed their
e orts in on addressing each of these hal marks. A broad consensus formed that
this would be the best way to al eviate the pain and su ering of those who are
aging.
There is little doubt that the list of hal marks, though incomplete, comprises
the beginnings of a rather strong tactical manual for living longer and healthier
lives. Interventions aimed at slowing any one of these hal marks may add a few
years of wel ness to our lives. If we can address al of them, the reward could be
vastly increased average lifespans.
As for pushing way past the maximum limit? Addressing these hal marks
might not be enough.
But the science is moving fast, faster now than ever before, thanks to the
accumulation of many centuries of knowledge, robots that analyze tens of
thousands of potential drugs each day, sequencing machines that read mil ions
of genes a day, and computing power that processes tril ions of bytes of data at
speeds that were unimaginable just a decade ago. Theories on aging, which were
slowly chipped away for decades, are now more easily testable and refutable.
Although it is in its early days, a new shift in thinking is again under way.
Once again we nd ourselves in a period of chaos—stil quite con dent that the
hal marks are accurate indicators of aging and its myriad symptoms but unable
to explain why the hal marks occur in the rst place.
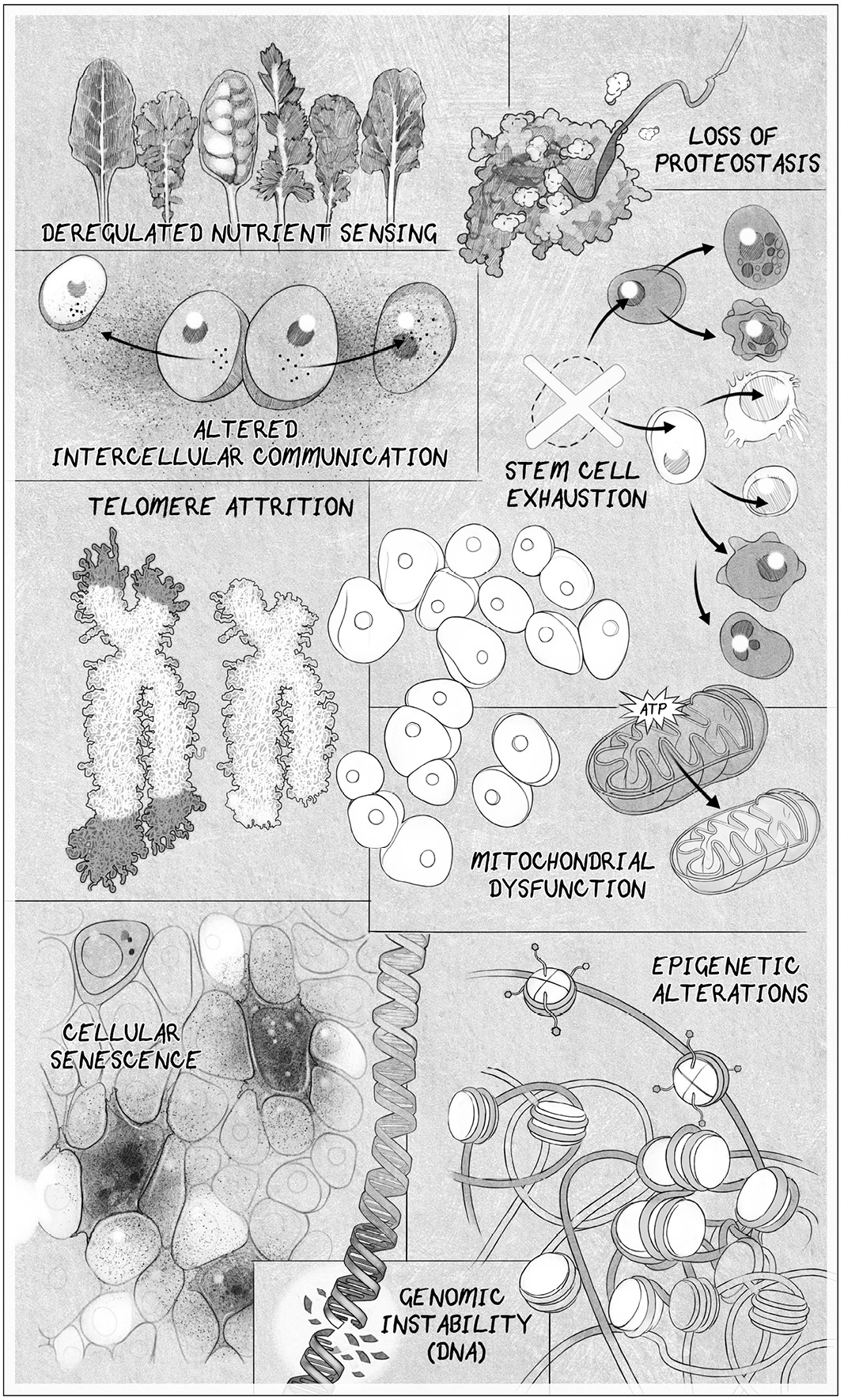
THE HALLMARKS OF AGING. Scientists have settled on eight or nine hal marks of aging. Address
one of these, and you can slow down aging. Address al of them, and you might not age.
It is time for an answer to this very old question.
Now, nding a universal explanation for anything—let alone something as
complicated as aging—doesn’t happen overnight. Any theory that seeks to
explain aging must not just stand up to scienti c scrutiny but provide a rational
explanation for every one of the pil ars of aging. A universal hypothesis that
seems to provide a reason for cel ular senescence but not stem cel exhaustion
would, for example, explain neither.
Yet I believe that such an answer exists—a cause of aging that exists upstream
of al the hal marks. Yes, a singular reason why we age.
Aging, quite simply, is a loss of information.
You might recognize that loss of information was a big part of the ideas that
Szilard and Medawar independently espoused, but it was wrong because it
focused on a loss of genetic information.
But there are two types of information in biology, and they are encoded
entirely di erently. The rst type of information—the type my esteemed
predecessors understood—is digital. Digital information, as you likely know, isbased on a nite set of possible values—in this instance, not in base 2 or binary,
coded as 0s and 1s, but the sort that is quaternary or base 4, coded as adenine,
thymine, cytosine, and guanine, the nucleotides A, T, C, G of DNA.
Because DNA is digital, it is a reliable way to store and copy information.
Indeed, it can be copied again and again with tremendous accuracy, no di erent
in principle from digital information stored in computer memory or on a DVD.
DNA is also robust. When I rst worked in a lab, I was shocked by how this
“molecule of life” could survive for hours in boiling water and thril ed that it was
recoverable from Neanderthal remains at least 40,000 years old. 23 The
advantages of digital storage explain why chains of nucleic acids have remained
the go-to biological storage molecule for the past 4 bil ion years.
The other type of information in the body is analog.
We don’t hear as much about analog information in the body. That’s in part
because it’s newer to science, and in part because it’s rarely described in terms of
information, even though that’s how it was rst described when geneticists
noticed strange nongenetic e ects in plants they were breeding.
Today, analog information is more commonly referred to as the epigenome,
meaning traits that are heritable that aren’t transmitted by genetic means.
The term epigenetics was rst coined in 1942 by Conrad H. Waddington, a
British developmental biologist, while working at Cambridge University. In the
past decade, the meaning of the word epigenetics has expanded into other areas
of biology that have less to do with heredity—including embryonic
development, gene switch networks, and chemical modi cations of DNA-
packaging proteins—much to the chagrin of orthodox geneticists in my
department at Harvard Medical School.
In the same way that genetic information is stored as DNA, epigenetic
information is stored in a structure cal ed chromatin. DNA in the cel isn’t
ailing around disorganized, it is wrapped around tiny bal s of protein cal ed
histones. These beads on a string self-assemble to form loops, as when you tidy
your garden hose on your driveway by looping it into a pile. If you were to play
tug-of-war using both ends of a chromosome, you’d end up with a six foot-long
string of DNA punctuated by thousands of histone proteins. If you could
somehow plug one end of the DNA into a power socket and make the histonesash on and o , a few cel s could do you for holiday lights.
In simple species, like ancient M. superstes and fungi today, epigenetic
information storage and transfer is important for survival. For complex life, it is
essential. By complex life, I mean anything made up of more than a couple of
cel s: slime molds, jel y sh, worms, fruit ies, and of course mammals like us.
Epigenetic information is what orchestrates the assembly of a human newborn
made up of 26 bil ion cel s from a single fertilized egg and what al ows the
genetical y identical cel s in our bodies to assume thousands of di erent
modalities.24
If the genome were a computer, the epigenome would be the software. It
instructs the newly divided cel s on what type of cel s they should be and what
they should remain, sometimes for decades, as in the case of individual brain
neurons and certain immune cel s.
That’s why a neuron doesn’t one day behave like a skin cel and a dividing
kidney cel doesn’t give rise to two liver cel s. Without epigenetic information,
cel s would quickly lose their identity and new cel s would lose their identity,
too. If they did, tissues and organs would eventual y become less and less
functional until they failed.
In the warm ponds of the primordial Earth, a digital chemical system was the
best way to store long-term genetic data. But information storage was also
needed to record and respond to environmental conditions, and this was best
stored in analog format. Analog data are superior for this job because they can be
changed back and forth with relative ease whenever the environment within or
outside the cel demands it, and they can store an almost unlimited number of
possible values, even in response to conditions that have never been encountered
before. 25
The unlimited number of possible values is why many audiophiles stil prefer
the rich sounds of analog storage systems. But even though analog devices have
their advantages, they have a major disadvantage. In fact, it’s the reason we’ve
moved from analog to digital. Unlike digital, analog information degrades over
time—fal ing victim to the conspiring forces of magnetic elds, gravity, cosmic
rays, and oxygen. Worse stil , information is lost as it’s copied.
No one was more acutely disturbed by the problem of information loss thanClaude Shannon, an electrical engineer from the Massachusetts Institute of
Technology (MIT) in Boston. Having lived through World War II, Shannon
knew rsthand how the introduction of “noise” into analog radio transmissions
could cost lives. After the war, he wrote a short but profound scienti c paper
cal ed “The Mathematical Theory of Communication” on how to preserve
information, which many consider the foundation of Information Theory. If
there is one paper that propel ed us into the digital, wireless world in which we
now live, that would be it. 26
Shannon’s primary intention, of course, was to improve the robustness of
electronic and radio communications between two points. His work may
ultimately prove to be even more important than that, for what he discovered
about preserving and restoring information, I believe, can be applied to aging.
Don’t be disheartened by my claim that we are the biological equivalent of an
old DVD player. This is actual y good news. If Szilard had turned out to be right
about mutations causing aging, we would not be able to easily address it, because
when information is lost without a backup, it is lost for good. Ask anyone who’s
tried to play or restore content from a DVD that’s had an edge broken o : what
is gone is gone.
But we can usual y recover information from a scratched DVD. And if I am
right, the same kind of process is what it wil take to reverse aging.
As cloning beautiful y proves, our cel s retain their youthful digital
information even when we are old. To become young again, we just need to nd
some polish to remove the scratches.
This, I believe, is possible.
A TIME TO EVERY PURPOSE
The Information Theory of Aging starts with the primordial survival circuit we
inherited from our distant ancestors.
Over time, as you might expect, the circuit has evolved. Mammals, for
instance, don’t have just a couple of genes that create a survival circuit, such as
those that rst appeared in M. superstes. Scientists have found more than twodozen of them within our genome. Most of my col eagues cal these “longevity
genes” because they have demonstrated the ability to extend both average and
maximum lifespans in many organisms. But these genes don’t just make life
longer, they make it healthier, which is why they can also be thought of as
“vitality genes.”
Together, these genes form a surveil ance network within our bodies,
communicating with one another between cel s and between organs by releasing
proteins and chemicals into the bloodstream, monitoring and responding to
what we eat, how much we exercise, and what time of day it is. They tel us to
hunker down when the going gets tough, and they tel us to grow fast and
reproduce fast when the going gets easier.
And now that we know these genes are there and what many of them do,
scienti c discovery has given us an opportunity to explore and exploit them; to
imagine their potential; to push them to work for us in di erent ways. Using
molecules both natural and novel, using technology both simple and complex,
using wisdom both new and old, we can read them, turn them up and down,
and even change them altogether.
The longevity genes I work on are cal ed “sirtuins,” named after the yeast
SIR2 gene, the rst one to be discovered. There are seven sirtuins in mammals,
SIRT1 to SIRT7, and they are made by almost every cel in the body. When I
started my research, sirtuins were barely on the scienti c radar. Now this family
of genes is at the forefront of medical research and drug development.
Descended from gene B in M. superstes, sirtuins are enzymes that remove
acetyl tags from histones and other proteins and, by doing so, change the
packaging of the DNA, turning genes o and on when needed. These critical
epigenetic regulators sit at the very top of cel ular control systems, control ing
our reproduction and our DNA repair. After a few bil ion years of advancement
since the days of yeast, they have evolved to control our health, our tness, and
our very survival. They have also evolved to require a molecule cal ed
nicotinamide adenine dinucleotide, or NAD. As we wil see later, the loss of
NAD as we age, and the resulting decline in sirtuin activity, is thought to be a
primary reason our bodies develop diseases when we are old but not when we areyoung.
Trading reproduction for repair, the sirtuins order our bodies to “buckle
down” in times of stress and protect us against the major diseases of aging:
diabetes and heart disease, Alzheimer’s disease and osteoporosis, even cancer.
They mute the chronic, overactive in ammation that drives diseases such as
atherosclerosis, metabolic disorders, ulcerative colitis, arthritis, and asthma.
They prevent cel death and boost mitochondria, the power packs of the cel .
They go to battle with muscle wasting, osteoporosis, and macular degeneration.
In studies on mice, activating the sirtuins can improve DNA repair, boost
memory, increase exercise endurance, and help the mice stay thin, regardless of
what they eat. These are not wild guesses as to their power; scientists have
established al of this in peer-reviewed studies published in journals such as
Nature, Cell, and Science.
And in no smal measure, because sirtuins do al of this based on a rather
simple program—the wondrous gene B in the survival circuit—they’re turning
out to be more amenable to manipulation than many other longevity genes.
They are, it would appear, one of the rst dominos in the magni cent Rube
Goldberg machine of life, the key to understanding how our genetic material
protects itself during times of adversity, al owing life to persist and thrive for
bil ions of years.
Sirtuins aren’t the only longevity genes. Two other very wel studied sets of
genes perform similar roles, which also have been proven to be manipulable in
ways that can o er longer and healthier lives.
One of these is cal ed target of rapamycin, or TOR, a complex of proteins
that regulates growth and metabolism. Like sirtuins, scientists have found TOR
—cal ed mTOR in mammals—in every organism in which they’ve looked for it.
Like that of sirtuins, mTOR activity is exquisitely regulated by nutrients. And
like the sirtuins, mTOR can signal cel s in stress to hunker down and improve
survival by boosting such activities as DNA repair, reducing in ammation
caused by senescent cel s, and, perhaps its most important function, digesting
old proteins. 27
When al is wel and ne, TOR is a master driver of cel growth. It senses theamount of amino acids that is available and dictates how much protein is created
in response. When it is inhibited, though, it forces cel s to hunker down,
dividing less and reusing old cel ular components to maintain energy and extend
survival—sort of like going to the junkyard to nd parts with which to x up an
old car rather than buying a new one, a process cal ed autophagy. When our
ancestors were unsuccessful in bringing down a wool y mammoth and had to
survive on meager rations of protein, it was the shutting down of mTOR that
permitted them to survive.
The other pathway is a metabolic control enzyme known as AMPK, which
evolved to respond to low energy levels. It has also been highly conserved among
species and, as with sirtuins and TOR, we have learned a lot about how to
control it.
These defense systems are al activated in response to biological stress. Clearly,
some stresses are simply too great to overcome—step on a snail, and its days are
over. Acute trauma and uncontrol able infections wil kil an organism without
aging that organism. Sometimes the stress inside a cel , such as a multitude of
DNA breaks, is too much to handle. Even if the cel is able to repair the breaks in
the short term without leaving mutations, there is information loss at the
epigenetic level.
Here’s the important point: there are plenty of stressors that wil activate
longevity genes without damaging the cel , including certain types of exercise,
intermittent fasting, low-protein diets, and exposure to hot and cold
temperatures (I discuss this in chapter 4). That’s cal ed hormesis. 28 Hormesis is
general y good for organisms, especial y when it can be induced without causing
any lasting damage. When hormesis happens, al is wel . And, in fact, al is better
than wel , because the little bit of stress that occurs when the genes are activated
prompts the rest of the system to hunker down, to conserve, to survive a little
longer. That’s the start of longevity.
Complementing these approaches are hormesis-mimicking molecules. Drugs
in development and at least two drugs on the market can turn on the body’s
defenses without creating any damage. It’s like making a prank cal to the
Pentagon. The troops and the Army Corps of Engineers are sent out, but there’s
no war. In this way, we can mimic the bene ts of exercise and intermittentfasting with a single pil (I discuss this in chapter 5).
Our ability to control al of these genetic pathways wil fundamental y
transform medicine and the shape of our everyday lives. Indeed, it wil change
the way we de ne our species.
And yes, I realize how that sounds. So let me explain why.
TWOTHE DEMENTED PIANIST
ON APRIL 15, 2003, NEWSPAPERS, television programs, and websites around the
world carried the story: the mapping of the human genome was complete.
There was just one pesky problem: it real y wasn’t. There were, in fact, huge
gaps in the sequence.
This wasn’t a case of the mainstream news media blowing things out of
proportion. Highly respected scienti c journals such as Science and Nature told
pretty much the same story. It also wasn’t a case of scientists overstating their
work. The truth is simply that, at the time, most researchers involved in the
thirteen-year, $1 bil ion project agreed that we’d come as close as we possibly
could—given the technology of the time—to identifying each of the 3 bil ion
base pairs in our DNA.
The parts of the genome that were missing, general y overlapping sections of
repetitive nucleotides, were just not considered important. These were areas of
the code of life that were once derided as “junk DNA” and that are now a little
better respected but stil general y disregarded as “noncoding.” From the
perspective of many of the best minds in science at the time, those regions were
little more than the ghosts of genomes past, mostly remnants of dead
hitchhiking viruses that had integrated into the genome hundreds of thousands
of years ago. The stu that makes us who we are, it was thought, had largely
been identi ed, and we had what we needed to propel forward ourunderstanding of what makes us human.
Yet by some estimates, that genetic dark matter accounts for as much as 69
percent of the total genome, 1 and even within the regions general y regarded as
“coding,” some scientists believe, up to 10 percent has yet to be decoded,
including regions that impact aging. 2
In the relatively short time that has come and gone since 2003, we have come
to nd out that within the famous double helix, there were sequences that were
not just unmapped but essential to our lives. Indeed, many thousands of
sequences had gone undetected because the original algorithms to detect genes
were written to disregard any gene less than 300 base pairs long. In fact, genes
can be as short as 21 base pairs, and today we’re discovering hundreds of them al
over the genome.
These genes tel our cel s to create speci c proteins, and these proteins are the
building blocks of the processes and traits that constitute human biology and
lived experiences. And as we get closer to identifying a complete sequence of our
DNA, we’ve come closer to having a “map” of the genes that control so much of
our existence.
Even once we have a complete code, though, there’s something we stil won’t
be able to nd.
We won’t be able to nd an aging gene.
We have found genes that impact the symptoms of aging. We’ve found
longevity genes that control the body’s defenses against aging and thus o er a
path to slowing aging through natural, pharmaceutical, and technological
interventions. But unlike the oncogenes that were discovered in the 1970s and
that have given us a good target for going to battle against cancer, we haven’t
identi ed a singular gene that causes aging. And we won’t.
Because our genes did not evolve to cause aging.
YEAST OF EDEN
My journey toward formulating the Information Theory of Aging was a longone. And in no smal part, it can be traced to the work of a scientist who toiled
without fame but whose work helped set the stage for a lot of the longevity
research being done around the world today.
His name was Robert Mortimer, and if there was one adjective that seemed
to come up more than any other about him after he passed away, it was “kind.”
“Visionary” was another. “Bril iant,” “inquisitive,” and “hardworking,” too.
But I’ve long been inspired by the example Mortimer set for his fel ow scientists.
Mortimer, who died in 2007, had played a tremendously important role in
elevating Saccharomyces cerevisiae from a seemingly lowly, single-cel ed yeast with
a sweet tooth (its name means “sugar-loving”) to its rightful place as one of the
world’s most important research organisms.
Mortimer col ected thousands of mutant yeast strains in his lab, many of
which had been developed right there at the University of California, Berkeley.
He could have paid for his research, and then some, by charging the thousands
of scientists he supplied through the university’s Yeast Genetic Stock Center.
But anyone, from impecunious undergraduates to tenured professors at the
world’s best-funded research institutions, could browse the center’s catalog,
request any strain, and have it promptly delivered for the cost of postage. 3
And because he made it so easy and so inexpensive, yeast research bloomed.
When Mortimer began working on S. cerevisiae alongside fel ow biologist
John Johnston4 in the 1950s, hardly anyone was interested in yeast. To most, it
didn’t seem we could learn much about our complex selves by studying a tiny
fungus. It was a struggle to convince the scienti c community that yeast could
be useful for something more than baking bread, brewing beer, and vinting
wine.
What Mortimer and Johnston recognized, and what many others began to
realize in the years to come, was that those tiny yeast cel s are not so di erent
from ourselves. For their size, their genetic and biochemical makeup is
extraordinarily complex, making them an exceptional y good model for
understanding the biological processes that sustain life and control lifespans in
large complex organisms such as ourselves. If you are skeptical that a yeast cel
can tel us anything about cancer, Alzheimer’s disease, rare diseases, or aging,
consider that there have been ve Nobel Prizes in Physiology or Medicineawarded for genetic studies in yeast, including the 2009 prize for discovering
how cel s counteract telomere shortening, one of the hal marks of aging. 5
The work Mortimer and Johnston did—and, in particular, a seminal paper in
1959 that demonstrated that mother and daughter yeast cel s can have vastly
di erent lifespans—would set the stage for a world-shattering change in the way
we view the limits of life. And by the time of Mortimer’s death in 2007, there
were some 10,000 researchers studying yeast around the globe.
Yes, humans are separated from yeast by a bil ion years of evolution, but we
stil have a lot in common. S. cerevisiae shares some 70 percent of our genes. And
what it does with those genes isn’t so di erent from what we do with them. Like
a whole lot of humans, yeast cel s are almost always trying to do one of two
things: either they’re trying to eat, or they’re trying to reproduce. They’re
hungry or they’re horny. As they age, much like humans, they slow down and
grow larger, rounder, and less fertile. But whereas humans go through this
process over the course of many decades, yeast cel s experience it in a week. That
makes them a pretty good place to start in the quest to understand aging.
Indeed, the potential for a humble yeast to tel us so much about ourselves—
and do so quite quickly relative to other research organisms—was a big part of
the reason I decided to begin my career by studying S. cerevisiae. They also smel
like fresh bread.
I met Mortimer in Vienna in 1992, when I was in my early 20s and attending
the International Yeast Conference—yes, there is such a thing—with my two
PhD supervisors, Professor Ian Dawes, a rule-avoiding Australian from the
University of New South Wales, 6 and Professor Richard Dickinson, a rule-
abiding Briton from the University of Cardi , Wales.
Mortimer was in Vienna to discuss a momentous scienti c endeavor: the
sequencing of the yeast genome. I was there to be inspired. And I was. 7 If I’d
harbored any doubts about my decision to dedicate the opening years of my
scienti c career to a single-cel ed fungus, they al went away when I came face to
face with people who were building great knowledge in a eld that had hardly
existed a few decades before.
It was shortly after that conference that one of the world’s top scientists inthe yeast eld, Leonard Guarente of the Massachusetts Institute of Technology,
came to Sydney on holiday to visit Ian Dawes. Guarente and I ended up at a
dinner together, and I made sure I was sitting opposite him.
I was then a graduate student using yeast to understand an inherited
condition cal ed maple syrup urine disease. As you might imagine from its name,
the disease is not something most polite people discuss over dinner. Guarente,
though, engaged me in a scienti c discussion with a curiosity and enthusiasm
that was nothing short of enchanting. The conversation soon turned to his latest
project—he had begun studying aging in yeast the past few months—work that
had its roots in the workable genetic map that Mortimer had completed in the
mid-1970s.
That was it. I had a passion for understanding aging, and I knew something
about wrangling a yeast cel with a microscope and micromanipulator. Those
were essential skil s needed to gure out why yeast age. That night, Guarente and
I agreed on one thing: if we couldn’t solve the problem of aging in yeast, we had
no chance in humans.
I didn’t just want to work with him. I had to work with him.
Dawes wrote him to tel him that I was keen to join his lab and I was “skil ed
at the bench.”
“It would be a pleasure to work with David,” he replied a few weeks later, the
same way he probably did to so many other enthusiastic applicants. “But he’s got
to come with his own funding.” Later I learned he had been excited only because
he’d thought I was the other student he’d met at dinner.
I had a foot in the door, but my chances were slim. At the time, foreigners
weren’t considered for prestigious postdoctoral awards in the United States, but
I insisted I be interviewed and paid for a ight to Boston myself. I was
interviewed by a giant in the stem cel eld, Douglas Melton, for a Helen Hay
Whitney Foundation Fel owship, which has been providing research support to
postdoctoral biomedical students since 1947. After waiting in line outside his
o ce with the other four candidates, I had my chance. This was my moment. I
don’t remember being nervous. I gured I probably wouldn’t get the award
anyway. So I went for it.
I told Melton about my lifelong quest to understand aging and nd “life-giving genes,” then sketched out on his whiteboard how the genes work and
what I’d be doing for the next three years if I got the money. To show my
gratitude, I gave him a bottle of red wine that I’d brought from Australia.
Afterward, two things became clear. One, don’t bring wine to an interview
because it can be seen as a bribe. And two, Melton must have liked what I said
and how I said it, because I ew home, got the fel owship, and then got onto a
plane back to Boston. It was, without a doubt, the most life-changing meeting of
my life. 8
At the time of my arrival, in 1995, I had expected to build our understanding
of aging by studying Werner syndrome, a terrible disease that occurs in less than
1 in 100,000 live births, with symptoms that include a loss of body strength,
wrinkles, gray hair, hair loss, cataracts, osteoporosis, heart problems, and many
other tel tale signs of aging—not among folks in their 70s and 80s but rather
among people in their 30s and 40s. Life expectancy for someone with Werner is
46 years.
Within two weeks of my arrival in the United States, though, a research team
at the University of Washington, headed by the wise and supportive grandfather
of aging research, George Martin, announced that they had found the gene that,
when mutated, causes Werner syndrome.9 It was de ating at the time to have
been “scooped,” but the discovery al owed me to take a bigger rst step toward
my ultimate objective. Indeed, it became the key to formulating the Information
Theory of Aging.
Now that the Werner gene, known as WRN, had been identi ed in humans,
the next step was to test if the similar gene in yeast had the same function. If so,
we could use yeast to more rapidly determine the cause of Werner syndrome and
perhaps help us better understand aging in general. I marched into Guarente’s
o ce to tel him I was now studying Werner’s syndrome in yeast and that’s how
we would solve aging.
In yeast, the equivalent of the WRN gene is Slow Growth Suppressor 1, or
SGS1. The gene was already suspected to code for a type of enzyme cal ed a
DNA helicase that untangles tangled strands of DNA before they break.
Helicases are especial y important in repetitive DNA sequences that are
inherently prone to tangling and breaking. Functionality of proteins, such as theones coded for by the Werner gene, is therefore vital, since more than half of our
genome is, in fact, repetitive.
Through a gene-swapping process in which cel s are tricked into picking up
extra pieces of DNA, we swapped out the functional SGS1 gene with a mutant
version. In e ect, we were testing to see if it was possible to give the yeast Werner
syndrome.
After the swap, the yeast cel s’ lifespan was cut in half. Ordinarily, this would
not have been news. Many events unrelated to aging—such as being eaten by a
mite, drying out on a grape, or being placed in an oven—can and do shorten the
lifespan of yeast cel s. And here we’d messed with their DNA, which could have
short-circuited the cel s in a thousand di erent ways to cause early death.
But those cel s weren’t just dying. They were dying after a precipitous decline
in health and function. As the SGS1 mutants became older, they slowed down
in their cel cycle. They grew larger. Both male and female “mating-type” genes
(descendants of gene A) were switched on at the same time, so they were sterile
and couldn’t mate. These were al known hal marks of aging in yeast. And it was
happening more quickly in the mutants we’d made. It certainly looked like a
yeast version of Werner’s.
Using specialized stains, we colored the DNA blue and used red for the
nucleolus, which sits inside the nucleus of al eukaryotic cel s. That made it
easier to see under the microscope what was happening at a cel ular level.
And what was happening was fascinating.
The nucleolus is a part of the nucleus in which ribosomal DNA, or rDNA,
resides. rDNA is copied into ribosomal RNA, which is used by ribosome
enzymes to stitch amino acids together to make every new protein.
In the aged SGS1 cel s, the nucleolus looked as if it had exploded. Instead of a
single red crescent swimming in a blue ocean, the nucleolus was scattered into
half a dozen smal islands. It was tragic and beautiful. The picture, which would
later appear in the August 1997 issue of the prestigious journal Science, stil
hangs in my o ce.
What happened next was both enchanting and il uminating. In response to
the damage, like rats to the cal of the Pied Piper, the protein cal ed Sir2—the
rst known sirtuin, which is encoded by the gene SIR2 10 and descended from
gene B—had moved away from the mating genes that control fertility and into
the nucleolus.
That was a beautiful sight to me, but it was a problem for the yeast. Sir2 has
an important job: it is an epigenetic factor, an enzyme that sits on genes, bundles
up the DNA, and keeps them silent. At the molecular level, Sir2 achieves this via
its enzymatic activity, making sure that chemicals cal ed acetyls don’t accumulate
on the histones and loosen the DNA packaging.
When sirtuins left the mating genes—the ones descended from gene A that
control ed fertility and reproduction—the mutant cel s turned on both male
and female genes, causing them to lose their sexual identity, just as in normal old
cel s, but much earlier.
I didn’t understand at rst why the nucleolus was exploding, let alone why
the sirtuins were moving toward it as the cel s grew older. I agonized over the
question for weeks.
And then one night, after a long day in the lab, I woke up with an idea.
It came in the space between sleep-deprived delirium and deep dreaming. The
wisps of a concept. A few words jumbled together. A muddled picture of
something. That was enough, though, to jolt me awake and pul me from my
bed.I grabbed my notebook and went to the kitchen. There, hunched over the
table in the early morning hours of October 28, 1996, I began to write.
I wrote for about an hour, jotting down ideas, drawing pictures, sketching
out graphs, formulating new equations.11 Scienti c observations that had
previously made no sense to me were fal ing perfectly into a larger picture.
Broken DNA causes genome instability, I wrote, which distracts the Sir2
protein, which changes the epigenome, causing the cel s to lose their identity
and become sterile while they xed the damage. Those were the analog scratches
on the digital DVDs. Epigenetic changes cause aging.
There was, I imagined, a singular process that control ed them al . Not acountless number of separate cel ular changes or diseases. Not even a set of
hal marks that could be addressed one at a time. There was something bigger—
and more singular—than any of that.
This was the foundation for understanding the survival circuit and its role in
aging.
The next day I showed Guarente my notes. I was excited; it felt like the
biggest idea I’d ever had. But I was nervous, too; afraid he would nd a hole in
my logic and tear it apart. Instead, he looked over my notebook quietly, asked a
few questions, and sent me on my way with six words.
“I like it,” he said. “Go prove it.”
THE RECITAL
To understand the Information Theory of Aging, we need to pay another visit to
the epigenome, the part of the cel that the sirtuins help control.
Up close, the epigenome is more complex and wonderful than anything we
humans have invented. It consists of strands of DNA wrapped around spooling
proteins cal ed histones, which are bound up into bigger loops cal ed chromatin,
which are bound up into even bigger loops cal ed chromosomes.
Sirtuins instruct the histone spooling proteins to bind up DNA tightly, while
they leave other regions to ail around. In this way, some genes stay silent, while
others can be accessed by DNA-binding transcription factors that turn genes
on. 12 Accessible genes are said to be in “euchromatin,” while silent genes are in
“heterochromatin.” By removing chemical tags on histones, sirtuins help
prevent transcription factors from binding to genes, converting euchromatin
into heterochromatin.
Every one of our cel s has the same DNA, of course, so what di erentiates a
nerve cel from a skin cel is the epigenome, the col ective term for the control
systems and cel ular structures that tel the cel which genes should be turned on
and which should remain o . And this, far more than our genes, is what actual y
controls much of our lives.
One of the best ways to visualize this is to think of our genome as a grandpiano.13 Each gene is a key. Each key produces a note. And from instrument to
instrument, depending on the maker, the materials, and the circumstances of
manufacturing, each wil sound a bit di erent, even if played the exact same way.
These are our genes. We have about 20,000 of them, give or take a few
thousand.14
Each key can also be played pianissimo (soft) or forte (with force). The notes
can be tenuto (held) or allegretto (played quickly). For master pianists, there are
hundreds of ways to play each individual key and endless ways to play the keys
together, in chords and combinations that create music we know as jazz,
ragtime, rock, reggae, waltzes, whatever.
The pianist that makes this happen is the epigenome. Through a process of
revealing our DNA or bundling it up in tight protein packages, and by marking
genes with chemical tags cal ed methyls and acetyls composed of carbon, oxygen,
and hydrogen, the epigenome uses our genome to make the music of our lives.
Yes, sometimes the size, shape, and condition of a piano dictate what a pianist
can do with it. It’s tough to play a concerto on an eighteen-key toy piano, and
it’s mighty hard to make beautiful music on an instrument that hasn’t been
tuned in fty years. Likewise, the genome certainly dictates what the epigenome
can do. A caterpil ar can’t become a human being, but it can become a butter y
by virtue of changes in epigenetic expression that occur during metamorphosis,
even though its genome never changes. Similarly, the child of two parents from a
long line of people with black hair and brown eyes isn’t likely to develop blond
hair and blue eyes, but twin agouti mice in the lab can turn out brown or
golden, depending on how much the Agouti gene is turned on during gestation
by environmental in uences on the epigenome, such as folic acid, vitamin B12,
genistein from soy, or the toxin bisphenol A. 15
Similarly, among monozygotic human twins, epigenetic forces can drive two
people with the same genome in vastly di erent directions. It can even cause
them to age di erently. You can see this clearly in side-by-side photographs of the
faces of smoking and nonsmoking twins; their DNA is stil largely the same, but
the smokers have bigger bags under their eyes, deeper jowls below their chins,
and more wrinkles around their eyes and mouths. They are not older, but
they’ve clearly aged faster. Studies of identical twins place the genetic in uenceson longevity at between 10 and 25 percent which, by any estimation, is
surprisingly low. 16
Our DNA is not our destiny.
Now imagine you’re in a concert hal . A virtuoso pianist is seated at a
gorgeously polished Steinway grand. The concerto begins. The music is
beautiful, breathtaking. Everything is perfect.
But then, a few minutes into the piece, the pianist misses a key. The rst time
it happens, it’s almost unnoticeable—an extra D, perhaps, in a chord that
doesn’t need that note. Embedded in so many perfectly played notes, hidden
among an otherwise awless chord in an otherwise perfect melody, it’s nothing
to worry about. But then, a few minutes later, it happens again. And then, with
increasing frequency, again and again and again.
It’s important to remember that there is nothing wrong with the piano. And
the pianist is playing most of the notes prescribed by the composer. She’s just
also playing some extra notes. Initial y, this is just annoying. Over time it
becomes unsettling. Eventual y it ruins the concerto. Indeed, we’d assume that
there was something wrong with the pianist. Someone might even rush onto the
stage to make sure she is al right.
Epigenetic noise causes the same kind of chaos. It is driven in large part by
highly disruptive insults to the cel , such as broken DNA, as it was in the original
survival circuit of M. superstes and in the old yeast cel s that lost their fertility.
And this, according to the Information Theory of Aging, is why we age. It’s why
our hair grays. It’s why our skin wrinkles. It’s why our joints begin to ache.
Moreover, it’s why each one of the hal marks of aging occurs, from stem cel
exhaustion and cel ular senescence to mitochondrial dysfunction and rapid
telomere shortening.
This is, I acknowledge, a bold theory. And the strength of a theory is based on
how wel it predicts the results of rigorous experiments, often mil ions of them,
the number of phenomena it can explain, and its simplicity. The theory was
simple, and it explained a lot. As good scientists, what we had left to do was to
try our best to disprove it and see how long it survived.
To get started, Guarente and I had to get our eyes on some yeast DNA.
We used a technique cal ed a Southern blot, a method of separating DNA
based on its size and conformation and lighting it up with a radioactive DNA
probe. In the rst experiment, we noticed something spectacular. Normal y, the
rDNA of a yeast cel that is made visible by a Southern blot is tightly packed, like
a new spool of rope, with a few barely visible wispy loops of supercoiled DNA.
But the rDNA of the yeast cel s we’d created in our lab—the Werner mutants
that seemed to be aging rapidly—were madly unpacking, like a vacuum-sealed
bag of yarn that had been ripped open.
LESSONS FROM YEAST CELLS ABOUT WHY WE AGE. In young yeast cel s, male and female
“mating-type information” (gene A) is kept in the “o ” position by the Sir2 enzyme, the rst
sirtuin (encoded by a descendant of gene B). The highly repetitive ribosomal DNA (rDNA) is
unstable, and toxic DNA circles form; these recombine and eventual y accumulate to toxic levels
in old cel s, kil ing them. In response to DNA circles and the perceived genome instability, Sir2
moves away from silent mating-type genes to help stabilize the genome. Both male and female
genes turn on, causing infertility, the main hal mark of yeast aging.
The rDNA was in a state of chaos. The genome, it seemed, was fragmenting.DNA was recombining and amplifying, showing up on the Southern blot as
dark spots and wispy circles, depending on how coiled up and twisted they were.
We cal ed those loops extrachromosomal ribosomal DNA circles, or ERCs, and
they were accumulating as the mutants aged.
If we had indeed induced aging, then we would see this same pattern emerge
in yeast cel s that had aged normal y.
We don’t count the age of a single yeast cel with birthday candles. They
simply don’t last that long. Instead, aging in yeast is measured by the number of
times a mother cel divides to produce daughter cel s. In most cases, a yeast cel
gets to about 25 divisions before it dies. That, however, makes obtaining old
yeast cel s an exceptional y chal enging task. Because by the time an average yeast
cel expires, it is surrounded by 225, or 33 mil ion, of its descendants.
It took a week of work, a lot of sleepless nights, and a whole lot of ca einated
beverages to col ect enough regular old cel s. The next day, when I developed the
lm to visualize the rDNA, what I saw astounded me. 17
Just like the mutants, the normal old yeast cel s were packed with ERCs.
That was a “Eureka!” moment. Not proof—a good scientist never has proof
of anything—but the rst substantial con rmation of a theory, the foundation
upon which I and others would build more discoveries in the years to come.
The rst testable prediction was if we put an ERC into very young yeast cel s
—and we devised a genetic trick to do that—the ERCs would multiply and
distract the sirtuins, and the yeast cel s would age prematurely, go sterile, and die
young—and they did. We published that work in December 1997 in the
scienti c journal Cell, and the news broke around the world: “Scientists gured
out a cause of aging.”
It was there and then that Matt Kaeberlein, a PhD student at the time,
arrived at the lab. His rst experiment was to insert an extra copy of SIR2 into
the genome of yeast cel s to see if it could stabilize the yeast genome and delay
aging. When the extra SIR2 was added, ERCs were prevented, and he saw a 30
percent increase in the yeast cel s’ lifespan, as we’d been hoping. Our hypothesis
seemed to be standing up to scrutiny: the fundamental, upstream cause of
sterility and aging in yeast was the inherent instability of the genome.
What emerged from those initial results in yeast, and another decade ofpondering and probing mammalian cel s, was a completely new way to
understand aging, an information theory that would reconcile seemingly
disparate factors of aging into one universal model of life and death. It looked
like this:
Youth → broken DNA → genome instability → disruption of DNA
packaging and gene regulation (the epigenome) → loss of cel identity →
cel ular senescence → disease → death.
The implications were profound: if we could intervene in any of these steps, we
might help people live longer.
But what if we could intervene in al of them? Could we stop aging?
Theories must be tested and tested and tested some more—not just by one
scientist but by many. And to that end, I was fortunate to have been put onto a
research team that included some of the most bril iant and insightful scientists
in the world.
There was Lenny Guarente, our indefatigable mentor. There was also Brian
Kennedy, who started the yeast-aging project in Lenny’s lab and has since played
a tremendously important role in understanding premature aging diseases and
the impact of genes and molecules that increase health and longevity in model
organisms. There were Monica Gotta and Susan Gasser at the University of
Geneva, who are now some of the most in uential researchers in the eld of
gene regulation; Shin-ichiro Imai, now a professor at Washington University,
who discovered that sirtuins are NAD-utilizing enzymes and now does research
into how the body controls sirtuins; Kevin Mil s, who ran a lab in Maine, then
became a cofounder of and chief scienti c o cer at Cyteir Therapeutics, which
develops novel ways to ght cancer and autoimmune diseases; Nicanor
Austriaco, who started the project with Brian, now teacher of biology and
theology at Providence Col ege, a great combo; Tod Smeal, chief scienti c o cer
of cancer biology at the global pharmaceutical company Eli Lil y; David
Lombard, who is now a researcher in the eld of aging at the University of
Michigan; Matt Kaeberlein, a professor at the University of Washington, who is
testing molecules on dog longevity; David McNabb, whose lab at the Universityof Arkansas has made key and lifesaving discoveries about fungal pathogens;
Bradley Johnson, an expert on human aging and cancer at the University of
Pennsylvania; and Mala Murthy, a prominent neuroscientist now at Princeton.
Again and again I have been greatly privileged in the matter of those who
work around me. And that was never truer than it was in Guarente’s lab at MIT.
It was a dream team, and I often felt humbled by the people with whom I was
surrounded.
When I began my career in this eld, I dreamt of publishing just one study in
a top-tier journal. During those years, our group was publishing one every few
months.
We demonstrated that the redistribution of Sir2 to the nucleolus is a response
to numerous DNA breakages, which happen as a result of ERCs multiplying
and inserting back into the genome or joining together to form superlarge
ERCs. When Sir2 moves to combat DNA instability, it causes sterility in old,
bloated yeast cel s. That was the rst step of the survival circuit, though at the
time we had no idea that it was as ancient and as essential to our very existence as
it turned out to be.
We told the world that we could give yeast a Werner-like syndrome, causing
exploded nucleoli.18 We described the ways in which mutants of SGS1, those we’d plagued with the yeast equivalent to the Werner syndrome mutation,
accumulated ERCs more rapidly, leading to premature aging and a shortened
lifespan. 19 Crucial y, by demonstrating that if you add an ERC to young cel s
they age prematurely, we had crucial evidence that ERCs don’t just happen
during aging, they cause it. And by arti cial y breaking the DNA in the cel and
watching the cel ular response, we showed why sirtuins move—to help with
DNA repair.20 That turned out to be the second step of the survival circuit.21
The DNA damage that gave rise to the ERCs was distracting Sir2 from the
mating-type genes, causing them to become sterile, a hal mark of yeast aging.
It was epigenomic noise in its purest form.
It took another twenty years to learn if those ndings in yeast were relevant to
organisms more complex than yeast. We mammals have seven sirtuin genes that
have evolved a variety of functions beyond what simple SIR2 can do. Three of
them, SIRT1, SIRT6, and SIRT7, are critical to the control of the epigenome and DNA repair. The others, SIRT3, SIRT4, and SIRT5, reside inmitochondria, where they control energy metabolism, while SIRT2 buzzes
around the cytoplasm, where it controls cel division and healthy egg
production.
There had been many clues along the way. Brown University’s Stephen
Helfand showed that adding extra copies of the dSir2 gene to fruit ies
suppresses epigenetic noise and extends their lifespan. We found that SIRT1 in
mammals moves from silent genes to help repair broken DNA in mouse and
human cel s. 22 But the true extent to which the survival circuit is conserved between yeast and humans wasn’t ful y known until 2017, when Eva Bober’s
team at the Max Planck Institute for Heart and Lung Research in Bad Nauheim,
Germany, reported that sirtuins stabilize human rDNA. 23 Then, in 2018, Katrin
Chua at Stanford University found that, by stabilizing human rDNA, sirtuins
prevent cel ular senescence—essential y the same antiaging function as we had
found for sirtuins in yeast twenty years earlier.24
That was an astonishing revelation: over a bil ion years of separation between
yeast and us, and, in essence, the circuit hadn’t changed.
By the time those ndings appeared, though, it was clear to me that
epigenomic noise was a likely catalyst of human aging. Two decades of research
had already been leading us in that direction.25
In 1999, I moved from MIT across the river to Harvard Medical School,
where I set up a new lab on aging. There I was hoping to answer a new question
that had increasingly been occupying my thoughts.
I had noticed that yeast cel s fed with lower amounts of sugar were not just
living longer, but their rDNA was exceptional y compact—signi cantly delaying
the inevitable ERC accumulation, catastrophic numbers of DNA breaks,
nucleolar explosion, sterility, and death.
Why was that happening?
THE SURVIVAL CIRCUIT COMES OF AGE
Our DNA is constantly under attack. On average, each of our forty-sixchromosomes is broken in some way every time a cel copies its DNA,
amounting to more than 2 tril ion breaks in our bodies per day. And that’s just
the breaks that occur during replication. Others are caused by natural radiation,
chemicals in our environment, and the X-rays and CT scans that we’re subjected
to. If we didn’t have a way to repair our DNA, we wouldn’t last long. That’s
why, way back in primordium, the ancestors of every living thing on this planet
today evolved to sense DNA damage, slow cel ular growth, and divert energy to
DNA repair until it was xed—what I cal the survival circuit.
Since the yeast work, evidence that yeast aren’t so di erent from us has
continued to accumulate. In 2003, Michael McBurney from the University of
Ottawa in Canada discovered that mouse embryos manipulated to be unable to
produce one of the seven sirtuin enzymes, SIRT1, couldn’t last past the
fourteenth day of development—about two-thirds of the way into a mouse’s
gestation period. 26 Among the reasons, the team reported in the journal Cancer
Cell, was an impaired ability to respond to and repair DNA damage. 27 In 2006,
Frederick Alt, Katrin Chua, and Raul Mostovslavsky at Harvard showed that
mice engineered to lack SIRT6 underwent the typical signs of aging faster along
with shortened lifespans. 28 When the scientists knocked out a cel ’s ability to create this vital protein, the cel lost its ability to repair double-strand DNA
breaks, just as we had showed in yeast back in 1999.
If you are skeptical, and you should be, you might assume these SIRT mutant
mice could just be sick and, therefore, short lived. But adding in more copies of
the sirtuin genes SIRT1 and SIRT6 does just the opposite: it increases the health
and extends the lifespan of mice, just as adding extra copies of the yeast SIR2
gene does in yeast. 29 Credit for these discoveries goes to two of my previous col eagues, Shin-ichiro Imai, my former drinking buddy at the Guarente lab, and
Haim Cohen, my rst postdoc at Harvard.
In yeast, we had shown that DNA breaks cause sirtuins to relocalize away
from silent mating-type genes, causing old cel s to become sterile. That was a
simple system, and we’d gured it out in a few years.
But is the survival circuit causing aging in mammals? What parts of thesystem survived the bil ion years, and which are yeast speci c? Those questions
are on the cutting edge of human knowledge right now, but the answers are
beginning to reveal themselves.
What I’m suggesting is that the SIR2 gene in yeast and the SIRT genes in
mammals are al descendants of gene B, the original gene silencer in M.
superstes. 30 Its original job was to silence a gene that control ed reproduction.
In mammals, the sirtuins have since taken on a variety of new roles, not just
as control ers of fertility (which they stil are). They remove acetyls from
hundreds of proteins in the cel : histones, yes, but also proteins that control cel
division, cel survival, DNA repair, in ammation, glucose metabolism,
mitochondria, and many other functions.
I’ve come to think of sirtuins as the directors of a multifaceted disaster
response corps, sending out a variety of specialized emergency teams to address
DNA stability, DNA repair, cel survivability, metabolism, and cel -to-cel
communication. In a way, this is like the command center for the thousands of
utility workers who descended upon Louisiana and Mississippi in the wake of
Hurricane Katrina in 2005. Most of the workers weren’t from the Gulf Coast,
but they came, did their level best to x what was broken, and then went home.
Some were working in the storm-ravaged communities for a few days and others
for a few weeks before returning to their normal lives. And for most, it wasn’t
the rst or last time they had done something like that; anytime there’s a mass
disaster that impacts utilities, they swoop in to help.
When they’re home, those folks take care of the typical business of being at
home: paying bil s, mowing lawns, coaching basebal , whatever. But when
they’re away, helping keep places like the Gulf Coast from descending into
anarchy—a condition that would have had disastrous results for the rest of the
nation—a lot of those things have to be put on hold.
When sirtuins shift from their typical priorities to engage in DNA repair,
their epigenetic function at home ends for a bit. Then, when the damage is xed
and they head back to home base, they get back to doing what they usual y do:
control ing genes and making sure the cel retains its identity and optimal
function.
But what happens when there’s one emergency after another to tend to?Hurricane after hurricane? Earthquake after earthquake? The repair crews are
away from home a lot. The work they normal y do piles up. The bil s come due,
then overdue, and then the folks from col ections start cal ing. The grass grows
too long, and soon the president of the neighborhood association is sending
nastygrams. The basebal team goes coachless, and the team devolves into the
Bad News Bears. And most of al , one of the most important things they do
while at home—reproducing—doesn’t get done. This form of hormesis, the
original survival circuit, works ne to keep organisms alive in the short term. But
unlike longevity molecules that simply mimic hormesis by tweaking sirtuins,
mTOR, or AMPK, sending out the troops on fake emergencies, these real
emergencies create life-threatening damage.
What could cause so many emergencies? DNA damage. And what causes
that? Wel , over time, life does. Malign chemicals. Radiation. Even normal DNA
copying. These are the things that we’ve come to believe are the causes of aging,
but there is a subtle but vital shift we have to make in that manner of thinking.
It’s not so much that the sirtuins are overwhelmed, though they probably are
when you are sunburned or get an X-ray; what’s happening every day is that the
sirtuins and their coworkers that control the epigenome don’t always nd their
way back to their original gene stations after they are cal ed away. It’s as if a few
emergency workers who came to address the damage done in the Gulf Coast by
Katrina had lost their home address. Then disaster strikes again and again, and
they must redeploy.
Wherever epigenetic factors leave the genome to address damage, genes that
should be o , switch on and vice versa. Wherever they stop on the genome, they
do the same, altering the epigenome in ways that were never intended when we
were born.
Cel s lose their identity and malfunction. Chaos ensues. The chaos
materializes as aging. This is the epigenetic noise that is at the heart of our
uni ed theory.
How does the SIR2 gene actual y turn o genes? SIR2 codes for a specialized
protein cal ed a histone deacetylase, or HDAC, that enzymatical y cleaves the
acetyl chemical tags from histones, which, as you’l recal , causes the DNA tobundle up, preventing it from being transcribed into RNA.
When the Sir2 enzyme is sitting on the mating-type genes, they remain silent
and the cel continues to mate and reproduce. But when a DNA break occurs,
Sir2 is recruited to the break to remove the acetyl tags from the histones at the
DNA break. This bundles up the histones to prevent the frayed DNA from
being chewed back and to help recruit other repair proteins. Once the DNA
repair is complete, most of the Sir2 protein goes back to the mating-type genes
to silence them and restore fertility. That is, unless there is another emergency,
such as the massive genome instability that occurs when ERCs accumulate in
the nucleoli of old yeast cel s.
For the survival circuit to work and for it to cause aging, Sir2 and other
epigenetic regulators must occur in “limiting amounts.” In other words, the cel
doesn’t make enough Sir2 protein to simultaneously silence the mating-type
genes and repair broken DNA; it has to shuttle Sir2 between the various places
on an “as-needed” basis. This is why adding an extra copy of the SIR2 gene
extends lifespan and delays infertility: cel s have enough Sir2 to repair DNA
breaks and enough Sir2 to silence the mating-type genes.31
Over the past bil ion years, presumably mil ions of yeast cel s have
spontaneously mutated to make more Sir2, but they died out because they had
no advantage over other yeast cel s. Living for 28 divisions was no advantage over
those that lived for 24 and, because Sir2 uses up energy, having more of the
protein may have even been a disadvantage. In the lab, however, we don’t notice
any disadvantage because the yeast are given more sugar than they could possibly
ever eat. By adding extra copies of the SIR2 gene, we gave the yeast cel s what
evolution failed to provide.
If the information theory is correct—that aging is caused by overworked
epigenetic signalers responding to cel ular insult and damage—it doesn’t so
much matter where the damage occurs. What matters is that it is being damaged
and that sirtuins are rushing al over the place to address that damage, leaving
their typical responsibilities and sometimes returning to other places along the
genome where they are silencing genes that aren’t supposed to be silenced. This
is the cel ular equivalent of distracting the cel ular pianist.
To prove that, we needed to break some mouse DNA.It’s not hard to intentional y break DNA. You can do it with mechanical
shearing. You can do it with chemotherapy. You can do it with X-rays.
But we needed to do it with precision—in a way that wouldn’t create
mutations or impact regions that a ect any cel ular function. In essence, we
needed to attack the wastelands of the genome. To do that, we got our hands on
a gene similar to Cas9, the CRISPR gene-editing tool from bacteria that cuts
DNA at precise places.
The enzyme we chose for our experiments comes from a goopy yel ow slime
mold cal ed Physarum polycephalum, which literal y means “many-headed
slime.” Most scientists believe that this gene, cal ed I- Ppo I, is a parasite that serves
only to copy itself. When it cuts the slime mold genome, another copy of I- Ppo I
is inserted. It is the epitome of a sel sh gene.
That’s in a slime mold, its native habitat. But when I- Ppo I nds itself in a
mouse cel , it doesn’t have al the slime mold machinery to copy itself. So it oats
around and cuts DNA at just a few places in the mouse genome, and there is no
copying process. Instead, the cel has no problem pasting the DNA strands back
together, leaving no mutations, which is exactly what we were looking for to
engage the survival circuit and distract the sirtuins. DNA-editing genes such as
Cas9 and I- Ppo I are nature’s gifts to science.
To create a mouse to test the information theory, we inserted I- Ppo I into a
circular DNA molecule cal ed a plasmid, along with al the regulatory DNA
elements needed to control the gene, and then inserted that DNA into the
genome of a mouse embryonic stem cel line we were culturing in plastic dishes
in the lab. We then injected the genetical y modi ed stem cel s into a 90-cel
mouse embryo cal ed a blastocyst, implanted it into a female mouse’s uterus, and
waited about twenty days for a baby mouse to show up.
This al sounds complicated, but it’s not. After some training, a col ege
student can do it. It’s such a commodity these days, you can even order a mouse
out of a catalog or pay a company to make you one to your speci cations.
The baby mice were born perfectly normal, as expected, since the cutting
enzyme was switched o at that stage. We cal ed them a ectionately “ICE mice,”
ICE standing for “Inducible Changes to the Epigenome.” The “inducible” part
of the acronym is vital—because there’s nothing di erent about these mice untilwe feed them a low dose of tamoxifen. This is an estrogen blocker that is
normal y used to treat human cancers, but in this case, we’d engineered the
mouse so that tamoxifen would turn on the I- Ppo I gene. The enzyme would go
to work, cutting the genome and slightly overwhelming the survival circuit,
without kil ing any cel s. And since tamoxifen has a half-life of only a couple of
days, removing it from the mice’s food would turn o the cutting.
The mice might have died. They might have grown tumors. Or they might
have been perfectly ne, no worse o than if they’d received a dental X-ray.
Nobody had ever done this before in a mouse, so we didn’t know. But if our
hypothesis about epigenetic instability and aging was correct, the tamoxifen
would work like the potion that Fred and George Weasley used to age themselves
in Harry Potter and the Goblet of Fire.
And it worked. Like wizardry, it did.
During the treatment, the mice were ne, oblivious to the DNA cutting and
sirtuin distraction. But a few months later, I got a cal from a postdoc who was
taking care of our lab’s animals while I was on a trip to my lab in Australia.
“One of the mice is real y sick,” she said. “I think we need to put it down.”
I asked her to text me a photo of the mouse she was talking about. When the
photo came over my phone, I couldn’t help but laugh.
“That’s not a sick mouse,” I replied. “That’s an old mouse.”
“David,” she said, “I think you’re mistaken. It says here that it’s the sister of
these other mice in the cage, and they’re perfectly normal.”
Her confusion was understandable. At 16 months old, a regular lab mouse
stil has a thick coat of fur, a sturdy tail, a muscular gure, perky ears, and clear
eyes. A tamoxifen-triggered ICE mouse at the same age has thinning, graying
hair, a bent spine, paper-thin ears, and cloudy eyes.
Remember, we’d done nothing to change the genome. We’d simply broken
the mice’s DNA in places where there aren’t any genes and forced the cel to
paste, or “ligate,” them back together. Just to make sure, later we broke the
DNA in other places, too, with the same results. Those breaks had induced a
sirtuin response. When those xers went to work, their absence from their
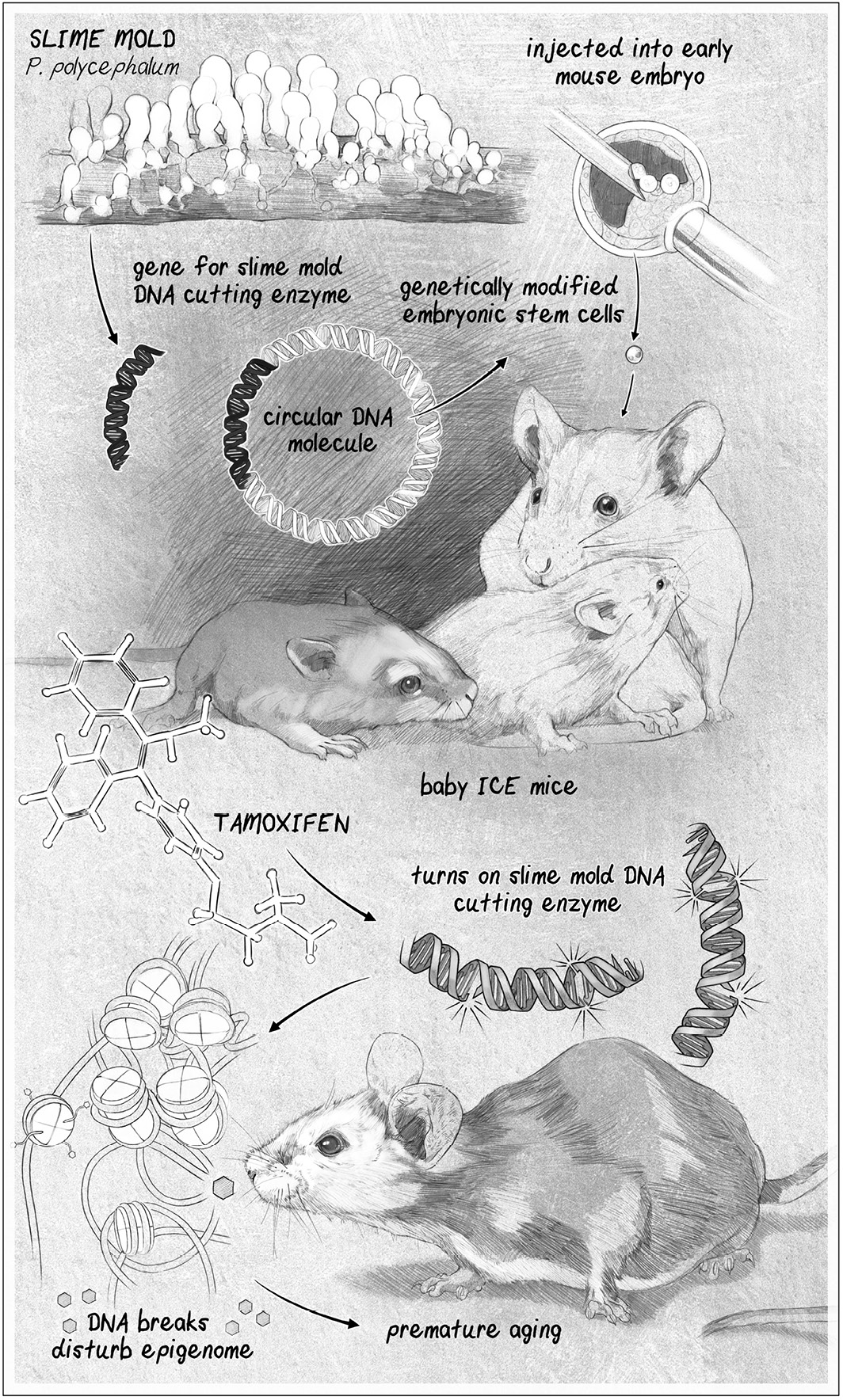
normal duties and presence on other parts of the genome altered the ways in
which lots of genes were being expressed at the wrong time.
THE MAKING OF THE ICE MOUSE TO TEST IF THE CAUSE OF AGING MIGHT BE
INFORMATION LOSS. A gene from a slime mold that encodes an enzyme that cuts DNA at a
speci c place was inserted into a stem cel and injected into an embryo to generate the ICE
mouse. Turning on the slime mold gene cut the DNA and distracted the sirtuins, causing the
mouse to undergo aging.
Those ndings were aligned to discoveries being made by Trey Ideker and
Kang Zhang, at UC San Diego, and Steve Horvath, at UCLA. Steve’s name
stuck, and today he’s the namesake of the Horvath Clock—an accurate way of
estimating someone’s biological age by measuring thousands of epigenetic marks
on the DNA, cal ed methylation. We tend to think of aging as something that
begins happening to us at midlife, because that’s when we start to see signi cant
changes to our bodies. But Horvath’s clock begins ticking the moment we are
born. Mice have an epigenetic clock, too. Were the ICE mice older than their
siblings? Yes, they were—about 50 percent older.
We’d found life’s master clock winder.In another manner of thinking, we’d scratched up the DVD of life about 50
percent faster than it normal y gets scratched. The digital code that is, and was,
the basic blueprint for our mice was the same as it had always been. But the
analog machine built to read that code was able to pick up only bits and pieces
of the data.
Here’s the vital takeaway: we could age mice without a ecting any of the
most commonly assumed causes of aging. We hadn’t made their cel s mutate. We
hadn’t touched their telomeres. We hadn’t messed with their mitochondria. We
hadn’t directly exhausted their stem cel s. Yet the ICE mice were su ering from a
loss of body mass, mitochondria, and muscle strength and an increase in
cataracts, arthritis, dementia, bone loss, and frailty.
Al of the symptoms of aging—the conditions that push mice, like humans,
farther toward the precipice of death—were being caused not by mutation but
by the epigenetic changes that come as a result of DNA damage signals.
We hadn’t given the mice al of those ailments. We had given them aging.
And if you can give something, you can take it away.
FRUIT OF THE SAME TREE
Like the gnarled hands of giant zombies breaking free of the rocky soil, the
ancient bristlecone pine trees of California’s White Mountains strike haunting
silhouettes against the dewy morning sun.
The oldest of these trees have been here since before the pyramids of Egypt
were built, before the construction of Stonehenge, and before the last of the
wool y mammoths left our world. They have shared this planet with Moses,
Jesus, Muhammad, and the rst Buddha. Standing some two miles above sea
level, adding fractions of a mil imeter of growth to their twisted trunks each
year, defying lightning storms and periodic droughts, they are the epitome of
perseverance.
It’s easy to stand in wonder of these great and ancient things. It’s easy to be
swept away by their might and majesty. It’s easy to simply stare at them in awe.
But there’s another way to view these antediluvian patriarchs—a harder way, buta way in which we should seek to view every living thing on this planet: as our
teachers.
Bristlecones are, after al , our eukaryotic cousins. About half of their genes
are close relatives of ours.
Yet they do not age.
Oh, they add years to their lives—thousands upon thousands of them,
marked by the nearly microscopic rings hidden in their dense heartwood, which
also record in their size, shape, and chemical composition climate events long
past, as when the eruption of Krakatoa sent a cloud of ash around the globe in
1883, leaving a fuzzy ring of growth in 1884 and 1885, barely a centimeter from
the outer ring of bark that marks our current time. 32
Yet even over the course of many thousands of years, their cel s do not appear
to have undergone any decline in function. Scientists cal this “negligible
senescence.” Indeed, when a team from the Institute of Forest Genetics went
looking for signs of cel ular aging—studying bristlecones from 23 to 4,713 years
old—they came up empty-handed. Between young and old trees, their 2001
study found, there were no meaningful di erences in the chemical
transportation systems, in the rate of shoot growth, in the quality of the pol en
they produced, in the size of their seeds, or in the way those seeds germinated.33
The researchers also looked for deleterious mutations—the sorts of which
many scientists at the time expected to be a primary cause of aging. They found
none.34 I expect that if they were to look for epigenetic changes, they would similarly come up empty-handed.
Bristlecones are outliers in the biological world, but they are not unique in
their de ance of aging. The freshwater polyp known as Hydra vulgaris has also
evolved to defy senescence. Under the right conditions, these tiny cnidarians
have demonstrated a remarkable refusal to age. In the wild they might live for
only a few months, subject to the powers of predation, disease, and desiccation.
But in labs around the world they have been kept alive for upward of 40 years—
with no signs that they’l stop there—and indicators of health don’t di er
signi cantly between the very young and the very old.
A couple of species of jel y sh can completely regenerate from adult bodyparts, earning them the nickname “immortal jel ies.” Only the elegant moon jel y
Aurelia aurita from the US West Coast and the centimeter-long Turritopsis
dohrnii from the Mediterranean are currently known to regenerate, but I’m
guessing the majority of jel ies do. We just need to look. If you separate one of
these amazing animals into single cel s, the cel s jostle around until they form
clumps that then assemble back into a complete organism, like the T-1000
cyborg in Terminator 2, most likely resetting their aging clock.
Of course, we humans don’t want to be mashed into single cel s to be
immortal. What use is reassembling or spawning if you have no recol ection of
your present life? We may as wel be reincarnated.
What matters is what these biological equivalents of F. Scott Fitzgerald’s
backward-aging Benjamin Button teach us: that cel ular age can be ful y reset,
something I’m convinced we wil be able to do one day without losing our
wisdom, our memories, or our souls.
Though it’s not immortal, the Greenland shark Somniosus microcephalus is
stil an impressive animal and far more closely related to us. About the size of a
great white, it does not even reach sexual maturity until it is 150 years old.
Researchers believe the Arctic Ocean could be home to Greenland sharks that
were born before Columbus got lost in the New World. Radiocarbon dating
estimated that one very large individual may have lived more than 510 years, at
least up until it was caught by scientists so they could measure its age. Whether
this shark’s cel s undergo aging is an open scienti c question; very few biologists
had so much as looked at S. microcephalus until the past few years. At the very
least, this longest-living vertebrate undergoes the process of aging very, very
slowly.
Evolutionarily speaking, al of these life-forms are closer to us than yeast, and
just think of what we’ve learned about human aging from that tiny fungus. But
it is certainly forgivable to consider the distances between pine trees, hydrozoans,
cartilaginous sh, and mammals like ourselves on the enormous tree of life and
say, “No, these things are just too di erent.”
What, then, of another mammal? A warm-blooded, milk-producing, live-
birth-giving cousin?
Back in 2007, aboriginal hunters in Alaska caught a bowhead whale that,when butchered, was found to have the head of an old harpoon embedded in its
blubber. The weapon, historians would later determine, had been manufactured
in the late 1800s, and they estimated the whale’s age at about 130. That
discovery sparked a new scienti c interest in Balaena mysticetus, and later
research, employing an age-determining method that measures the levels of
aspartic acid in the lens of a whale’s eye, estimated that one bowhead was 211
years old when it was kil ed by native whalers.
That bowheads have been selected for exceptional longevity among mammals
should perhaps not be surprising. They have few predators and can a ord to
build a long-lived body and breed slowly. Most likely they maintain their survival
program on high alert, repairing cel s while maintaining a stable epigenome,
thereby making sure the symphony of the cel s plays on for centuries.
Can these long-lived species teach us how to live healthier and for longer?
In terms of their looks and habitats, pine trees, jel y sh, and whales are
certainly very di erent from humans. But in other ways, we’re very similar.
Consider the bowheads. Like us, they are complex, social, communicative, and
conscious mammals. We share 12,787 known genes, including some interesting
variants in a gene known as FOXO3. Also known as DAF-16, this gene was rst
identi ed as a longevity gene in roundworms by University of California at San
Francisco researcher Cynthia Kenyon. She found it to be essential for defects in
the insulin hormone pathway to double worm lifespan. Playing an integral role
in the survival circuit, DAF-16 encodes a smal transcription factor protein that
latches onto the DNA sequence TTGTTTAC and works with sirtuins to
increase cel ular survival when times are tough. 35
In mammals, there are four DAF-16 genes, cal ed FOXO1, FOXO3, FOXO4,
and FOXO6. If you suspect that we scientists sometimes intentional y
complicate matters, you’d be right, but not in this case. Genes in the same “gene
family” have ended up with di erent names because they were named before
DNA sequences were easily deciphered. It’s similar to the not uncommon
situation in which people have their genome analyzed and learn they have a
sibling on the other side of town. 36 DAF-16 is an acronym for dauer larvae formation. In German, “dauer” means “long lasting,” and this is actual y
relevant to this story. Turns out, worms become dauer when they are starved orcrowded, hunkering down until times improve. Mutations that activate DAF-16
extend lifespan by turning on the worm defense program even when times are
good.
I rst encountered FOXO/DAF-16 in yeast, where it is known as MSN2,
which stands for “multicopy suppressor of SNF1 (AMPK) epigenetic
regulator.” Like DAF-16, MSN2’s job in yeast is to turn on genes that push cel s
away from cel death and toward stress resistance.37 We discovered that when
calories are restricted MSN2 extends yeast lifespan by turning up genes that
recycle NAD, thereby giving the sirtuins a boost. 38
Hidden within the sometimes byzantine way scientists talk about science are
several repeating themes: low energy sensors (SNF1/AMPK), transcription
factors (MSN2/DAF-16/FOXO), NAD and sirtuins, stress resistance, and
longevity. This is no coincidence—these are al key parts of the ancient survival
circuit.
But what about FOXO genes in humans? Certain variants cal ed FOXO3 have
been found in human communities in which people are known to enjoy both
longer lifespans and healthspans, such as the people of China’s Red River
Basin. 39 These FOXO3 variants likely turn on the body’s defenses against diseases and aging, not just when times are tough but throughout life. If you’ve
had your genome analyzed, you can check if you have any of the known
variations of FOXO3 that are associated with a long life. 40 For example, having a C instead of a T variant at position rs2764264 is associated with longer life. Two
of our children, Alex and Natalie, inherited two Cs at this position, one from
Sandra and one from me, so al other genes being equal, and as long as they
don’t live terribly negative lifestyles, they should have greater odds of reaching
age 95 than I do, with my one C and one T, and substantial y greater than
someone with two Ts.
It’s worth pausing to consider how remarkable it is that we nd essential y
the same longevity genes in every organism on the planet: trees, yeast, worms,
whales, and humans. Al living creatures come from the same place in
primordium that we do. When we look through a microscope, we’re al made of
the same stu . We al share the survival circuit, a protective cel ular network that
helps us when times are tough. This same network is our downfal . Severe typesof damage, such as broken strands of DNA, cannot be avoided. They overwork
the survival circuit and change cel ular identity. We’re al subject to epigenetic
noise that should, under the Information Theory of Aging, cause aging.
Yet di erent organisms age at very di erent rates. And sometimes, it appears,
they do not age at al . What al ows a whale to keep the survival circuit on
without disrupting the epigenetic symphony? If the piano player’s skil s are lost,
how is it possible for a jel y sh to restore her ability?
These are the questions that have been guiding my thoughts as I have
considered where our research is headed. What might seem like fanciful ideas, or
concepts straight out of science ction, are rmly rooted in research. Moreover,
they are supported by the knowledge that some of our close relatives have
gured out a workaround to aging.
And if they can, we can, too.
THE LANDSCAPE OF OUR LIVES
Before most people could even fathom the idea of mapping our genome, before
we had the technology to map a cel ’s entire epigenome and understand how it
bundles DNA to turn genes on and o , the developmental biologist Conrad
Waddington was already thinking deeper.
In 1957, the professor of genetics, from the University of Edinburgh, was
trying to understand how an early embryo could possibly be transformed from a
col ection of undi erentiated cel s—each one exactly like the next and with the
exact same DNA—to the thousands of di erent cel types in the human body.
Perhaps not coincidental y, Waddington’s ponderings came in the dawning years
of the digital revolution, at the same time that Grace Hopper, the mother of
computer programming, was laying the foundation for the rst widely used
computer language, COBOL. In essence, what Waddington was seeking to
ascertain was how cel s, al running on the same code, could possibly produce
di erent programs.
There had to be something more than genetics at play: a program thatcontrol ed the code.
Waddington conceived of an “epigenetic landscape,” a three-dimensional
relief map that represents the dynamic world in which our genes exist. More
than half a century later, Waddington’s landscape remains a useful metaphor to
understand why we age.
On the Waddington map, an embryonic stem cel is represented by a marble
at the top of a mountain peak. During embryonic development, the marble rol s
down the hil and comes to rest in one of hundreds of di erent val eys, each
representing a di erent possible cel type in the body. This is cal ed
“di erentiation.” The epigenome guides the marbles, but it also acts as gravity
after the cel s come to rest, ensuring that they don’t move back up the slope or
hop over into another val ey.
The nal resting place is known as the cel ’s “fate.” We used to think this was
a one-way street, an irreversible path. But in biology there is no such thing as
fate. In the last decade, we’ve learned that the marbles in the Waddington
landscape aren’t xed; they have a terrible tendency to move around over time.
At the molecular level, what’s real y going on as the marble rol s down the
slope is that di erent genes are being switched on and o , guided by
transcription factors, sirtuins and other enzymes such as DNA
methyltransferases (DNMTs) and histone methyltransferases (HMTs), which
mark the DNA and its packing proteins with chemical tags that instruct the cel
and its descendants to behave in a certain way.
What’s not general y appreciated, even in scienti c circles, is how important
the stability of this information is for our long-term health. You see, epigenetics
was long the purview of scientists who study the very beginnings of life, not
folks like me who are studying the other end of things.
Once a marble has settled in Waddington’s landscape, it tends to stay there. If
al goes wel with fertilization, the embryo develops into a fetus, then a baby,
then a toddler, then a teenager, then an adult. Things tend to go wel in our
youth. But the clock is ticking.
Every time there’s a radical adjustment to the epigenome, say, after DNA
damage from the sun or an X-ray, the marbles are jostled—envision a smal
earthquake that ever so slightly changes the map. Over time, with repeatedearthquakes and erosion of the mountains, the marbles are moved up the sides
of the slope, toward a new val ey. A cel ’s identity changes. A skin cel starts
behaving di erently, turning on genes that were shut o in the womb and were
meant to stay o . Now it is 90 percent a skin cel and 10 percent other cel types,
al mixed up, with properties of neurons and kidney cel s. The cel becomes inept
at the things skin cel s must do, such as making hair, keeping the skin supple,
and healing when injured.
In my lab we say the cel has ex-differentiated.
Each cel is succumbing to epigenetic noise. The tissue made up of thousands
of cel s is becoming a melange, a medley, a miscel aneous set of cel s.
As you’l recal , the epigenome is inherently unstable because it is analog
information—based on an in nite number of possible values—and thus it’s
di cult to prevent the accumulation of noise and nearly impossible to duplicate
without some information loss. The earthquakes are a fact of life. The landscape
is always changing.
If the epigenome had evolved to be digital rather than analog, the val ey wal s
would be the equivalent of 100 miles high and vertical, and gravity would be
superstrong, so the marbles could never jump over into a new val ey. Cel s would
never lose their identity. If we were built this way, we could be healthy for
thousands of years, perhaps longer.
But we are not built this way. Evolution shapes both genomes and
epigenomes only enough to ensure su cient survival to ensure replacement—
and perhaps, if we are lucky, just a little bit more—but not immortality. So our
val ey wal s are only slightly sloped, and gravity isn’t that strong. A whale that
lives two hundred years has probably evolved steeper val ey wal s and its cel s
maintain their identity for twice as long as ours do. Yet even whales don’t live
forever.
I believe the blame lies with M. superstes and the survival circuit. The
repeated shu ing of sirtuins and other epigenetic factors away from genes to
sites of broken DNA, then back again, while helpful in the short term, is
ultimately what causes us to age. Over time, the wrong genes come on at the
wrong time and in the wrong place.
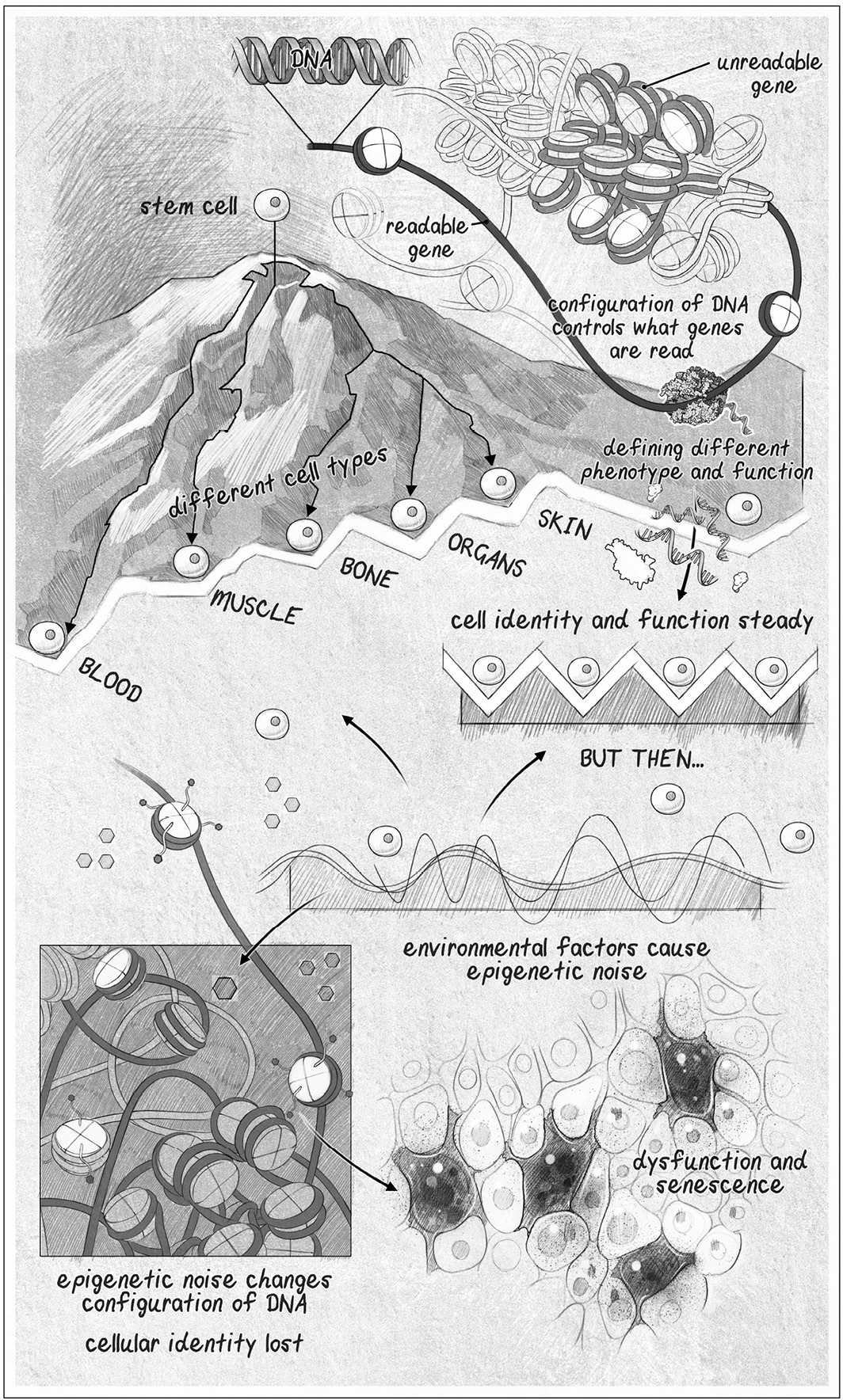
As we saw in the ICE mice, when you disrupt the epigenome by forcing it to
deal with DNA breaks, you introduce noise, leading to an erosion of the
epigenetic landscape. The mice’s bodies turned into chimeras of misguided,
malfunctioning cel s.
THE CHANGING LANDSCAPE OF OUR LIVES. The Waddington landscape is a metaphor for
how cel s nd their identity. Embryonic cel s, often depicted as marbles, rol downhil and land
in the right val ey that dictates their identity. As we age, threats to survival, such as broken DNA,
activate the survival circuit and rejigger the epigenome in smal ways. Over time, cel s
progressively move towards adjacent val eys and lose their original identity, eventual y
transforming into zombielike senescent cel s in old tissues.
That’s aging. This loss of information is what leads each of us into a world of
heart disease, cancer, pain, frailty, and death.
If the loss of analog information is the singular reason why we age, is there
anything we can do about it? Can we stabilize the marbles, keeping the val ey
wal s high and the gravity strong?
Yes. I can say with con dence that there is.
REVERSAL COMES OF AGERegular exercise “is a commitment,” says Benjamin Levine, a professor at the
University of Texas. “But I tel people to think of exercise as part of personal
hygiene, like brushing their teeth. It should be something we do as a matter of
course to keep ourselves healthy. ”41
I’m sure he’s right. Most people would exercise a lot more if going to the gym
were as easy as brushing their teeth.
Perhaps one day it wil be. Experiments in my lab indicate it is possible.
“David, we’ve got a problem,” a postdoctoral researcher named Michael
Bonkowski told me one morning in the fal of 2017 when I arrived at the lab.
That’s seldom a good way to start the day.
“Okay,” I said, taking a deep breath and preparing for the worst. “What is it?”
“The mice,” Bonkowski said. “They won’t stop running.”
The mice he was talking about were 20 months old. That’s roughly the
equivalent of a 65-year-old human. We had been feeding them a molecule
intended to boost the levels of NAD, which we believed would increase the
activity of sirtuins. If the mice were developing a running addiction, that would
be a very good sign.
“But how can that be a problem?” I said. “That’s great news!”
“Wel ,” he said, “it would be if not for the fact that they’ve broken our
treadmil .”
As it turned out, the treadmil tracking program had been set up to record a
mouse running for only up to three kilometers. Once the old mice got to that
point, the treadmil shut down. “We’re going to have to start the experiment
again,” Bonkowski said.
It took a few moments for that to sink in.
A thousand meters is a good, long run for a mouse. Two thousand meters—
ve times around a standard running track—would be a substantial run for a
young mouse.
But there’s a reason why the program was set to three kilometers. Mice simply
don’t run that far. Yet these elderly mice were running ultramarathons.
Why? One of our key ndings, in a study we published in 2018, 42 was that when treated with an NAD-boosting molecule that activated the SIRT1enzyme, the elderly mice’s endothelial cel s, which line the blood vessels, were
pushing their way into areas of the muscle that weren’t getting very much blood
ow. New tiny blood vessels, capil aries, were formed, supplying badly needed
oxygen, removing lactic acid and toxic metabolites from muscles, and reversing
one of the most signi cant causes of frailty in mice and in humans. That was
how these old mice suddenly became such mighty marathoners.
Because the sirtuins had been activated, the mice’s epigenomes were
becoming more stable. The val ey wal s were growing higher. Gravity was
growing stronger. And Waddington’s marbles were being pushed back to where
they belonged. The lining of the capil aries was responding as if the mice were
exercised. It was an exercise mimetic, the rst of its kind, and a sure sign that
some aspects of age reversal are possible.
We stil don’t know everything about why this happens. We don’t know what
sorts of molecules wil work best for activating sirtuins or in what doses.
Hundreds of di erent NAD precursors have been synthesized, and there are
clinical trials in progress to answer that question and more.
But that doesn’t mean we need to wait to take advantage of al that we’ve
learned about engaging the epigenetic survival circuit and living longer and
healthier lives. We don’t need to wait to take advantage of the Information
Theory of Aging.
There are steps we can take right now to live much longer and much healthier
lives. There are things we can do to slow, stop, and even reverse aspects of aging.
But before we talk about what steps we might take to combat aging, before I
can explain the science-backed interventions that have the greatest promise for
fundamental y changing the way we think about growing old, before we even
begin to talk about the treatments and therapies that wil be game changers for
our species, we need to answer one very important question:
Should we?
THREETHE BLIND EPIDEMIC
IT WAS MAY 10, 2010, and London was abuzz. Chelsea Footbal Club had just won
its fourth national championship by devastating Wigan Athletic, 8–0, on the
nal day of Premier League play. Meanwhile, Gordon Brown announced that he
would be stepping down as prime minister in response to a disastrous
parliamentary result for his Labour Party, which had lost more than ninety seats
in the previous week’s general election.
With the eyes of the English sports world on one part of London and the
attention of the British political universe on another, the goings-on at Carlton
House Terrace were missed by al but the most attentive observers of the
president, council, and fel ows of the Royal Society of London for Improving
Natural Knowledge.
More simply known as the Royal Society, the world’s oldest national
scienti c organization was established in 1660 to promote and disseminate “new
science” by big thinkers of the day such as Sir Francis Bacon, the
Enlightenment’s promulgator of “the prolongation of life.” 1 Be tting its rich
scienti c history, the society has held annual scienti c events ever since.
Highlights have included lectures by Sir Isaac Newton on gravity, Charles
Babbage on his mechanical computer, and Sir Joseph Banks, who had just
arrived back from Australia with a bounty of more than a thousand preserved
plants that were al new to science.
Even today, in a post-Enlightenment world, most of the events at the societyare fascinating if not world changing. But the two-day meeting that commenced
in the spring of 2010 was nothing short of that, for gathered together on that
Monday and Tuesday was a motley group of researchers who were meeting to
discuss an important “new science.”
The gathering had been cal ed by geneticist Dame Linda Partridge,
bioanalytics pioneer Janet Thornton, and molecular neuroscientist Gil ian
Bates, al luminaries in their respective elds. The attendee list was no less
impressive. Cynthia Kenyon spoke about her landmark work on a single
mutation in the IGF-1 receptor gene that had doubled the lifespan of
roundworms by activating DAF-162—work that was rst suggested by Partridge
to be a worm-speci c aberration3 but soon forced her and other leading
researchers to confront long-held beliefs that aging could be control ed by a
single gene. Thomas Nyström, from the University of Gothenburg, reported his
discovery that Sir2 not only is important for genomic and epigenomic stability
in yeast, it also prevents oxidized proteins from being passed on to young
daughter cel s.
Brian Kennedy, a former Guarente student who was about to assume the
presidency of the Buck Institute for Research on Aging, explained the ways in
which genetic pathways that had been similarly conserved in a diverse array of
species were likely to play similar roles in mammalian aging. Andrzej Bartke
from Southern Il inois University, former PhD adviser to Michael “Marathon
Mouse” Bonkowski, talked about how dwarf mice can live up to twice as long as
normal mice, a record. Molecular biologist María Blasco explained how old
mammalian cel s are more likely than young cel s to lose their identity and
become cancerous. And geneticist Nir Barzilai spoke of genetic variants in long-
lived humans and his belief that al aging-related diseases can be substantial y
prevented and human lives can be considerably extended with one relatively easy
pharmaceutical intervention.
Over the course of those two days, nineteen presenting scientists from some
of the best research institutions in the world moved toward a provocative
consensus and began to build a compel ing case that would chal enge
conventional wisdom about human health and disease. Summarizing the
meeting for the society later that fal , the biogerontologist David Gems wouldwrite that advances in our understanding of organismal senescence are al leading
to a momentous singular conclusion: that aging is not an inevitable part of life
but rather a “disease process with a broad spectrum of pathological
consequences. ”4 In this way of thinking, cancer, heart disease, Alzheimer’s, and
other conditions we commonly associate with getting old are not necessarily
diseases themselves but symptoms of something greater.
Or, put more simply and perhaps even more seditiously: aging itself is a
disease.
THE LAW OF HUMAN MORTALITY
If the idea that aging is a disease sounds strange to you, you’re not alone.
Physicians and researchers have been avoiding saying that for a long time. Aging,
we’ve long been told, is simply the process of growing old. And growing old has
long been seen as an inevitable part of life.
We see aging, after al , in nearly everything around us and, in particular, the
things around us that look anything like us. The cows and pigs in our farms age.
The dogs and cats in our homes do, too. The birds in the sky. The sh in the sea.
The trees in the forest. The cel s in our petri dishes. It always ends the same way:
dust to dust.
The connection between death and aging is so strong that the inevitability of
the former governed the way we came to de ne the latter. When European
societies rst began keeping public death certi cates in the 1600s, aging was a
respected cause of death. Descriptions such as “decrepitude” or “feebleness due
to old age” were commonly accepted explanations for death. But according to
the seventeenth-century English demographer John Graunt, who wrote Natural
and Political Observations Mentioned in a Following Index, and Made upon the
Bills of Mortality, so were “fright,” “grief,” and “vomiting.”
As we’ve moved forward in time, we’ve moved away from blaming death on
old age. No one dies anymore from “getting old.” Over the past century, the
Western medical community has come to believe not only that there is always a
more immediate cause of death than aging but that it is imperative to identifythat cause. In the past few decades, in fact, we’ve become rather fussy about this.
The World Health Organization’s International Classification of Diseases, a
list of il nesses, symptoms, and external causes of injury, was launched in 1893
with 161 headings. Today there are more than 14,000, and in most places where
records of death are kept, doctors and public health o cials use these codes to
record both immediate and underlying causes of disability and death.5 That, in
turn, helps medical leaders and policy makers around the globe make public
health decisions. Broadly speaking, the more often a cause shows up on a death
certi cate, the more attention society gives to ghting it. This is why heart
disease, type 2 diabetes, and dementia are major focuses of research and
interventionary medical care, while aging is not, even though aging is the greatest
cause of al those diseases.
Age is sometimes considered an underlying factor at the end of someone’s life,
but doctors never cite it as an immediate reason for death. Those who do run the
risk of raising the ire of bureaucrats, who are prone to send the certi cate back to
the doctor for further information. Even worse, they are likely to endure the
ridicule of their peers. David Gems, the deputy director of the Institute of
Healthy Ageing at University Col ege London and the same man who wrote the
report from the Royal Society meeting on “the new science of aging,” told
Medical Daily in 2015 that “the idea that people die of pure aging, without
pathology, is nuts. ”6
But this misses the point. Separating aging from disease obfuscates a truth
about how we reach the ends of our lives: though it’s certainly important to
know why someone fel from a cli , it’s equal y important to know what
brought that person to the precipice in the rst place.
Aging brings us to the precipice. Give any of us 100 years or so, and it brings
us al there.
In 1825, the British actuary Benjamin Gompertz, a learned member of the
Royal Society, tried to explain this upward limit with a “Law of Human
Mortality,” essential y a mathematical description of aging. He wrote, “It is
possible that death may be the consequence of two general y co-existing causes;
the one, chance, without previous disposition to death or deterioration; theother, a deterioration, or an increased inability to withstand destruction. ”7
The rst part of the law says that there is an internal clock that ticks away at
random, like the chance a glass at a restaurant wil break; essential y a rst-order
rate reaction, similar to radioactive decay, with some glasses lasting far longer
than most. The second part says that, as time passes, due to an unknown
runaway process, humans experience an exponential increase in their probability
of death. By adding these two components together, Gompertz could accurately
predict deaths due to aging: the number of people alive after 50 drops
precipitously, but there is a tail at the end where some “lucky” people remain
alive beyond what you’d expect. His equations made his relatives, Sir Moses
Monte ore and Nathan Mayer Rothschild, owners of the Al iance Insurance
Company, a lot of money.
What Gompertz could not have known, but would have appreciated, is that
most organisms obey his law: ies, roundworms, mice, even yeast cel s. For larger
organisms, we don’t know exactly what the two clocks are, but we do know in
yeast cel s: the chance clock is the formation of an rDNA circle, and the
exponential clock is the replication and exponential increase in the numbers of
rDNA circles, with the resulting movement of Sir2 away from the silent mating-
type genes that causes sterility.8
Humans are more complicated, but in the nineteenth century, British
mortality rates were becoming amenable to simple mathematical modeling
because they were increasingly avoiding not-from-aging deaths: childbirth,
accidents, and infections. This increasingly revealed the underlying and
exponential incidence of death due to internal clocks as being the same as it ever
was. During those times, the probability of dying doubled every eight years, an
equation that left very little room for survivors after the age of 100.
That cap has general y held true ever since, even as the global average life
expectancy jumped twenty years between 1960 and today. 9 That’s because al
that doubling adds up quickly. So even though most people who live in
developed nations can now feel con dent that they wil make it to 80, these days
the chances that any of us wil reach a century is just 3 in 100. Getting to 115 is a
1-in-100-mil ion proposition. And reaching 130 is a mathematical improbabilityof the highest order.
At least it is right now.
THE MORTAL BREEZE
Back in the mid-1990s, when I was pursuing my PhD at Australia’s University of
New South Wales, my mother, Diana, was found to have a tumor the size of an
orange in her left lung.
As she was a lifelong smoker, I’d suspected it was coming. It was the one
thing we had argued about more than anything else. When I was a young boy, I
used to steal her cigarettes and hide them. It infuriated her. The fact that she
didn’t respond to my pleas to stop smoking infuriated me, too.
“I have lived a good life. The rest is a bonus,” she would say to me in her early
40s.“Do you know how lucky you are to have been born? You’re throwing your
life away! I won’t come visit you in hospital when you get cancer,” I would say.
When the cancer nal y arrived about a decade later, I wasn’t angry. Tragedy
has a way of vanquishing anger. I drove to the hospital, determined to solve any
problem.
My mother was responsible for her own actions, but she was also a victim of
an unscrupulous industry. Tobacco alone doesn’t kil people; it’s the
combination of tobacco, genetics, and time that most often leads to death. She
was diagnosed with cancer at the age of 50. That’s twenty-one years earlier than
the rst diagnosis in the average lung cancer patient. It’s also how old I am now.
In one way of thinking, my mother was unfortunate to develop cancer at
such a young age. After her back was opened up, rows of ribs were cut from her
spine, and major arteries were rerouted, she lived the rest of her life with just one
lung, which certainly impacted her quality of life and ensured that she had only
a few years of good life left.
On the genetics front, my mother was also unfortunate. Everyone in my
family, from my grandmother to my youngest son, has had their genes analyzed
by one of the companies that o er these services. When my mother had hersdone, she learned, albeit after she had cancer, that she had inherited a mutation
in the SERPINA1 gene, which is implicated in chronic obstructive pulmonary
disease or emphysema. That meant her clock was ticking even faster. After her
left lung was removed, her right lung was the sole provider of oxygen, but the
de ciency in SERPINA1 meant that white blood cel s attacked her remaining
lung, destroying the tissue as if it were an invader. Eventual y the lung gave out.10
In another way of thinking, though, my mother was very lucky—she had the
come-to-God moment that many smokers need to go to battle with the
tremendously powerful forces of addiction in time to save herself, and she spent
another two decades on this planet. She traveled the world, visiting eighteen
di erent countries. She met her grandchildren. She saw me give a TED Talk at
the Sydney Opera House. For this we must certainly credit the doctors who
removed her cancerous lung, but we should also acknowledge the positive
impact of her age. One of the best ways to predict whether someone wil survive
a disease, after al , is to take a look at how old he or she is when diagnosed—and
my mother was, relatively speaking, very young.
This is key. We know that smoking accelerates the aging clock and makes you
more likely to die than a nonsmoker—15 years earlier, on average. So, we have
fought it with public health campaigns, class action lawsuits, taxes on tobacco
products, and legislation. We know that cancer makes you more likely to die,
and we’ve fought it with bil ions of dol ars’ worth of research aimed at ending it
once and for al .
We know that aging makes you more likely to die, too, but we’ve accepted it
as part of life.
It’s also worth noting that even before my mother was diagnosed with lung
cancer—indeed, even before the cancerous cel s in her lungs began growing out
of control—she was aging. And in that way, of course, she was hardly unique.
We know that the process of aging begins long before we notice it. And with the
unfortunate exceptions of those whose lives are taken by the early onset of a
hereditary ailment or a deadly pathogen, most people begin to experience at least
some of the e ects of aging long before they are impacted by the accumulation
of diseases we commonly associate with growing old. At the molecular level, this
starts to happen at a time in our lives that many of us stil look and feel young.Girls who go through puberty earlier than normal, for example, have an
accelerated epigenetic clock. At that age, we can’t hear the mistakes of the
concert pianist.11 But they are there, even as a teenager.
In our 40s and 50s, we don’t often think about what it feels like to grow old.
When I give talks about my research, sometimes I bring an “age suit” and ask a
young volunteer to wear it. A neck brace reduces mobility in the neck, lead-lined
jackets and wraps al over the body simulate weak muscles, earplugs reduce
hearing, and ski goggles simulate cataracts. After a few minutes of walking
around in the suit, the test subject is very relieved to take it o —and fortunately
can do so.
“Imagine wearing it for a decade,” I say.
To put yourself into an aged mind-set, try this little experiment. Using your
nondominant hand, write your name, address, and phone number while circling
your opposite foot counterclockwise. That’s a rough approximation of what it
feels like.
Di erent functions peak at di erent times for di erent people, but physical
tness, in general, begins to decline in our 20s and 30s. Men who run middle-
distance races, for instance, are fastest around the age of 25, no matter how hard
they train after that. The best female marathoners can stay competitive wel into
their late 20s and early 30s, but their times begin to rise quickly after 40.
Occasional y, exceptional y t outliers—such as National Footbal League
quarterback Tom Brady, National Women’s Soccer League defender Christie
Pearce, Major League Basebal out elder Ichiro Suzuki, and tennis legend
Martina Navratilova—demonstrate that professional athletes can stay
competitive into their 40s, but almost no one remains at the highest levels of
these or most other professional sports much past their mid-40s. Even someone
as resilient as Navratilova peaked when she was in her early 20s through her early
30s.There are some simple tests to determine how biologicaly old you probably
are. The number of push-ups you can do is a good indicator. If you are over 45
and can do more than twenty, you are doing wel . The other test of age is the
sitting-rising test (SRT). Sit on the oor, barefooted, with legs crossed. Lean
forward quickly and see if you can get up in one move. A young person can. Amiddle-aged person typical y needs to push o with one of their hands. An
elderly person often needs to get onto one knee. A study of people 51 to 80 years
found that 157 out of 159 people who passed away in 75 months had received
less than perfect SRT scores.
Physical changes happen to everyone. Our skin wrinkles. Our hair grays. Our
joints ache. We start groaning when we get up. We begin to lose resilience, not
just to diseases but to al of life’s bumps and bruises.
Fortunately, a hip fracture for a teenager is a very rare event that nearly
everyone is expected to bounce back from. At 50, such an injury could be a life-
altering event but general y not a fatal one. It’s not long after that, though, that
the risk factor for people who su er a broken hip becomes terrifyingly high.
Some reports show that up to half of those over the age of 65 who su er a hip
fracture wil die within six months.12 And those who survive often live the rest
of their lives in pain and with limited mobility. At 88, my grandmother Vera
tripped on a rumpled carpet and broke her upper femur. During surgery to
repair the damage, her heart stopped on the operating table. Though she
survived, her brain had been starved for oxygen. She never walked again and died
a few years later.
Wounds also heal much more slowly with age—a phenomenon rst
scienti cal y studied during World War I by the French biophysicist Pierre
Lecomte du Noüy, who noted a di erence in the rate of healing between
younger and older wounded soldiers. We can see this in even starker relief when
we look at the di erences in the ways children and the elderly heal from wounds.
When a child gets a cut on her foot, a noninfected wound wil heal quite
quickly. The only medicine most kids need when they get hurt like this is a kiss, a
Band-Aid, and some assurance that everything wil be okay. For an elderly
person, a foot injury is not just painful but dangerous. For older diabetics, in
particular, a smal wound can be deadly: The ve-year mortality rate for a foot
ulcer in a diabetic is greater than 50 percent. That’s higher than the death rates
for many kinds of cancer. 13
Chronic foot wounds, by the way, are not rare; we just don’t hear much
about them. They almost always begin with seemingly benign rubbing on
increasingly numb and fragile soles—but not always. My friend DavidArmstrong, at the University of Southern California, a passionate advocate for
increasing our focus on preventing diabetic foot injuries, often tel s the story of
one of his patients, who had a nail stuck in his foot for four days. The patient
noticed it only because he wondered where the tapping sound on the oor was
coming from.
Smal and large diabetic foot wounds rarely heal. They can look as though
someone has taken an apple corer to the bal s of both feet. The body doesn’t
have enough blood ow and cel regeneration capacity, and bacteria thrive in this
meaty, moist environment. Right now, 40 mil ion people, bedridden and
waiting for death, are living this nightmare. There’s almost nothing that can be
done for them except to cut back the dead and dying tissue, then cut some more,
and then some more. From there, robbed of upright mobility, misery is your
bedfel ow and thankful y death is nigh. In the United States alone, each year,
82,000 elderly people have a limb amputated. That’s ten every hour. Al this
pain, al this cost, comes from relatively minor initial injuries: foot wounds.
The older we get, the less it takes for an injury or il ness to drive us to our
deaths. We are pushed closer and closer to the precipice until it takes nothing
more than a gentle wind to send us over. This is the very de nition of frailty.
If hepatitis, kidney disease, or melanoma did the sorts of things to us that
aging does, we would put those diseases on a list of the deadliest il nesses in the
world. Instead, scientists cal what happens to us a “loss of resilience,” and we
general y have accepted it as part of the human condition.
There is nothing more dangerous to us than age. Yet we have conceded its
power over us. And we have turned our ght for better health in other
directions.
WHACK-A-MOLE MEDICINE
There are three large hospitals within a few minutes walk of my o ce. Brigham
and Women’s Hospital, Beth Israel Deaconess Medical Center, and Boston
Children’s Hospital are focused on di erent patient populations and medicalspecialties, but they’re al set up the same way.
If we were to take a walk into the lobby of Brigham and Women’s and head
over to the sign by the elevator, we’d get a lay of this nearly universal medical
landscape. On the rst oor is wound care. Second oor: orthopedics. Third
oor: gynecology and obstetrics. Fourth oor: pulmonary care.
At Boston Children’s, the di erent medical specialties are similarly separated,
though they are labeled in a way more be tting the young patients at this
amazing hospital. Fol ow the signs with the boats for psychiatry. The owers wil
take you to the cystic brosis center. The sh wil get you to immunology.
And now over to Beth Israel. This way to the cancer center. That way to
dermatology. Over here for infectious diseases.
The research centers that surround these three hospitals are set up in much
the same way. In one lab you’l nd researchers working to cure cancer. In
another they’re ghting diabetes. In yet another they’re working on heart
disease. Sure, there are geriatricians, but they almost always take care of the
already sick, thirty years too late. They treat the aged—not the aging. No
wonder so few doctors today are choosing to specialize in this area of medicine.
There’s a reason why hospitals and research institutions are organized in this
way. Most of our modern medical culture has been built to address medical
problems one by one—a segregation that owes itself in no smal part to our
obsession with classifying the speci c pathologies leading to death.
There was nothing wrong with this setup when it was established hundreds
of years ago. And by and large, it stil works today. But what this approach
ignores is that stopping the progression of one disease doesn’t make it any less
likely that a person wil die of another. Sometimes, in fact, the treatment for one
disease can be an aggravating factor for another. Chemotherapy can cure some
forms of cancer, for instance, but it also makes people’s bodies more susceptible
to other forms of cancer. And as we learned in the case of my grandmother Vera,
something as seemingly routine as orthopedic surgery can make patients more
susceptible to heart failure.
Because the stakes are so exceptional y high for the individual patients being
treated in these places, a lot of people don’t recognize that a battle won on any of
these individual fronts won’t make much of a di erence against the Law ofHuman Mortality. Surviving cancer or heart disease doesn’t substantial y
increase the average human lifespan, it just decreases the odds of dying of cancer
or heart disease.
The way doctors treat il ness today “is simple,” wrote S. Jay Olshansky, a
demographer at the University of Il inois. “As soon as a disease appears, attack
that disease as if nothing else is present; beat the disease down, and once you
succeed, push the patient out the door until he or she faces the next chal enge;
then beat that one down. Repeat until failure. ”14
The United States spends hundreds of bil ions of dol ars each year ghting
cardiovascular disease.15 But if we could stop al cardiovascular disease—every
single case, al at once—we wouldn’t add many years to the average lifespan; the
gain would be just 1.5 years. The same is true for cancer; stopping al forms of
that scourge would give us just 2.1 more years of life on average, because al other
causes of death stil increase exponential y. We’re stil aging, after al .
Aging in its nal stages is nothing like a bushwalk, where a bit of rest, a drink
of water, a nutritional bar, and some fresh socks can get you another dozen miles
before sunset. It’s more like a fast sprint over an ever-higher and ever-closer set of
hurdles. One of those hurdles wil eventual y send you for a tumble. And once
you’ve fal en one time, if you do get up, the odds of fal ing again just keep
getting higher. Take away one hurdle, and the path forward is real y no less
precarious. That’s why the current solutions, which are focused on curing
individual diseases, are both very expensive and very ine ective when it comes to
making big advances in prolonging our healthspans. What we need are
medicines that knock down all the hurdles.
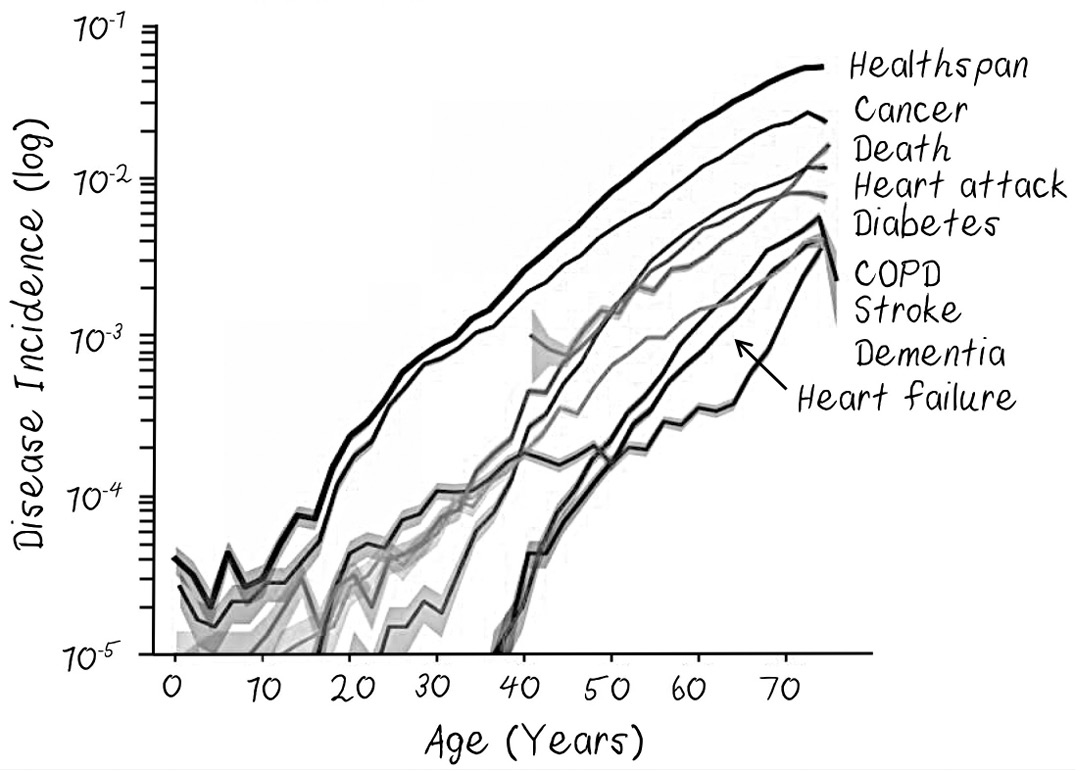
WHY TREATING ONE DISEASE AT A TIME HAS LITTLE IMPACT ON LIFESPAN. The graph
shows an exponential increase in disease as each year passes after the age of 20. It’s hard to appreciate exponential graphs. If I were to draw this graph with a linear Y-axis, it would be two
stories tal . What this means is your chance of developing a lethal disease increases by a
thousandfold between the ages of 20 and 70, so preventing one disease makes little di erence to
lifespan.
Source: Adapted from A. Zenin, Y. Tsepilov, S. Sharapov, et al., “Identi cation of 12 Genetic
Loci Associated with Human Healthspan,” Communications Biology 2 (January 2019).
Thanks to statins, triple-bypass surgeries, de bril ators, transplants, and other
medical interventions, our hearts are staying alive longer than ever. But we
haven’t been nearly so attentive to our other organs, including the most
important one of al : our brains. The result is that more of us are spending more
years su ering from brain-related maladies, such as dementia.
Eileen Crimmins, who studies health, mortality, and global aging at the
University of Southern California, has observed that even though average
lifespans in the United States have increased in recent decades, our healthspans
have not kept up. “We have reduced mortality more than we prevented
morbidity,” she wrote in 2015. 16
So prevalent is the combined problem of early mortality and morbidity that
there is a statistic for it: the disability-adjusted life year, or DALY, which
measures the years of life lost from both premature death and poor state of
health. The Russian DALY is the highest in Europe, with twenty- ve lost years
of healthy life per person. In Israel, it is an impressive ten years. In the United
States, the number is a dismal twenty-three. 17
The average age of death can vary rather signi cantly over time, and isa ected by many factors, including the prevalence of obesity, sedentary lifestyles,
and drug overdoses. Similarly, the very idea of poor health is both subjective and
measured di erently from place to place, and so researchers are divided on
whether the DALY is rising or declining in the United States. But even the more
optimistic assessments suggest that the numbers have largely been static in recent
years. To me, that in itself is an indictment of the US system; like other advanced
countries, we should be making tremendous progress toward reducing the
DALY and other measures of morbidity, yet, at best, it seems we’re treading
water. We need a new approach.
It doesn’t take studies and statistics to know what’s happening, though. It’s
al around us, and the older we get, the more obvious it becomes. We get to 50
and begin to notice we look like our parents, with graying hair and an increasing
number of wrinkles. We get to 65, and if we haven’t faced some form of disease
or disability yet, we consider ourselves fortunate. If we’re stil around at 80, we
are almost guaranteed to be combating an ailment that has made life harder, less
comfortable, and less joyful. One study found that 85-year-old men are
diagnosed with an average of four di erent diseases, with women of that age
su ering from ve. Heart disease and cancer. Arthritis and Alzheimer’s. Kidney
disease and diabetes. Most patients have several additional undiagnosed diseases,
including hypertension, ischemic heart disease, atrial bril ation, and
dementia.18 Yes, these are di erent ailments with di erent pathologies, studied
in di erent buildings at the National Institutes of Health and in di erent
departments within universities.
But aging is a risk factor for al of them.
In fact, it’s the risk factor. Truly, by comparison, little else matters.
The nal years of my mother’s life serve as a good example. Like almost
everyone else, I recognized that smoking would increase my mother’s chances of
getting lung cancer. I also knew why: cigarette smoke contains a chemical cal ed
benzo(a)pyrene, which binds to guanine in DNA, induces double-strand breaks,
and causes mutations. The repair process also causes epigenetic drift and
metabolic changes that cancer cel s thrive on, in a process we’ve cal ed
geroncogenesis.19
The combination of genetic and epigenetic changes induced by years ofexposure to cigarette smoke increases the chances of developing lung cancer
about vefold.
That’s a big increase. And because of it—and the devastatingly high health
costs associated with treating cancer—the majority of the world’s nations
sponsor smoking cessation programs. Most countries also put health warnings
on cigarette packaging, some with horri c color pictures of tumors and
blackened extremities. Most countries have passed laws against certain kinds of
tobacco advertising. And most have sought to decrease consumption through
punitive taxes.20
Al of that to prevent a vefold increase in a few kinds of cancer. And having
watched my mother su er from that kind of cancer, I’l be the rst to say it’s
total y worth it. From both an economic and emotional point of view, these are
good investments.
But consider this: though smoking increases the risk of getting cancer
vefold, being 50 years old increases your cancer risk a hundredfold. By the age
of 70, it is a thousandfold. 21
Such exponential y increasing odds also apply to heart disease. And diabetes.
And dementia. The list goes on and on. Yet there is not a country in the world
that has committed any signi cant resources to help its citizens combat aging. In
a world in which we seem to agree on very little, the feeling that “it’s just the way
it goes” is almost universal.
A GLORIOUS FIGHT
Aging results in physical decline.
It limits the quality of life.
And it has a speci c pathology.
Aging does al this, and in doing so it ful l s every category of what we cal a
disease except one: it impacts more than half the population.
According to The Merck Manual of Geriatrics, a malady that impacts less
than half the population is a disease. But aging, of course, impacts everyone. The
manual therefore cal s aging an “inevitable, irreversible decline in organ functionthat occurs over time even in the absence of injury, il ness, environmental risks,
or poor lifestyle choices.”
Can you imagine saying that cancer is inevitable and irreversible? Or
diabetes? Or gangrene?
I can. Because we used to say that.
Al of these may be natural problems, but that doesn’t make them inevitable
and irreversible—and it sure doesn’t make them acceptable. The manual is
wrong about aging.
But being wrong has never stopped conventional wisdom from negatively
impacting public policy. And because aging isn’t a disease by the commonly
accepted de nition, it doesn’t t nicely into the system we’ve built for funding
medical research, drug development, and the reimbursement of medical costs by
insurance companies. Words matter. De nitions matter. Framing matters. And
the words, de nitions, and framing we use to describe aging are al about
inevitability. We didn’t just throw in the towel before the ght began, we threw
it in before we even knew there was a ght to be had.
But there is a ght. A glorious and global one. And, I think, a winnable one.
There’s no good reason why we have to say that something that happens to
49.9 percent of the population is a disease while something that happens to 50.1
percent of the population is not. In fact, that’s a backward way of approaching
problems that lends itself to the whack-a-mole system of medicine we’ve set up
in hospitals and research centers around the world.
Why would we choose to focus on problems that impact smal groups of
people if we could address the problem that impacts everyone—especial y if, in
doing so, we could signi cantly impact al those other, smal er problems?
We can.
I believe that aging is a disease. I believe it is treatable. I believe we can treat it
within our lifetimes. And in doing so, I believe, everything we know about
human health wil be fundamental y changed.
If you are not yet convinced that aging is a disease, I want to let you in on a
secret. I have a window into the future. In 2028, a scientist wil discover a new
virus, cal ed LINE-1. It wil turn out that we are al infected with it. We get it
from our parents. It wil turn out that the LINE-1 virus is responsible for mostother major diseases: diabetes, heart disease, cancer, dementia. It causes a slow,
horrible chronic disorder, and al humans eventual y succumb to it, even if they
have a low-grade infection. Fortunately, the world pours bil ions of dol ars into
nding a cure. In 2033, a company wil succeed in making a vaccine that
prevents LINE-1 infections. New generations who are vaccinated at birth wil
live fty years longer than their parents did—it wil turn out that that’s our
natural lifespan and we had no idea. The new generation of healthy humans wil
pity previous generations, who blindly accepted that physical decline at 50 was
natural and an 80-year life was a life wel lived.
Of course, this is a science ction story I just invented. But it might be truer
than you think.
A few recent studies have suggested that the so-cal ed sel sh genes we al carry
in our genome, actual y cal ed LINE-1 elements, replicate and cause cel ular
havoc as we get older, accelerating our physical demise. We’l discuss them in
more detail later, but for now, it’s the idea I want to focus on because it raises
important questions: Does it matter whether LINE-1 comes from your parents
directly or via a virus? Would you want to eradicate LINE-1 from humanity or
let it grow in your kids and in ict horrible diseases on them? Would you say that
LINE-1 causes a disease or not?
If not, is it simply because more than half of al people carry it?
Whether it’s a virus, a sel sh DNA element, or simply the makeup of our
cel s that causes these health problems, what’s the di erence? The end result is
the same.
The belief that aging is a natural process is deep-rooted. So even if I’ve
somewhat convinced you that aging should be considered a disease, let’s do
another thought experiment.
Imagine that everyone on our planet typical y lives to 150 years in good
health. Your family, though, doesn’t. You become wrinkled, gray-haired,
diabetic, and frail at 80. Upon seeing these poor, unfortunate souls in this poor,
unfortunate state of existence, what doctor would not diagnose your family with
a disease, name it after him- or herself, and publish horrid photos of you with
your eyes blacked out in medical journals? Communities would raise money tounderstand and nd a cure for your family’s wretched inheritance.
That was exactly what happened when the German physician Otto Werner
rst described a condition that causes people to look and feel as though they are
80 when they are in their 40s. That’s Werner syndrome, the disease I was
studying when I rst arrived at MIT in the 1990s. Nobody said I was studying
something that is inevitable or irreversible. Nobody said it was crazy to cal
Werner syndrome a disease or to work to nd a breakthrough therapy. Nobody
told me or the Werner patients that “that’s just the way it goes.”
In front of us is the deadliest and costliest disease on the planet, a disease that
almost no one is working on. It is as if the planet is in a stupor. If your rst
thought is “But I don’t want to live past 90,” let me assure you: I don’t want you
to live a year longer than you wish.
But before you make your decision, let’s do one nal thought experiment.
Imagine that a clerk at City Hal has found a mistake on your birth
certi cate. It turns out that you are actual y 92 years old.
“You’l get a new one in the mail,” the clerk says. “Have a nice day.”
Do you feel any di erent now that you are 92? Nothing else has changed in
your life—just a few numbers on your identi cation. Do you suddenly want to
kil yourself?
Of course not. When we stay healthy and vibrant, as long as we feel young
physical y and mental y, our age doesn’t matter. That’s true whether you are 32,
52, or 92. Most middle-aged and older adults in the United States report feeling
ten to twenty years younger than their age, because they stil feel healthy. And
feeling younger than your age predicts lower mortality and better cognitive
abilities later in life.22 It’s a virtuous cycle, as long as you keep pedaling.
But no matter how you feel at this moment in your life, even with a positive
outlook and a healthy lifestyle, you have a disease. And it’s going to catch up to
you, sooner rather than later, unless something is done.
I acknowledge that cal ing aging a disease is a radical departure from the
mainstream view of health and wel -being, which has established an array of
medical interventions addressing the various causes of death. That framework
evolved, however, largely because we didn’t understand why aging occurs. Up
until very recently, the best thing we had was a list of aging hal marks. TheInformation Theory of Aging could change that.
There is nothing wrong with using the hal marks to guide interventions. We
can probably have a positive impact on people’s lives by addressing each of them.
It’s possible that interventions aimed at slowing telomere deterioration wil
improve people’s long-term wel -being. Maintaining proteostasis, preventing
deregulation of nutrient sensing, thwarting mitochondrial dysfunction,
stopping senescence, rejuvenating stem cel s, and decreasing in ammation might
al be ways to delay the inevitable. Indeed, I work with students, postdocs, and
companies around the globe that are developing solutions to each one of these
hal marks and hope to continue. 23 Anything we can do to al eviate su ering we
should do.
But we’re stil building nine dams on nine tributaries.
In coming together to tackle the “new science of aging,” as the attendees of
the Royal Society meeting termed this ght in their 2010 meeting, increasing
numbers of scientists are starting to acknowledge the possibility and potential
inherent in heading upstream.
Together we can build a single dam—at the source. Not just intervene when
things go wrong. Not just slow things down. We can eliminate the symptoms of
aging altogether.
This disease is treatable.
PART IIWHAT WE’RE LEARNING
(THE PRESENT)
FOURLONGEVITY NOW
EVERY DAY I WAKE UP to an inbox ful of messages from people from al over the
world. The tide ebbs and ows but always takes the form of a ash ood in the
wake of newly announced research from my team or others.
“What should I be taking?” they ask.
“Can you tel me what I need to do to get admitted into one of the human
trials?” they implore.
“Can you extend the lifespan of my daughter’s hamster?” I kid you not.
Some of the letters are much sadder than others. One man recently wrote to
o er to contribute a donation to my lab in honor of his mother, who had passed
away after su ering terribly through many years of age-related il ness. “I feel
compel ed to help, even in some smal way, to prevent this from happening to
someone else,” he wrote. The next day, a woman whose father had been
diagnosed with Alzheimer’s wrote to ask if there were any way to get him
admitted into a study. “I would do anything, take him anywhere, spend every
last cent I have,” she pleaded. “He is the only family I have and I cannot bear the
thought of what is about to happen to him.”
There is great reason for hope on the not-so-distant horizon, but those
battling against the ravages of aging right now must do so in a world in which
most doctors have never even thought about why we age, let alone how to treat
aging.
Some of the medical therapies and life-extending technologies discussed inthis book are already here. Others are a few years away. And there are more to
discuss that are a decade or so down the road; we’l get to those as wel .
But even without access to this developing technology, no matter who you
are, where you live, how old you are, and how much you earn, you can engage
your longevity genes, starting right now.
That’s what people have been doing for centuries—without even knowing it
—in centenarian-heavy places such as Okinawa, Japan; Nicoya, Costa Rica; and
Sardinia, Italy. These are, you might recognize, some of the places the writer Dan
Buettner introduced to the world as so-cal ed Blue Zones starting in the mid-
2000s. Since that time, the primary focus for those seeking to apply lessons from
these and other longevity hot spots has been on what Blue Zone residents eat.
Ultimately this resulted in the distil ation of “longevity diets” that are based on
the commonalities in the foods eaten in places where there are lots of
centenarians. And overwhelmingly that advice comes down to eating more
vegetables, legumes, and whole grains, while consuming less meat, dairy
products, and sugar.
And that’s not a bad place to start—in fact, it’s a great place to start. There is
widespread disagreement, even among the best nutritionists in the world, as to
what constitutes the “best” diet for H. sapiens. That’s likely because there is no
best diet; we’re al di erent enough that our diets need to be subtly and
sometimes substantial y di erent, too. But we’re also al similar enough that
there are some very broad commonalities: more veggies and less meat; fresh food
versus processed food. We al know this stu , though applying it can be a
chal enge.
A big part of the reason so many people aren’t wil ing to face up to that
chal enge is because we’ve always thought of aging as an inevitable part of life. It
might come a little earlier for some and a little later for others, but we’ve always
been told that it’s coming for us al .
That’s what we used to say about pneumonia, in uenza, tuberculosis, and
gastrointestinal conditions, too. In 1900, those four il nesses accounted for
about half of the deaths in the United States and—if you managed to live long
enough—you could be virtual y assured that one of them would get youeventual y.
Today, deaths among people su ering from tuberculosis and gastrointestinal
conditions are exceedingly rare. And pneumonia and in uenza claim less than 10
percent of the lives taken by those conditions a little more than a century ago—
with most of those deaths now among individuals weakened by aging.
What changed? In no smal part it was framing. Advances in medicine,
innovations in technology, and better information to guide our lifestyle
decisions resulted in a world in which we didn’t have to accept the idea that
these diseases were “just the way it goes.”
We don’t have to accept aging like that, either.
But even among those who wil have the most immediate access to
pharmaceuticals and technologies that wil be emerging to o er longer and
healthier lives in the next few decades, reaching an optimal lifespan and
healthspan won’t be as easy as ipping a switch.
There wil always be good and bad choices. And that starts with what we put
into our bodies.
And what we don’t.
GO, FAST
After twenty- ve years of researching aging and having read thousands of
scienti c papers, if there is one piece of advice I can o er, one sure re way to stay
healthy longer, one thing you can do to maximize your lifespan right now, it’s
this: eat less.
This is nothing revolutionary, of course. As far back as Hippocrates, the
ancient Greek physician, doctors have been espousing the bene ts of limiting
what we eat, not just by rejecting the deadly sin of gluttony, as the Christian
monk Evagrius Ponticus counseled in the fourth century, but through
“intentional asceticism.”
Not malnutrition. Not starvation. These are not pathways to more years, let
alone better years. But fasting—al owing our bodies to exist in a state of want,
more often than most of us al ow in our privileged world of plenty—isunquestionably good for our health and longevity.
Hippocrates knew this. Ponticus knew this. So, too, did Luigi Cornaro, a
fteenth-century Venetian nobleman who could, and probably should, be
considered the father of the self-help book.
The son of an innkeeper, Cornaro made a fortune as an entrepreneur and
lavishly spent his money on wine and women. By his mid-30s, he was exhausted
by food, drink, and sex—the poor guy—and resolved to limit himself in each
regard. The historical record is a bit vague on the details of his sex life after that
fateful decision,1 but his diet and drinking habits have been wel documented:
he ate no more than twelve ounces of food and drank two glasses of wine each
day.“I accustomed myself to the habit of never fuly satisfying my appetite, either
with eating or drinking,” Cornaro wrote in his First Discourse on the Temperate
Life, “always leaving the table wel able to take more.” 2
Cornaro’s discourses on the bene ts of la vita sobria might have fal en into
obscurity had he not provided such compel ing personal proof that his advice
had merit: he published his guidance when he was in his 80s, and in exceptional
health, no less, and he died in 1566 at nearly (and some sources say more than)
100 years old.
In more recent times, Professor Alexandre Guéniot, the president of the Paris
Medical Academy just after the turn of the twentieth century, was famed for
living on a restricted diet. It is said that his contemporaries mocked him—for
there was no science at that time to back his suspicion that hunger would lead to
good health, just his gut hunch—but he outlived them, one and al . He nal y
succumbed at the age of 102.
The rst modern scienti c explorations of the lifelong e ects of a severely
restricted diet began during the last days of World War I. That’s when the
longtime biochemical col aborators Lafayette Mendel and Thomas Osborne—
the duo who had discovered vitamin A—discovered, along with researcher Edna
Ferry, that female rats whose growth was stunted due to lack of food early in life
lived much longer than those that ate plenty. 3
Picking up on that evidence in 1935, a now-famous Cornel Universityprofessor named Clive McCay demonstrated that rats fed a diet containing 20
percent indigestible cel ulose—cardboard, essential y—lived signi cantly longer
lives than those that were fed a typical lab diet. Studies conducted over the next
eighty years demonstrated again and again that calorie restriction without
malnutrition, or CR, leads to longevity for al sorts of life-forms. Hundreds of
mouse studies have been done since to test the e ects of calories on health and
lifespan, mostly on male mice.
Reducing calories works even in yeast. I rst noticed this in the late 1990s.
Cel s fed with lower doses of glucose were living longer, and their DNA was
exceptional y compact—signi cantly delaying the inevitable ERC
accumulation, nucleolar explosion, and sterility.
If this happened only in yeast, it would merely be interesting. But because we
knew that rodents also lived longer when their food was restricted—and later
learned that this was the case for fruit ies, as wel 4—it was apparent that this
genetic program was very old, perhaps nearly as old as life itself.
In animal studies, the key to engaging the sirtuin program appears to be
keeping things on the razor’s edge through calorie restriction—just enough food
to function in healthy ways and no more. This makes sense. It engages the
survival circuit, tel ing longevity genes to do what they have been doing since
primordial times: boost cel ular defenses, keep organisms alive during times of
adversity, ward o disease and deterioration, minimize epigenetic change, and
slow down aging.
But this has, for obvious reasons, proven a chal enge to test on humans in a
control ed scienti c setting. Sadly, it’s not hard to nd instances in which
humans have had to go without food, but those periods are general y times in
which food insecurity results in malnutrition, and it would be a chal enge to
keep a test group of humans on the razor’s edge for the long periods of time that
would be required for comprehensive control ed studies.
As far back as the 1970s, though, there have been observational studies that
strongly suggested long-term calorie restriction could help humans live longer
and healthier lives, too.
In 1978 on the island of Okinawa, famed for its large number ofcentenarians, bioenergetics researcher Yasuo Kagawa learned that the total
number of calories consumed by schoolchildren was less than two-thirds of what
children were getting in mainland Japan. Adult Okinawans were also leaner,
taking in about 20 percent fewer calories than their mainland counterparts.
Kagawa noted that not only were the lifespans of Okinawans longer, but their
healthspans were, too—with signi cantly less cerebral vascular disease,
malignancy, and heart disease.5
In the early 1990s, the Biosphere 2 research experiment provided another
piece of evidence. For two years, from 1991 to 1993, eight people lived inside a
three-acre, closed ecological dome in southern Arizona, where they were
expected to be reliant on the food they were growing inside. Green thumbs they
weren’t, though, and the food they farmed turned out to be insu cient to keep
the participants on a typical diet. The lack of food wasn’t bad enough to result
in malnutrition, but it did mean that the team members were frequently hungry.
One of the prisoners (and by “prisoners” I mean “experimental subjects”)
happened to be Roy Walford, a researcher from California whose studies on
extending life in mice are stil required reading for scientists entering the aging
eld. I have no reason to suspect that Walford sabotaged the crops, but the
coincidence was rather fortuitous for his research; it gave him an opportunity to
test his mouse-based ndings on human subjects. Because they were thoroughly
medical y monitored before, during, and after their two-year stint inside the
dome, the participants gave Walford and other researchers a unique opportunity
to observe the numerous biological e ects of calorie restriction. Tel ingly, the
biochemical changes they saw in their bodies closely mirrored those Walford had
seen in his long-lived calorie-restricted mice, such as decreased body mass (15 to
20 percent), blood pressure (25 percent), blood sugar level (21 percent), and
cholesterol levels (30 percent), among others.6
In recent years, formal human studies have begun, but it has turned out to be
quite di cult to get volunteer human subjects to reduce their food intake and
maintain that level of consumption over long periods. As my col eagues Leonie
Heilbronn and Eric Ravussin wrote in The American Journal of Clinical
Nutrition in 2003, “the absence of adequate information on the e ects of good-
quality, calorie-restricted diets in nonobese humans re ects the di cultiesinvolved in conducting long-term studies in an environment so conducive to
overfeeding. Such studies in free-living persons also raise ethical and
methodologic issues. ”7 In a report published in The Journals of Gerontology in
2017, a Duke University research team described how it sought to limit 145
adults to a diet of 25 percent fewer calories than is typical y recommended for a
healthy lifestyle. People being people, the actual calorie restriction achieved was,
on average, about 12 percent over two years. Even that was enough, however, for
the scientists to see a signi cant improvement in health and a slowdown in
biological aging based on changes in blood biomarkers. 8
These days, there are many people who have embraced a lifestyle that permits
signi cantly reduced caloric intake; about a decade ago, before fasting’s most
recent revival, some of them visited my lab at Harvard.
“Isn’t it hard to do what you do?” I asked Meredith Averil and her husband,
Paul McGlothin, at the time members of CR Society International and stil very
much advocates for calorie restriction, who limit themselves to about 75 percent
of the calories typical y recommended by doctors and sometimes quite a bit less
than that. “Don’t you just feel hungry al the time?”
“Sure, at rst,” McGlothin told me. “But you get used to it. We feel great!”
At lunch that day, McGlothin expounded upon the merits of eating organic
baby food and slurped down something that looked to me like orange mush. I
also noticed that both he and Averil were wearing turtlenecks. It wasn’t winter.
And most folks in my lab are perfectly comfortable in T-shirts. But with so little
fat on their bodies, they needed the extra warmth. Then in his late 60s,
McGlothin showed no signs that his diet might slow him down. He was the
CEO of a successful marketing company and a former New York State chess
champion. He didn’t look much younger than his age, though; in large part, I
suspect this was because a lack of fat exposes wrinkles, but his blood
biochemistry suggested otherwise. On his 70th birthday, his health indicators,
from blood pressure and LDL cholesterol to resting heart rate and visual acuity,
were typical of those of a much younger person. 9 Indeed, they resembled those
seen in the long-lived rats on calorie restriction.
It’s true that what we know about the impact of lifelong calorie restriction inhumans comes down to short-term studies and anecdotal experiences. But one
of our close relatives has o ered us insights into the longitudinal bene ts of this
lifestyle.
Since the 1980s, a long-term study of calorie restriction in rhesus monkeys—
our close genetic cousins—has produced stunningly compel ing results. Before
the study, the maximum known lifespan for any rhesus monkey was 40 years.
But of twenty monkeys in the study that lived on calorie-restricted diets, six
reached that age, which is roughly equivalent to their reaching 120 in human
terms.
To hit that mark, the monkeys didn’t need to live on a calorie-restricted diet
for their entire lives. Some of the test subjects were started on a 30 percent
reduction regimen when they were middle-aged monkeys. 10
CR works to extend the lifespan of mice, even when initiated at 19 months of
age, the equivalent of a 60- to 65-year-old human, but the earlier the mice start
on CR, the greater the lifespan extension.11 What these and other animal studies
tel us is that it’s hard to “age out” of the longevity bene ts of calorie restriction,
but it’s probably better to start earlier than later, perhaps after age 40, when
things real y start to go downhil , molecularly speaking.
That doesn’t make a CR diet a good plan for everyone. Indeed, even Rozalyn
Anderson, a former trainee of mine who’s now a famous professor at the
University of Wisconsin and a lead researcher in the rhesus study, says a 30
percent calorie-reduced diet for humans, long term, amounted in her mind to a
“bonkers diet. ”12
It’s certainly not bonkers for everyone, though, especial y considering that
calorie restriction hasn’t been demonstrated only to lengthen life but also to
forestal cardiac disease, diabetes, stroke, and cancer. It’s not just a longevity
plan; it’s a vitality plan.
It’s nonetheless a hard sel for many people. It takes strong wil power to avoid
the fridge at home or snacks at work. There’s an adage in my eld: if calorie
restriction doesn’t make you live longer, it wil certainly make you feel that way.
But it turns out that’s okay, because research is increasingly demonstrating
that many of the bene ts of a life of strict and uncompromising calorie
restriction can be obtained in another way. In fact, that way might be even better.THE PERIODIC TABLE
To ensure a genetic response to a lack of food, hunger doesn’t need to be the
status quo. Once we’ve grown accustomed to stress, after al , it’s no longer as
stressful.
Intermittent fasting, or IF—eating normal portions of food but with
periodic episodes without meals—is often portrayed as a new innovation in
health. But long before my friend Valter Longo at the University of California,
Los Angeles, began touting the bene ts of IF, scientists had been studying the
e ects of periodic calorie restriction for the better part of a century.
In 1946, University of Chicago researchers Anton Carlson and Frederick
Hoelzel subjected rats to periodic food restriction and found, when they did,
that those that went hungry every third day lived 15 to 20 percent longer than
their cousins on a regular diet.13
At the time it was believed that fasting provided the body with a “rest. ”14
That’s very much the opposite of what we now know about what happens at a
cel ular level when we subject our bodies to the stress of going without food.
Either way, Carlson and Hoelzel’s work provided valuable information on the
long-term results of irregular calorie restriction.
It’s not clear whether the pair applied what they’d learned to their own lives,
but both lived relatively long lives for their time. Carlson died at the age of 81.
Hoelzel made it to 74, despite having subjected himself over the years to
experiments that included swal owing gravel, glass beads, and bal bearings to
study how long it would take for such objects to pass through his system. And
people say I’m crazy.
Today, human studies are con rming that once-in-a-while calorie restriction
can have tremendous health results, even if the times of fasting are quite
transient.
In one such study, participants ate a normal diet most of the time but turned
to a signi cantly restricted diet consisting primarily of vegetable soup, energy
bars, and supplements for ve days each month. Over the course of just threemonths, those who maintained the “fasting mimicking” diet lost weight,
reduced their body fat, and lowered their blood pressure, too. Perhaps most
important, though, the participants had lower levels of a hormone made
primarily in the liver cal ed insulin-like growth factor 1, or IGF-1. Mutations in
IGF-1 and the IGF-1 receptor gene are associated with lower rates of death and
disease and found in abundance in females whose families tend to live past
100.15
Levels of IGF-1 have been closely linked to longevity. The impact is so strong,
in fact, that in some cases it can be used to predict—with great accuracy—how
long someone wil live, according to Nir Barzilai and Yousin Suh, who research
aging at the Albert Einstein Col ege of Medicine at Yeshiva University in New
York.
Barzilai and Suh are geneticists whose research focuses on centenarians who
have made it to 100—and beyond—without su ering from any age-related
diseases. That unique population is a vital study group, because its members
provide a model for aging the way most people say they want to age—not
accepting that additional years of life need to come with additional years of
misery.
When we nd clusters of these people, we see that in some cases it doesn’t
actual y matter what they put into their bodies. They carry gene variants that
seem to put them into a state of fasting no matter what they eat. As anyone who
has ever known a centenarian can attest, it doesn’t take a lifetime of making 100
percent healthy decisions to reach 100. When Barzilai’s team studied nearly 500
Ashkenazi Jews over the age of 95, they saw that many engaged in the same sorts
of behaviors doctors have long been tel ing us to shun: eating fried foods,
smoking, and just sitting around and drinking a little too much. Barzilai once
asked one of his centenarian study subjects why she hadn’t listened to her
doctors over the years when they had strongly advised her to end her lifelong
smoking habit. “I’ve had four doctors tel me smoking would kil me,” she said
with a wry smile, “and wel , al four are dead now, aren’t they?”
Some people are simply winners in the genetic lottery. The rest of us have
some extra work to do. But the good news is that the epigenome is mal eable.
Since it’s not digital, it’s easier to impact. We can control the behavior of thisanalog element of our biology by how we live our lives.
The important thing is not just what we eat but the way we eat. As it turns
out, there is a strong correlation between fasting behavior and longevity in Blue
Zones such as Ikaria, Greece, “the island where people forget to die,” where one-
third of the population lives past the age of 90 and almost every older resident is
a staunch disciple of the Greek Orthodox church and adheres to a religious
calendar that cal s for some manner of fasting more than half the year. 16 On many days, that means no meat, dairy products, or eggs and sometimes no wine
or olive oil, either—for some Greeks, that’s just about everything. Additional y,
many Greeks observe periods of total fasting before taking Holy Communion. 17
Other longevity hot spots, such as Bama County in southern China, are
places where people have access to good, healthy food but choose to forgo it for
long periods each day. 18 Many of the centenarians in this region have spent their
lives eschewing a morning meal. They general y eat their rst smal meal of the
day around noon, then share a larger meal with their families at twilight. In this
way, they typical y spend sixteen hours or more of each day without eating.
When we investigate places like this, and as we seek to apply research about
fasting to our modern lives, we nd that there are scores of ways to calorie
restrict that are sustainable, and many take the form of what has come to be
known as periodic fasting—not being hungry al the time but using hunger
some of the time to engage our survival circuit.
Over time, some of these ways of limiting food wil prove to be more e ective
than others. A popular method is to skip breakfast and have a late lunch (the
16:8 diet). Another is to eat 75 percent fewer calories for two days a week (the
5:2 diet). If you’re a bit more adventurous, you can try skipping food a couple of
days a week (Eat Stop Eat), or as the health pundit Peter Attia does, go hungry
for an entire week every quarter. The permutations of these various models for
extending life and health are being worked out in animals and wil be worked
out in people, too. The short-term studies are promising. I suspect the long-term
research wil be, too. In the meantime, however, almost any periodic fasting diet
that does not result in malnutrition is likely to put your longevity genes to work
in ways that wil result in a longer, healthier life.
It doesn’t take any money to eat this way. In fact, it saves money. Moreover,people who are not accustomed to being able to gorge themselves whenever they
want might be in a better position to be successful at going a few days each
month with a lot less food.
At least at this juncture in the evolution of our customs around food,
though, for many people any form of fasting is a nonstarter.
I’ve tried calorie restriction. I can’t do it. Feeling hungry isn’t fun, and food is
just too pleasurable. Lately, I have taken to periodic fasting—skipping a meal or
two each day—but I admit that it’s mostly unintentional. I simply forget to eat.
So far, though, we’ve talked only about engaging the survival circuit by
limiting how much we eat, but what we eat is also important.
AMINO RIGHT
We’d die quite quickly without amino acids, the organic compounds that serve
as the building blocks for every protein in the human body. Without them—and
in particular the nine essential amino acids that our bodies cannot make on their
own—our cel s can’t assemble the life-giving enzymes needed for life.
Meat contains al nine of the essential amino acids. That’s easy energy, but it
doesn’t come without a cost. Actual y, a lot of costs. Because no matter how you
feel about the morals of the matter, meat is murder—on our bodies. So can we
just avoid protein? Ironical y, protein is what satiates us. Same for mice. Same for
swarming locusts in need of nutrients, which is why they eat each other.19 It would appear that animal life can’t easily limit protein in the diet without some
hunger pains.
There isn’t much debate on the downsides of consumption of animal
protein. Study after study has demonstrated that heavily animal-based diets are
associated with high cardiovascular mortality and cancer risk. Processed red
meats are especial y bad. Hot dogs, sausage, ham, and bacon might be gloriously
delicious, but they’re ingloriously carcinogenic, according to hundreds of
studies that have demonstrated a link between these foods and colorectal,
pancreatic, and prostate cancer.20 Red meat also contains carnitine, which gut
bacteria convert to trimethylamine N-oxide, or TMAO, a chemical that issuspected of causing heart disease.
That doesn’t mean a little red meat wil kil you—the diet of hunter-gatherers
is a mix of plants packed with ber and nutrients, mixed with some red meat and
sh in moderation21—but if you’re interested in a long and healthy life, your
diet probably needs to look a lot more like a rabbit’s lunch than a lion’s dinner.
When we substitute animal protein with more plant protein, studies have
shown, al -cause mortality fal s signi cantly.22
From an energy perspective, the good news is that there isn’t a single amino
acid that can’t be obtained by consuming plant-based protein sources. The bad
news is that, unlike most meats, weight for weight, any given plant usual y
delivers limited amounts of amino acids.
From a vitality perspective, though, that’s great news. Because a body that is
in short supply of amino acids overal , or any single amino acid for a spel , is a
body under the very sort of stress that engages our survival circuits.
You’l recal that when the enzyme known as mTOR is inhibited, it forces
cel s to spend less energy dividing and more energy in the process of autophagy,
which recycles damaged and misfolded proteins. That act of hunkering down
ends up being good for prolonged vitality in every organism we’ve studied. What
we’re coming to learn is that mTOR isn’t impacted only by caloric restriction.23
If you want to keep mTOR from being activated too much or too often,
limiting your intake of amino acids is a good way to start, so inhibiting this
particular longevity gene is real y as simple as limiting your intake of meat and
dairy.
It’s also increasingly clear that al essential amino acids aren’t equal. Rafael de
Cabo at the National Institutes of Health, Richard Mil er at the University of
Michigan, and Jay Mitchel at Harvard Medical School have found over the years
that feeding mice a diet with low levels of the amino acid methionine works
particularly wel to turn on their bodily defenses, to protect organs from hypoxia
during surgery, and to increase healthy lifespan by 20 percent.24 One of my
former students, Dudley Lamming, who now runs a lab at the University of
Wisconsin, demonstrated that methionine restriction causes obese mice to shed
most of their fat—and fast. Even as the mice, which Lamming cal ed “couch
potatoes,” continued to eat as much as they wanted and shun exercise, they stillost about 70 percent of their fat in a month, while also lowering their blood
glucose levels.25
We can’t live without methionine. But we can do a better job of restricting
the amount of it we put into our bodies. There’s a lot of methionine in beef,
lamb, poultry, pork, and eggs, whereas plant proteins, in general, tend to contain
low levels of that amino acid—enough to keep the light on, as it were, but not
enough to let biological complacency set in.
The same is true for arginine and the three branched-chain amino acids,
leucine, isoleucine, and valine, al of which can activate mTOR. Low levels of
these amino acids correlate with increased lifespan26 and in human studies, a
decreased consumption of branched-chain amino acids has been shown to
improve markers of metabolic health signi cantly. 27
We can’t live without them, but most of us can de nitely stand to get less of
them, and we can do that by lowering our consumption of foods that many
people consider to be the “good animal proteins,” chicken, sh, and eggs—
particularly when those foods aren’t being used to recover from physical stress or
injury.
Al of this might seem counterintuitive; amino acids, after al , are often
considered helpful. And they can be. Leucine, for instance, is wel known to
boost muscle, which is why it’s found in large quantities in the protein drinks
that bodybuilders often chug before, during, and after workouts. But that
muscle building is coming in part because leucine is activating mTOR, which
essential y cal s out to your body, “Times are good right now, let’s disengage the
survival circuit.” 28 In the long run, however, protein drinks may be preventing
the mTOR pathway from providing its longevity bene ts. Studies in which
leucine is completely eliminated from a mouse’s diet have demonstrated that just
one week without this particular amino acid signi cantly reduces blood glucose
levels, a key marker of improved health. 29 So a little leucine is necessary, of course, but a little goes a long way.
Al of these ndings may explain why vegetarians su er signi cantly lower
rates of cardiovascular disease and cancer than meat eaters.30 The reduction of
amino acids—and thus the inhibition of mTOR—isn’t the only thing at play in
that equation. The lower calorie content, increased polyphenols, and feeling ofsuperiority over your fel ow human beings are also helpful. Al of these, except
the last, are valid explanations for why vegetarians live longer and stay healthier.
Even if we eat a low-protein, vegetable-rich diet, we may live longer, but we
won’t maximize our lifespans—because putting our bodies into nutritional
adversity isn’t going to maximal y trigger our longevity genes. We need to induce
some physical adversity, too. If that doesn’t happen, we miss a key opportunity
to trigger our survival circuits further. Like a beautiful sports car driven only a
block and back on Sunday mornings, our longevity genes wil go tragical y
underutilized.
With so much horsepower under the hood, we just have to re up the engine
and take it out for a spin.
DO SWEAT IT
There’s a reason why for centuries exercise has been the go-to prescription for
vitality. But that reason isn’t what most people—or even many doctors—think.
In the nearly four hundred years since the English physician Wil iam Harvey
discovered that blood ows around the body in an intricate network of tubes,
doctors thought that exercise improves health by moving blood through the
circulatory system faster, ushing out the buildup of plaque.
That’s not how it works.
Yes, exercise improves blood ow. Yes, it improves lung and heart health. Yes,
it gives us bigger, stronger muscles. But more than any of that—and indeed,
what is responsible for much of that—is a simple thing that happens at a much
smal er scale: the cel ular scale.
When researchers studied the telomeres in the blood cel s of thousands of
adults with al sorts of di erent exercise habits, they saw a striking correlation:
those who exercised more had longer telomeres. And according to one study
funded by the Centers for Disease Control and Prevention and published in
2017, individuals who exercise more—the equivalent of at least a half hour of
jogging ve days a week—have telomeres that appear to be nearly a decade
younger than those who live a more sedentary life. 31 But why would exercising delay the erosion of telomeres?If you think about how our longevity genes work—employing those ancient
survival circuits—this al makes sense. Limiting food intake and reducing the
heavy load of amino acids in most diets aren’t the only ways to activate longevity
genes that order our cel s to shift into survival mode. Exercise, by de nition, is
the application of stress to our bodies. It raises NAD levels, which in turn
activates the survival network, which turns up energy production and forces
muscles to grow extra oxygen-carrying capil aries. The longevity regulators
AMPK, mTOR, and sirtuins are al modulated in the right direction by exercise,
irrespective of caloric intake, building new blood vessels, improving heart and
lung health, making people stronger, and, yes, extending telomeres. SIRT1 and
SIRT6, for example, help extend telomeres, then package them up so they are
protected from degradation. Because it’s not the absence of food or any
particular nutrient that puts these genes into action; instead it is the hormesis
program governed by the survival circuit, the mild kind of adversity that wakes
up and mobilizes cel ular defenses without causing too much havoc.
There’s real y no way around this. We al need to be pushing ourselves,
especial y as we get older, yet only 10 percent of people over the age of 65 do.32
The good news is that we don’t have to exercise for hours on end. One recent
study found that those who ran four to ve miles a week—for most people,
that’s an amount of exercise that can be done in less than 15 minutes per day—
reduce their chance of death from a heart attack by 40 percent and al -cause
mortality by 45 percent.33 That’s a massive e ect.
In another study, researchers reviewed the medical records of more than
55,000 people and cross-referenced those documents with death certi cates
issued over fteen years. 34 Among 3,500 deaths, they weren’t particularly
surprised to see that those who had told their doctors they were runners were far
less likely to die of heart disease. Even when the researchers adjusted for obesity
and smoking, the runners were less likely to have died during the years of the
study. The big shock was that the health bene ts were remarkably similar no
matter how much running the people had done. Even about ten minutes of
moderate exercise a day added years to their lives. 35
There is a di erence between a leisurely walk and a brisk run, however. Toengage our longevity genes ful y, intensity does matter. Mayo Clinic researchers
studying the e ects of di erent types of exercise on di erent age groups found
that although many forms of exercise have positive health e ects, it’s high-
intensity interval training (HIIT)—the sort that signi cantly raises your heart
and respiration rates—that engages the greatest number of health-promoting
genes, and more of them in older exercisers. 36
You’l know you are doing vigorous activity when it feels chal enging. Your
breathing should be deep and rapid at 70 to 85 percent of your maximum heart
rate. You should sweat and be unable to say more than a few words without
pausing for breath. This is the hypoxic response, and it’s great for inducing just
enough stress to activate your body’s defenses against aging without doing
permanent harm.37
We’re stil working to understand what al of the longevity genes do, but one
thing is already clear: many of the longevity genes that are turned on by exercise
are responsible for the health bene ts of exercise, such as extending telomeres,
growing new microvessels that deliver oxygen to cel s, and boosting the activity
of mitochondria, which burn oxygen to make chemical energy. We’ve known for
a long time that these bodily activities fal as we age. What we also know now is
that the genes most impacted by exercise-induced stress can bring them back to
the levels associated with youth. In other words: exercise turns on the genes to
make us young again at a cel ular level.
Often I’m asked, “Can I just eat what I want and run o the extra calories?”
My answer is “Unlikely.” When you give rats a high-calorie diet and al ow them
to burn o the energy, lifespan extension is minimal. Same for a CR diet. If you
make food l ing but not as calori c, some of the health bene ts are lost. Being
hungry is necessary for CR to work because hunger helps turn on genes in the
brain that release longevity hormones, at least according to a recent study by
Dongsheng Cai at the Albert Einstein Col ege of Medicine. 38
Would a combination of fasting and exercise lengthen your lifespan?
Absolutely. If you manage to do both these things: congratulations, you are wel
on your way.
But there is plenty more you can do.
THE COLD FRONTBefore arriving in Boston in my early 20s, I’d spent my whole life in Australia.
Cultural y, everything worked out just ne. Within a week, I’d gured out which
markets carried Vegemite, the black yeast spread that some might say requires
some pretty signi cant epigenetic programming as a child to enjoy as an adult. It
took a bit longer to track down the best places for meat pies, Violet Crumble,
Tim Tams, and musk sticks, but eventual y I gured out how to get al of those
tastes of home, too. And it didn’t take long before I stopped caring that folks in
the United States seem to have a hard time di erentiating between Australian
and British accents. (It’s not that hard; Aussie accents are sexier.)
The toughest part was the cold.
As a boy, I thought I knew what cold was. When the temperature at
Observatory Hil , Sydney’s o cial weather station for more than a century,
approached freezing (it hasn’t actual y fal en below freezing in modern history),
that was cold.
Boston was a whole di erent world. A real y frigid one.
I invested in coats, sweaters, and long underwear and spent a lot of time
indoors. Like a lot of postdoctoral fel ows, I often worked through the night. I
truly was committed to my work, but the truth is that part of the calculus for
not going home, on many nights, was that I didn’t want to go outside.
These days I wish I’d taken a di erent approach. I wish I’d just told myself to
tough it out. To take a walk in the bitter cold. To dip my toes into the Charles
River in the middle of January. Because as it turns out, exposing your body to
less-than-comfortable temperatures is another very e ective way to turn on your
longevity genes.
When the world takes us out of the thermoneutral zone—the smal range of
temperatures that don’t require our bodies to do any extra work to stay warm or
cool o —al sorts of things happen. Our breathing patterns shift. The blood
ow to and through our skin—the largest organ in our body—changes. Our
heart rates speed up or slow down. These reactions aren’t happening just
“because.” Al of these reactions have genetic roots dating back to M. superstes’s
ght for survival al those bil ions of years ago.
Homeostasis, the tendency for living things to seek a stable equilibrium, is auniversal biological principle. Indeed, it is the guiding force of the survival
circuit. And thus we see it everywhere we look—especial y on the low end of the
thermometer.
As scientists have increasingly turned their attention to the impacts of
reduced food intake on the human body, it has quickly become clear that calorie
restriction has the e ect of reducing core body temperature. It wasn’t at rst
clear whether this contributed to prolonged vitality or was simply a by-product
of al of the changes happening in the bodies of organisms exposed to this
particular sort of stress.
Back in 2006, though, a team from the Scripps Research Institute genetical y
engineered some lab mice to live their lives a half degree cooler than normal—a
feat they accomplished by playing a trick on the mice’s biological thermostat.
The team inserted copies of the mouse UCP2 gene into the mice’s
hypothalamus, which regulates the skin, sweat glands, and blood vessels. UCP2
short-circuited mitochondria in the hypothalamus so they produced less energy
but more heat. That, in turn, caused the mice to cool down about half a degree
Celsius. The result was a 20 percent longer life for female mice, the equivalent of
about seven additional healthy human years, while male mice got an extension of
12 percent. 39

COLD ACTIVATES LONGEVITY GENES. Sirtuins are switched on by cold, which in turn activates
protective brown fat in our back and shoulders. Image: The author enduring “cold therapy” at
the Massachusetts Institute of Technology in 1999.
The gene involved—which has a human analog—wasn’t just a piece of the
complex machinery that tricked the hypothalamus into thinking the mice’s
bodies were warmer than they were. It was also a gene that has been connected
time and time again to longevity. Five years earlier, a joint team of researchers
from Beth Israel Deaconess Medical Center and Harvard Medical School
showed that mice age faster when their UCP2 gene is nul i ed. 40 And in 2005,
Stephen Helfand and his team, then at the University of Connecticut Health
Center, had demonstrated that targeted upregulation of an analogous gene
could extend the lifespans of fruit ies by 28 percent in females and 11 percent
in males.41 Then, in 2017, the connection between the UCP2 gene and aging
came ful circle, thanks to researchers from Université Laval in Quebec: not only
could UCP2 make mice “run cold,” the Canadian team demonstrated, but
colder temperatures could change the way the gene operated, too—through its
ability to rev up brown adipose tissue.42
Also known as “brown fat,” this mitochondria-rich substance was, untilrecently, thought to exist only in infants. Now we know that it is found in
adults, too, although the amount of it decreases as we age. Over time, it becomes
harder and harder to nd; it mingles with white fat and is spread out even more
unevenly across the body. It “hangs out” in di erent areas in di erent people,
sometimes in the abdomen, sometimes across the upper back. That makes
researching it in humans a bit of a chal enge: it general y takes a PET scan—
which requires the injection of radioactive glucose—to locate it. Rodent studies,
however, have provided signi cant insights into the correlation between brown
fat and longevity.
One study of genetical y engineered Ames dwarf mice, for instance,
demonstrated that the function of brown fat is enhanced in these remarkably
long-lived animals. 43 Other studies have shown that animals with abundant
brown fat or subjected to shivering cold for three hours a day have much more of
the mitochondrial, UCP-boosting sirtuin, SIRT3, and experience signi cantly
reduced rates of diabetes, obesity, and Alzheimer’s disease. 44
That is why we need to learn more about how to chemical y substitute for
brown adipose tissue thermogenesis.45 Chemicals cal ed mitochondrial
uncouplers can mimic the e ects of UCP2, al owing protons to leak through
mitochondrial membranes, like dril ing holes in a dam at a hydroelectric plant.
The result is not cold but heat as a by-product of the mitochondrial short
circuit.
The sweet-smel ing mitochondrial uncoupler cal ed 2,4-dinitrophenol
(DNP) was used for making explosives in the First World War, and it soon
became apparent that employees exposed to the chemical were rapidly losing
weight, with one employee even dying from overexposure.46 In 1933, doctors
Windsor Cutting and Maurice Tainter, from the Stanford University School of
Medicine, summarized a series of their papers showing that DNP markedly
increases metabolic rate. 47 That same year, despite Tainter and Cutting’s
warnings about “certain potential dangers,” twenty companies started sel ing it
in the United States, as did others in Great Britain, France, Sweden, Italy, and
Australia.
It worked wel —too wel , in fact.
Just one year later, speaking before the American Public Health Association,Tainter said, “The interest in and enthusiasm for this product were so great that
its wide-spread use has become a matter of some concern in public health. The
total amount of the drug being used is astonishing.”
Moments later he dropped a bombshel : “during the past year, the Stanford
Clinics have supplied . . . over 1,200,000 capsules of dinitrophenol of 0.1 gm.
each.” 48
Over 1 mil ion capsules? From one university? In one year? That is
astonishing. And that was in 1933, when California had an eighth of its present
population. Three pounds of weight per person per week were reportedly being
shed. The public was relieved—something finally worked. Obesity was going to
be a thing of the past.
But the metabolic party didn’t last long. People began to die from overdoses,
and other long-term side e ects showed up. DNP was declared “extremely
dangerous and not t for human consumption” in the United States Federal
Food, Drug, and Cosmetic Act of 1938. As a curious aside, the legislation was
written by Senator Royal Copeland, a homeopathic physician who, only days
before he died, entrenched protections for natural supplements that today fuel a
largely unregulated industry with revenues of $122 bil ion.
The act rightly banned a dangerous substance but dashed hopes that obesity
would be a thing of the past.49 Anecdotal y, DNP continued to be prescribed to
Russian soldiers during World War II to keep them warm, 50 and today some
unscrupulous people sel it on the internet. But they do so at their peril. In 2018,
Bernard Rebelo was sentenced to seven years in prison for the death of a woman
to whom he sold DNP. In the United States, there have been sixty-two
documented deaths since 1918, though there were likely many more than that.51
One thing is clear: DNP is extremely dangerous. Eating less at each meal,
moving more, and focusing on plant-based foods are much safer options.
Another thing you can try is activating the mitochondria in your brown fat
by being a bit cold. The best way to do this might be the simplest—a brisk walk
in a T-shirt on a winter day in a city such as Boston wil do the trick. Exercising
in the cold, in particular, appears to turbocharge the creation of brown adipose
tissue. 52 Leaving a window open overnight or not using a heavy blanket while you sleep could help, too.This hasn’t gone unnoticed by the health and wel ness industry. Being cold is
hot right now. Cryotherapy—a few minutes in a box superchil ed to −110°C or
−166°F—is an increasingly popular method of inducing a helping of this sort of
stress to our bodies, although the research is stil a ways away from being
conclusive as to how, why, and even whether it truly works. 53 That didn’t stop
me from accepting an invitation from Joe Rogan, the media mogul and
comedian, to go with him to a cryotherapy spa. Three minutes standing in my
underwear at Mars temperatures may have activated my brown fat and al the
great health bene ts that go with that. At the very least, it left me invigorated
and grateful to be alive.
As with most things in life, it’s probably best to change your lifestyle when
you are young, because making brown fat becomes harder as you get older. If
you choose to expose yourself to the cold, moderation wil be key. Similar to
fasting, the greatest bene ts are likely to come for those who get close to, but not
beyond, the edge. Hypothermia is not good for our health. Neither is frostbite.
But goose bumps, chattering teeth, and shivering arms aren’t dangerous
conditions—they’re simply signs that you’re not in Sydney. And when we
experience these conditions often enough, our longevity genes get the stress they
need to order up some additional healthy fat.
What happens on the other side of the thermostat? The picture is a bit less
clear, but we have some promising leads from our friend S. cerevisiae. We know
from work in my lab that raising the temperature of yeast—from 30°C to 37°C,
just below the limits of what those single-cel ed organisms can sustain—turns on
the PNC1 gene and boosts their NAD production, so their Sir2 proteins can
work that much harder. What’s fascinating is not so much that these
temperature-stressed cel s lived 30 percent longer but that the mechanism was
the same as that evoked by calorie restriction.
Is heat good for human bodies, too? Possibly, but not exactly in the same way.
Because we are warm-blooded animals, our enzymes haven’t evolved a tolerance
for large changes in temperature. You can’t just raise your core body temperature
and expect to live longer. But as my northern German wife, Sandra, likes to
point out, there are a lot of bene ts to exposing your skin and lungs to high temperatures, at least temporarily.Continuing an ancient Roman tradition, many northern and eastern
Europeans regularly partake in “sauna bathing” for relaxation and health
reasons. The Finns are the most dedicated, with the majority of men reporting
using a sauna once a week, year round. Sandra tel s me it’s pronounced “ZOW-
na” not “saw-nah,” and that no home should be without one. I’m sticking with
saw-nah to avoid sounding like a snicklefritz, but when it comes to housing
construction, Sandra may be on to something.
A 2018 study conducted in Helsinki found that “physical function, vitality,
social functioning, and general health were signi cantly better among sauna
users than non-users,” although the researchers were correct to point out that
part of the e ect could be due to the fact that those who are sick or disabled
don’t go to the sauna. 54
A more convincing study fol owed a group of more than 2,300 middle-aged
men from eastern Finland for more than twenty years. 55 Those who used a
sauna with great frequency—up to seven times a week—enjoyed a twofold drop
in heart disease, fatal hearts attacks, and al -cause mortality events over those
who heat bathed once per week.
None of the sauna studies dug deep enough to tel us why temporary heat
exposure may be so good for us. If yeast is any guide, NAMPT, the gene in our
bodies that recycles NAD, may be in on the act. NAMPT is turned on by a
variety of adversity triggers, including fasting and exercise, which makes more
NAD so the sirtuins can work hard at making us healthier.56 We have never
tested if NAMPT is turned on by heat, but that would be something to do.
Either way, one thing is clear: it does us little good to spend our entire lives in the
thermoneutral zone. Our genes didn’t evolve for a life of pampered comfort. A
little stress to induce hormesis once in a while likely goes a long way.
But dealing with biological adversity is one thing. Overwhelming genetic
damage is another.
DON’T ROCK THE LANDSCAPE
A bit of adversity or cel ular stress is good for our epigenome because itstimulates our longevity genes. It activates AMPK, turns down mTOR, boosts
NAD levels, and activates the sirtuins—the disaster response teams—to keep up
with the normal wear and tear that comes from living on planet Earth.
But “normal” is the operative word, because when it comes to aging,
“normal” is bad enough. When our sirtuins have to respond to many disasters—
especial y those that cause double-strand DNA breaks—these epigenetic
signalers are forced to leave their posts and head to other places on the genome
where DNA breaks have occurred. Sometimes they make their way back home.
Sometimes they don’t.
We can’t prevent al DNA damage—and we wouldn’t want to because it’s
essential for the function of the immune system and even for consolidating our
memories57—but we do want to prevent extra damage.
And there’s a lot of extra damage to be had out there.
Cigarettes, for starters. There aren’t many legal vices out there that are worse
for your epigenome than the deadly concoction of thousands of chemicals
smokers put into their bodies every day. There’s a reason why smokers seem to
age faster: they do age faster. The DNA damage that results from smoking keeps
the DNA repair crews working overtime, and likely the result is the epigenetic
instability that causes aging. And although I’m not likely to be the rst person
you’l hear this from, it nonetheless bears repeating: smoking is not a private,
victimless activity. The levels of DNA-damaging aromatic amines in cigarette
smoke are about fty to sixty times as high in secondhand as in rsthand
smoke. 58 If you do smoke, it is worth trying to quit.
Don’t smoke? That’s great, but even without smoke there’s re. In much of
the developed world—and increasingly in the developing world as wel —we’re
practical y bathing in DNA-damaging chemicals. In some places—cities with
lots of people and lots of cars, especial y—the simple act of breathing is enough
to do extra damage to your DNA. But it would also be wise to be wary of the
PCBs and other chemicals found in plastics, including many plastic bottles and
take-out containers. 59 (Avoid microwaving these; it releases even more PCBs.)
Exposure to azo dyes, such as aniline yel ow, which is used in everything from
reworks to the yel ow ink in home printers, can also damage our DNA.60 And
organohalides—compounds that contain substituted halogen atoms and areused in solvents, degreasers, pesticides, and hydraulic uid—can also wreak
havoc on our genomes.
Nobody in his right mind would purposeful y ingest solvents, degreasers,
pesticides, and hydraulic uid, of course, but there’s plenty of damage to be had
in some of the things we do intentional y eat and drink. We’ve known for more
than half a century that N-nitroso compounds are present in food treated with
sodium nitrite, including some beers, most cured meats, and especial y cooked
bacon. In the decades since, we’ve learned that these compounds are potent
carcinogens. 61 What we’ve also come to understand is that cancer is just the start
of our nitrate-treated woes, because nitroso compounds can in ict DNA
breakage as wel 62—sending those overworked sirtuins back to work some more.
Then there’s radiation. Any source of natural or human-in icted radiation,
such as UV light, X-rays, gamma rays, and radon in homes (which is the second
most frequent cause of lung cancer besides smoking63) can cause additional
DNA damage, necessitating the cal -up of an epigenetic x-it team. As someone
who ies a lot for work, I think about this quite a bit—every time I go through
security, in fact. Most of the research on the current versions of airport scanners
suggests that they probably don’t do tremendous damage to our DNA, but
there’s been little attention given to their long-term impact on our epigenome
and the aging process. No one has ever tested what a mouse looks like two years
after being repeatedly exposed to these devices. The ICE mice tel us that
chromosome tickling is al that’s needed to accelerate aging. I’m aware the
radiation exposure from mil imeter-wave scanners is lower than that from
previous scanners. The security attendants at the machine tel travelers the
exposure is about the “same as the ight.” But with mil ions of ight miles under
my belt, why would I want to double the damage? Whenever possible, I take the
pre-check line or ask for a pat down instead.
If al of this makes you feel as if it’s impossible to completely avoid DNA
breaks and the epigenetic consequences of those breaks, wel , that’s true. The
natural and necessary act of replicating DNA causes DNA breaks, tril ions of
them throughout your body every day. You can’t avoid radon particles or cosmic
rays unless you live in a lead box at the bottom of the ocean. And even if you
were to move to a desert island, the sh you’d have to eat would likely containmercury, PCBs, PBDEs, dioxins, and chlorinated pesticides, al of which can
damage your DNA. 64 In our modern world, even with the most “natural”
lifestyle you can fol ow, this sort of DNA damage is inevitable.
No matter how old you are, even if you are a teenager, it is already happening
to you. 65 DNA damage has accelerated your clock, with implications at al stages
of life. Embryos and babies experience aging. What, then, of people in their 60s,
70s, and 80s? What of those individuals who are already frail and cannot restrict
their calories, go for a run, or make snow angels in the dead of winter? Is it too
late for them?
Not at al .
But if we’re all going to live longer and healthier lives—regardless of how
much epigenetic drift and aging we have experienced at this moment in time—
we might need some additional help.
FIVEA BETTER PILL TO SWALLOW
THE DREAM OF EXTENDING HUMAN lives did not begin in the early twenty- rst
century any more than the dream of human ight began in the early twentieth.
Nothing begins with science; it al begins with stories.
From Gilgamesh the Sumerian king, who is said to have reigned over Uruk
for 126 years, to Methuselah the patriarch in Hebrew scriptures, who is said to
have lived to the age of 969, humanity’s sacred stories testify to our deep-seated
fascination with longevity. Outside myths and parables, though, we had little
scienti c evidence of anyone succeeding in extending their life far beyond the
single century mark.
We had little hope of doing so without a deep understanding of how life
works. That is knowledge, albeit stil imperfect, that some of my col eagues and I
believe we nal y possess.
It wasn’t until 1665 that “England’s Leonardo,” Robert Hooke, published
Micrographia, in which he reported seeing cel s in cork bark. That discovery
launched us into the modern era of biology. But centuries would pass before we
had any clue about how cel s work at the molecular scale. That knowledge could
come only from the combination of a series of great leaps in microscopy,
chemistry, physics, genetics, nanoengineering, and computing power.
To understand how aging occurs, we must journey down into the subcel ular
nanoworld, heading down to the cel , piercing the outer membrane, and
traveling into the nucleus. From there, we head down to the scale of amino acidsand DNA. At this size, it is obvious why we don’t live forever.
Until we understood life at the nanoscale, even why we live was a mystery.
The bril iant Austrian theoretical physicist Erwin Schrödinger, the man who
developed quantum physics (and yes, that famous thought experiment involving
a both-dead-and-alive cat) was ummoxed when he tried to explain life. In 1944,
he threw up his hands and declared that living matter “is likely to involve ‘other
laws of physics’ hitherto unknown.” 1 That was the best he could do at the time.
But things moved quickly in the decades to come. And today, the answer to
Schrödinger’s 1944 book, What Is Life? , if not ful y answered, is certainly close
to being so.
Turns out, there is no new law required to explain life. At the nanoscale, it is
merely an ordered set of chemical reactions, concentrating and assembling atoms
that would normal y never assemble, or breaking apart molecules that would
normal y never disintegrate. Life does this using proteinaceous Pac-Men cal ed
enzymes made up of coils and layered mats of amino acid chains.
Enzymes make life possible by taking advantage of fortuitous molecular
movements. Every second you are alive, thousands of glucose molecules are
captured within each of your tril ions of cel s by an enzyme cal ed glucokinase,
which fuses glucose molecules to phosphorus atoms, tagging them for energy
production. Most of the energy created is used by a multicomponent RNA and
protein complex cal ed a ribosome, whose primary job is to capture amino acids
and fuse them with other amino acids to make fresh proteins.
Does this sort of talk make your eyes gloss over? You are not alone, and you
are not to blame. We teachers have done society a great disservice by making cool
science boring. Textbooks and scienti c papers depict biology as a static, two-
dimensional world. Chemicals are drawn as sticks, biochemical pathways are
arrows, DNA is a line, a gene is a rectangle, and enzymes are ovals, drawn
thousands of times larger relative to the cel than they actual y are.
But once you understand how cel s actual y work, they are the most amazing
things. The problem with conveying this wonder in a classroom is that cel s exist
in four dimensions and buzz around with speeds and on scales we humans
cannot perceive or even conceive. To us, the second and the mil imeter are short
divisions of time and space, but to an enzyme about 10 nanometers across andvibrating every quadril ionth of a second, a mil imeter is the size of a continent
and a second is more than a year.2
Consider catalase, a ubiquitous, regular-sized enzyme that can break apart
and detoxify 10,000 molecules of hydrogen peroxide per second. A mil ion of
them could t inside an E. coli bacterium, a mil ion of which could t on the
head of a pin. 3 These numbers aren’t just hard to imagine; they are
inconceivable.
In each cel are a total of 75,000 enzymes like catalase,4 al thrown together,
jostling around in a slightly salty sea. At the nanoscale, water is gelatinous, and
molecular events are more violent than a category 5 hurricane, with molecules
thrown together at speeds we would perceive as a thousand miles per hour.
Enzymatic reactions are one-in-a-thousand events, but at the nanoscale one-in-a-
thousand events can occur thousands of times a second, enough to sustain life.
If this sounds chaotic, it is, but we need this chaos for order to emerge.
Without it, the molecules that must come together to sustain life would not nd
each other, and they would not fuse. The human sirtuin enzyme cal ed SIRT1
serves as a good example. Precise vibrating sockets on SIRT1 simultaneously
clasp onto an NAD molecule and the protein it wants to strip the acetyls from,
such as a histone or FOXO3. The two captured molecules immediately lock
together, just before SIRT1 rips them apart in a di erent way, producing
vitamin B3 and acetylated adenine ribose as waste products that are recycled back
to NAD.
More important is the fact that the target protein has now been stripped of
the acetyl chemical group that was holding it at bay. Now the histone can pack
DNA more tightly to silence genes, and FOXO3 has had its shackles removed,
al owing it to go turn on a defense program of protective genes.
If the chaos ended and our enzymes suddenly stopped doing what they do,
we would al be dead within a few seconds. Without energy and cel defenses,
there can be no life. M. superstes would never have emerged from the scum and
its descendants would never have been capable of comprehending the words on
this page.
And so, at the fundamental level, life is rather simple: we exist by the grace ofan order created from chaos. When we toast to life, we real y should be toasting
to enzymes.
By studying life at this level, we’ve also learned something rather important—
something the Nobel Prize–winning physicist Richard Feynman expressed
succinctly: “There is nothing in biology yet found that indicates the inevitability
of death. This suggests to me that it is not at al inevitable and that it is only a
matter of time before biologists discover what it is that is causing us the
trouble.” 5
It’s true: there are no biological, chemical, or physical laws that say life must
end. Yes, aging is an increase in entropy, a loss of information leading to disorder.
But living things are not closed systems. Life can potential y last forever, as long
as it can preserve critical biological information and absorb energy from
somewhere in the universe. This doesn’t mean we could be immortal tomorrow
—no more than we could have own to the moon on December 18, 1903.
Science moves forward with smal steps and big steps, but always one step at a
time.
Here’s the remarkable thing: the rst steps have actual y been available to us
since the times of Gilgamesh and Methuselah, and indeed from the time of M.
superstes. And, in the past few centuries, and by accident even earlier than that,
we have discovered ways to chemical y modulate enzymes with molecules we cal
medicines.
Now that we know how life works and have the tools to change it at a genetic
and epigenetic level, we can build upon this very old wisdom. And when it
comes to the goal of extending healthy lifespans, the easiest measures to use are
the various drugs that we already know can impact human aging.
THE WORLD’S GREATEST EASTER EGG
Rapa Nui, a remote volcanic island 2,300 miles west of Chile, is commonly
known as Easter Island and even better known for the nearly nine hundred giant
stone heads that line the island’s perimeter. What should be just as wel known
—and perhaps one day wil be—is the story of how the island came to be thesource of the world’s most e ective lifespan-extending molecule.
Back in the mid-1960s, a team of scientists traveled to the island. The
researchers were not archaeologists seeking answers about the origins of the moai
statues but rather biologists looking for endemic microorganisms.
In the dirt beneath one of the island’s famed stone heads, they discovered a
new actinobacterium. That single-cel ed organism was Streptomyces
hygroscopicus, and when it was isolated by a pharmaceutical researcher, Suren
Sehgal, it soon became clear that the actinobacterium secreted an antifungal
compound. Sehgal named that compound rapamycin, in honor of the island
where it was discovered, and began looking for ways to process it as a potential
remedy for fungal conditions such as athlete’s foot.6 The compound looked
promising for that purpose, but when the Montreal lab where Sehgal worked
was shuttered in 1983, he was directed to destroy the compound.
He couldn’t bring himself to do that, though. Instead he spirited a few vials
of the bacterium out of the lab and kept them in his freezer at home until the
late 1980s, when he convinced his bosses at a new lab in New Jersey to let him
resume studying it.
It wasn’t long before researchers discovered that the compound was an
e ective suppressor of the immune system. That would end its potential as an
antifungal—there are plenty of remedies for athlete’s foot that don’t come at the
cost of lowered immunity—but it gave scientists a new attribute to study.
Even in the 1960s, researchers knew that one of the most common reasons
for an organ transplant to fail is that the recipient patient’s body rejects it. Could
rapamycin lower the immune response enough to ensure the organ would be
accepted? Indeed it could.
It is for this reason that if you were to make a pilgrimage to Rapa Nui, you
might come upon a smal plaque at the site where S. hygroscopicus was
discovered. “At this site,” the plaque reads in Portuguese, “soil samples were
obtained in January 1965 that al owed the production of rapamycin, a substance
that inaugurated a new era for patients who need organ transplants.”
I suspect that a larger plaque may soon be in order, because the discovery of
S. hygroscopicus set into motion a tremendous amount of research, much of
which is stil ongoing and some of which has the potential to prolong vitality forcountless other people. Because in recent years it has become clear that
rapamycin isn’t just an antifungal compound and it isn’t just an immune system
suppressor; it’s also one of the most consistently successful compounds for
extending life.
We know this from experiments on a diverse menagerie of model organisms
in labs around the world. And much as my own research began with
experiments with yeast, much of the initial work that has been done to
understand rapamycin was completed on S. cerevisiae. If you put 2,000 normal
yeast cel s into a culture, a few wil remain viable after six weeks. But if you feed
those yeast cel s rapamycin, in six weeks about half wil stil be healthy. 7 The drug
wil also increase the number of daughter cel s mothers can produce by
stimulating the production of NAD.
Fruit ies fed rapamycin live about 5 percent longer.8 And smal doses of
rapamycin given to mice when they are already in the nal months of their
normal lives results in 9 to 14 percent longer lives, depending on whether they
are male or female, which translates to about a decade of healthy human life. 9
We’ve known for a long time that greater parental age is a risk factor for
disease in the next generation. That’s the power of epigenetics. But mice treated
with rapamycin buck this trend. When researchers from the German Center for
Neurodegenerative Diseases inhibited mTOR in mice born to older fathers, the
negative impact of having an old parent went away. 10
Want to know what the world’s most prominent arbiters of great science
think about the potential of TOR and the molecules that inhibit it to change
the world? The three men who discovered TOR in yeast, Joseph Heitman,
Michael Hal , and Rao Movva, are on a lot of people’s shortlists for the Nobel
Prize in Medicine or Physiology. My col eague across the river at MIT, David
Sabatini, who identi ed mTOR, was named a Clarivate Citation Laureate for
having his work cited most frequently in top-tier peer-reviewed journals; the
Clarivate list has predicted more than forty Nobel Prize winners since 2002.11
Rapamycin isn’t a panacea. Longer-lived animals might not fare as wel on it
as shorter-lived ones do; it’s been shown to be toxic to kidneys at high doses over
extended periods of time; and it might suppress the immune system over time.
That doesn’t mean TOR inhibition is a dead end, though. It might be safe insmal or intermittent doses—that worked in mice to extend lifespan12 and in
humans dramatical y improved the immune responses of elderly people to a u
vaccine.13
There are hundreds of researchers from the TOR inhibition side of the
family working in universities and biotech companies to identify “rapalogs,”
which are compounds that act on TOR in ways similar to rapamycin but have
greater speci city and less toxicity.14
The quality of the people involved in this line of research and development
makes it hard to bet against TOR inhibition as a pathway to greater human
health and vitality. But even if rapalogs don’t pan out, there’s another
pharmaceutical pathway to prolonged vitality that has already proven to be both
e ective and relatively safe.
PENNIES FOR PROLONGED VITALITY
Galega officinalis is a lovely ower, with stacks of delicate purple petals that seem
locked in a reverent bow to the world.
Also known as goat’s rue, a rather unfortunate name, and French lilac, a far
more charming sobriquet, it has been used as an herbal medicine in Europe for
centuries, owing to a chemical composition rich in guanidine, a smal chemical
in human urine that serves as an indicator of healthy protein metabolism. In the
1920s, doctors began to prescribe guanidine as a way to lower blood glucose
levels in patients with diabetes.
In 1922, a 14-year-old boy named Leonard Thompson, who was dying in a
Toronto hospital, became the rst diabetic patient to be given an injection of a
novel pancreatic peptide hormone that had shown great promise in animal
studies. Two weeks later he was given another, and news of his exceptional
improvement spread quickly around the world. Type 1 diabetes, which occurs
when the pancreas doesn’t produce enough of the hormones needed to alert the
body to sugar, is now widely treated by supplemental insulin. But the ght was
not over.
The type 2 version of the disease, so-cal ed age-associated diabetes, occurswhen the pancreas is able to make enough insulin but the body is deaf to it. The
9 percent of al adults global y with this disease need a drug that restores their
body’s sensitivity to insulin so cel s take up and use the sugar that’s coursing
through their bloodstreams. That’s important for at least two reasons: it gives
the overworked pancreas a rest, and it prevents spikes of freely oating sugar
from essential y caramelizing proteins in the body. Recent results indicate high
blood sugar can also speed up the epigenetic clock.
Thanks to an increasingly sedentary lifestyle and the abundance of sugars and
carbohydrates on every supermarket shelf around the globe, high blood sugar is
causing the premature deaths of 3.8 mil ion people a year. These deaths do not
come quickly and compassionately but in horri c ways, with blindness, kidney
failure, stroke, open foot wounds, and limb amputations.
As they considered this disease in the mid-1950s, the pharmacist Jan Aron
and the physician Jean Sterne—both Frenchmen who would have been
exceptional y familiar with the purple- owering plant so ubiquitous in their
native land—decided to reinvestigate the potential of French lilac derivatives to
ght type 2 diabetes in ways insulin doesn’t. 15
In 1957, Sterne published a paper demonstrating the e ectiveness of oral
dimethyl biguanide to treat type 2 diabetes. The drug, now most commonly
cal ed metformin, has since become one of the most widely taken and e ective
medicines on the globe. It’s among the medications on the World Health
Organization’s Model List of Essential Medicines, a catalog of the most e ective,
safe, and cost-e ective therapies for the world’s most prevalent medical
conditions. As a generic medication, it costs patients less than $5 a month in
most of the world. Except for an extremely rare condition cal ed lactic acidosis,
the most common of the side e ects is some stomach discomfort. Many people
mitigate that side e ect by taking the medication as a coated tablet or with a glass
of milk or a meal, but even when that doesn’t work, the mild upset feeling comes
with a bit of a side bene t: it tends to discourage overeating.
What place does a diabetes medication have in a conversation about
prolonging vitality? Perhaps it would have no place at al if not for the fact that, a
few years ago, researchers noticed a curious phenomenon: people taking
metformin were living notably healthier lives—independent, it seemed, of itse ect on diabetes. 16
In mice, even a very low dose of metformin has been shown by Rafael de
Cabo’s lab at the National Institutes of Health to increase lifespan by nearly 6
percent, though some have argued that the e ect is due mostly to weight loss.17
Either way, that amounts to the equivalent of ve extra healthy years for
humans, with an emphasis on healthy—the mice showed reduced LDL
cholesterol levels and improved physical performance.18 As the years have gone
by, the evidence has mounted. In twenty-six studies of rodents treated with
metformin, twenty- ve showed protection from cancer. 19
Like rapamycin, metformin mimics aspects of calorie restriction. But instead
of inhibiting TOR, it limits the metabolic reactions in mitochondria, slowing
down the process by which our cel ular powerhouses convert macronutrients
into energy.20 The result is the activation of AMPK, an enzyme known for its
ability to respond to low energy levels and restore the function of mitochondria.
It also activates SIRT1, one of my lab’s favorite proteins. Among other bene cial
e ects, metformin inhibits cancer cel metabolism, increases mitochondrial
activity, and removes misfolded proteins.21
A study of more than 41,000 metformin users between the ages of 68 and 81
concluded that metformin reduced the likelihood of dementia, cardiovascular
disease, cancer, frailty, and depression, and not by a smal amount. In one group
of already frail subjects, metformin use over the course of nine years reduced
dementia by 4 percent, depression by 16 percent, cardiovascular disease by 19
percent, frailty by 24 percent, and cancer by 4 percent. 22 In other studies, the
protective power of metformin against cancer has been far greater than that.
Though not al cancers are suppressed—prostate, bladder, renal, and esophageal
cancer seem recalcitrant—more than twenty- ve studies have shown a powerful
protective e ect, sometimes as great as a 40 percent lower risk, most notably for
lung, colorectal, pancreatic, and breast cancer.23
These aren’t just numbers. These are people whose lives were markedly
improved by using a single, safe drug that costs less than a cup of bad co ee.
If al metformin could do was reduce cancer incidence, it would stil be worth
prescribing widely. In the United States, the lifetime risk of being diagnosed
with cancer is greater than 40 percent. 24 But there’s a dividend beyond preventing cancer directly, a side e ect of living longer that most people don’tconsider: after age 90, your chances of dying of cancer drop considerably. 25 Of
course, people wil stil die of other conditions, but the tremendous pain and
costs associated with cancer would be signi cantly mitigated.
The beauty of metformin is that it impacts many diseases. Through the
power of AMPK activation, it makes more NAD and turns on sirtuins and
other defenses against aging as a whole—engaging the survival circuit upstream
of these conditions, ostensibly slowing the loss of epigenetic information and
keeping metabolism in check, so al organs stay younger and healthier.
Most of us assume that the e ects of a pil like metformin would take years to
produce any appreciable e ect on aging, but maybe not. An admittedly smal
study of healthy volunteers claimed that the DNA methylation age of blood cel s
is reversed within a week and, astoundingly, only ten hours after taking a single
850 mg pil of metformin. 26 But clearly more work is needed with greater
numbers of subjects to know for sure if metformin can delay the aging clock
over the long run.
In most countries, metformin isn’t yet prescriptible as an antiaging drug, but
for the hundreds of mil ions of people around the world who are diabetic, it’s
not a hard prescription to get. In some places, such as Thailand, metformin is
even available over the counter at every pharmacy—for just a few cents a pil . In
the rest of the world, even if you have prediabetes, it can be chal enging to
convince a doctor to prescribe you metformin. If you’ve been good to your
body, and greater than 93.5 percent of your blood’s hemoglobin isn’t irreversibly
bound to glucose—meaning it’s mostly the HbA1 type not HbA1c—you’re out
of luck, not just because the majority of physicians don’t know the data I just
shared with you, but because even if they did, aging isn’t yet considered a disease.
Among the people taking metformin—and leading the charge to evaluate its
long-term e ects on aging in humans—is Nir Barzilai, the Israeli American
physician and geneticist who, along with his col eagues at Albert Einstein
Col ege of Medicine, discovered several longevity gene variants in the insulin-like
growth hormone receptor that controls FOXO3, the cholesterol gene CETP,
and the sirtuin SIRT6, al of which seem to help ensure that some lucky peoplewith Ashkenazi Jewish ancestry remain healthy beyond 100.
Yes, although genes play a back-seat role to the epigenome, it does seem that
some people are genetical y primed for longevity at the digital level—enjoying
longer lives almost irrespective of how they live, thanks in part to gene variants
that stabilize their epigenomes, preventing the loss of analog information over
time. But Barzilai doesn’t see these people as winners so much as markers—they
represent the potential that most other humans have for long and healthy lives—
and he is fond of pointing out that even if we were never to extend lives past 120,
we know that 120 is possible. “So for most of us,” he has told me, “there are 40
good years stil on the table.”
Barzilai is leading the charge to make metformin the rst drug to be approved
to delay the most common age-related diseases by addressing their root cause:
aging itself. If Barzilai and his col eagues can show metformin has measurable
bene ts in the ongoing Targeting Aging with Metformin (TAME) study, the US
Food and Drug Administration has agreed to consider aging as a treatable
condition. That would be a game changer, the beginning of the end for a world
in which aging is “just the way it goes.”
Barzilai believes that day is coming. He has predicted that the traditional
Hebrew blessing “Ad me’ah ve-essrim shana,” or “May you live until 120,” may
soon need updating, for it wil be a wish not for a long life but for a very average
one.
STAC IT UP
Back in 1999, the story of the sirtuin longevity pathway we discovered in Lenny
Guarente’s lab at MIT was about to get even hotter.
We had nal y gured out a molecular cause of aging in yeast cel s, the rst
for any species. We were stil feeling the glow scientists get when they publish
new work that shows how smart they are. In a series of prominent papers that
had captured the imagination of the scienti c community, we’d reported that
the cause of yeast aging was the movement of Sir2 away from the mating-type
genes to deal with DNA breaks and a whole lot of ensuing genome instability. 27We’d shown that extra copies of the SIR2 gene could stabilize the rDNA and
extend lifespan. We’d linked genetic instability to epigenetic instability and
found one of the world’s rst true longevity genes—and the yeast hadn’t had to
go hungry to receive its bene ts.
But splicing extra copies of a gene into a single-cel ed organism is a much
easier endeavor than putting those copies into more complex creatures. It’s also
far less ethical y complicated. That’s why a few other researchers and I entered a
scienti c race to nd ways to ramp up sirtuin activity in mammals without
inserting extra sirtuin genes.
Here is where science becomes a matter of logical guesswork and some good
old-fashioned luck. Because there are more than 100 mil ion chemicals known to
science. Where do you even start?
Thankful y, Konrad Howitz was on the case. The Cornel -educated
biochemist was then the director of molecular biology for Biomol, a
Pennsylvania company that was a supplier of molecules for life science
researchers. Howitz was looking for chemicals that would inhibit the SIRT1
enzyme, so they could be sold to the growing number of scientists who were
starting to study the enzyme. In the process of evaluating di erent contenders,
he found two chemicals that, rather than inhibiting SIRT1, stimulated or
“activated” it, making it work ten times as fast. That was a serendipitous
discovery, not only because he was expecting to nd inhibitors but because
activators are very rare in nature. They are so rare, in fact, that most drug
companies don’t even bother fol owing up when one is discovered, guring it
must be a mistake.
The rst SIRT1-activating compound, or STAC, was a polyphenol cal ed
setin, which helps gives plants such as strawberries and persimmons their color
and is now known to also kil senescent cel s. The second was a molecule cal ed
butein, which can be found in numerous owering plants as wel as a toxic plant
known as the Chinese lacquer tree. Both had a signi cant e ect on SIRT1,
though not the sort of pedal-to-the-metal reaction that might make them ripe
for further research.
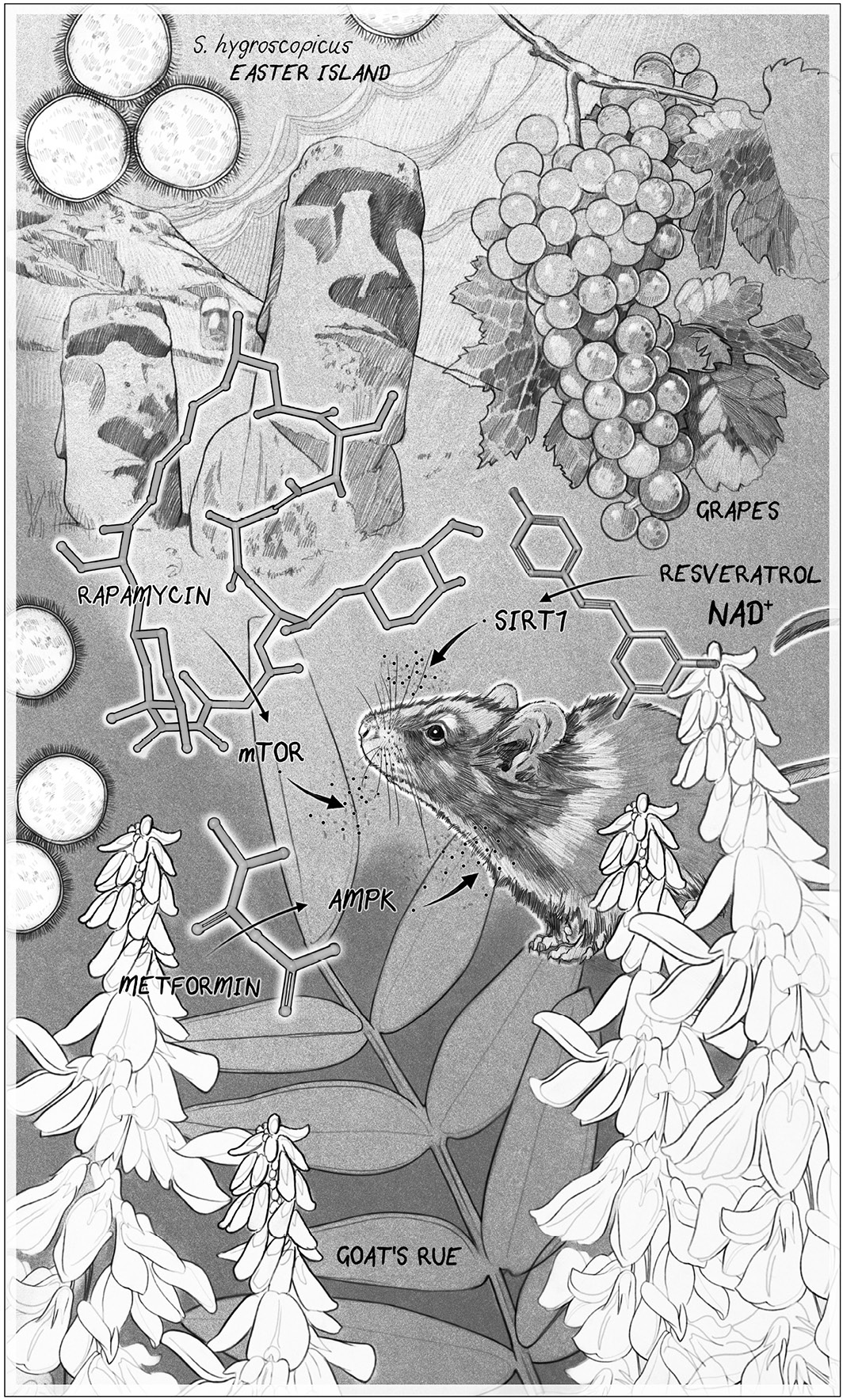
THE THREE MAIN LONGEVITY PATHWAYS, mTOR, AMPK, AND SIRTUINS, EVOLVED TO
PROTECT THE BODY DURING TIMES OF ADVERSITY BY ACTIVATING SURVIVAL
MECHANISMS. When they are activated, either by low-calorie or low-amino-acid diets, or by
exercise, organisms become healthier, disease resistant, and longer lived. Molecules that tweak
these pathways, such as rapamycin, metformin, resveratrol, and NAD boosters, can mimic the
bene ts of low-calorie diets and exercise and extend the lifespan of diverse organisms.
Howitz showed his initial results to Biomol’s founder and scienti c director,
Robert Zipkin, a bril iant chemist and entrepreneur who has an encyclopedic
knowledge of chemical structures. “Fisetin and butein, huh?” Zipkin said. “You
know what those two molecules look like? They’ve got an overlapping structure:
two phenolic rings connected by a bridge. You know what else has that
structure? Resveratrol.”
In 2002, antioxidants were al the rage. They might not have been the
antiaging and health panaceas some believed them to be, but that wasn’t yet
known. One of the antioxidants, scientists from the Karol Marcinkowski
University of Medical Sciences (now Poznan University of Medical Sciences in
Poland) had learned, was resveratrol, a natural molecule that is found in red wine
and that many plants produce in times of stress. 28 A few researchers had suggested that resveratrol might explain the “French paradox,” the fact that theFrench have lower rates of heart disease, even though their diet is relatively high
in foods containing saturated fat, such as butter and cheese.
Zipkin’s guess that resveratrol might have a similar e ect as setin and butein
was right on the money. When I studied it in my lab at Harvard, I saw that it
actual y far outperformed the other two molecules.
As a reminder, aging in yeast is often measured by the number of times a
mother cel divides to produce daughter cel s. In most cases, a yeast cel gets to
about twenty- ve divisions before it dies. Because the experiments required a
week of micromanipulation of cel s while looking down a microscope, and the
fewer times you put the cel s in the fridge to get some sleep, the longer the yeast
cel s live, I assembled a lab at home on my dining room table.
There, I saw something incredible: the resveratrol-fed yeast were slightly
smal er and grew slightly more slowly than untreated yeast, getting to an average
of thirty-four divisions before dying, as though they were calorie restricted. The
human equivalent would be an extra 50 years of life. We saw increases in
maximum lifespan, too—on resveratrol, they kept going past 35. We tested
resveratrol in yeast cel s with no SIR2 gene, and there was no e ect. We tested it
on calorie-restricted yeast, and saw no further increase in lifespan, suggesting
that the same pathway was being activated; this was how calorie restriction was
working.
It seemed like a joke’s punch line—not only had we found a calorie-
restriction mimetic, something that could extend longevity without hunger, but
we’d found it in a bottle of red wine.
Howitz and I were fascinated by the fact that resveratrol is produced in
greater quantities by grapes and other plants experiencing stress. We also knew
that many other health-promoting molecules, and chemical derivatives of them,
are produced in abundance by stressed plants; we get resveratrol from grapes,
aspirin from wil ow bark, metformin from lilacs, epigal ocatechin gal ate from
green tea, quercetin from fruits, and al icin from garlic. This, we believe, is
evidence of xenohormesis—the idea that stressed plants produce chemicals for
themselves that tel their cel s to hunker down and survive. Plants have survival
circuits, too, and we think we might have evolved to sense the chemicals theyproduce in times of stress as an early-warning system, of sorts, to alert our bodies
to hunker down as wel . 29
What this means, if it’s true, is that when we search for new drugs from the
natural world we should be searching the stressed-out ones: in stressed plants, in
stressed fungi, and even in the stressed microbiome populations in our guts. The
theory is also relevant to the foods we eat; plants that are stressed have higher
concentrations of xenohormetic molecules that may help us engage our own
survival circuits. Look for the most highly colored ones because xenohormetic
molecules are often yel ow, red, orange, or blue. One added bene t: they tend to
taste better. The best wines in the world are produced in dry, sun-exposed soil or
from stress-sensitive varietals such as Pinot Noir; as you might guess, they also
contain the most resveratrol. 30 The most delectable strawberries are those that
have been stressed by periods of limited water supply. And as anyone who has
grown leaf vegetables can attest, the best heads of lettuce come when the plants
are exposed to a one-two combo punch of heat and cold.31 Ever wonder why
organic foods, which are often grown under more stressful conditions, might be
better for you?
Resveratrol extended the lifespan of simple yeast cel s, but would it do the
same for other organisms? When my fel ow researcher Marc Tatar of Brown
University visited me in Boston, I gave him a smal vial of white, u y resveratrol
powder—marked only with the letter R—to try on insects in his lab. He took it
back to Rhode Island, mixed it with some yeast paste, and fed it to his fruit ies.
A few months later, I got a cal from him. “David!” he said. “What is this R
stu ?”
Under lab conditions, the fruit y Drosophila melanogaster typical y lives for
an average of forty or so days. “We added a week to their lives and sometimes
more than that,” Tatar told me. “On average, they’re living for more than fty
days.”
In human terms, that’s an additional fourteen years.
In my lab, resveratrol-fed roundworms also lived longer, an e ect that
required the worm sirtuin gene to be engaged. And when we gave resveratrol to
human cel s in culture dishes, they became resistant to DNA damage.
Later, when we fed resveratrol to obese mice at one year of age, somethinginteresting happened: the mice stayed fat, causing postdoctoral fel ow Joseph
Baur, now a professor at the University of Pennsylvania, to conclude that I’d
wasted more than a year of his time, jeopardizing his scienti c career with a
harebrained experiment. But when he and Rafael de Cabo, our col aborator at
the NIH, opened up the mice, they were shocked. The resveratrol mice looked
identical to mice on a normal diet, with healthy hearts, livers, arteries, and
muscles. They also had more mitochondria, less in ammation, and lower blood
sugar levels. The ones they didn’t dissect wound up living about 20 percent
longer than normal.32
Other researchers went on to show in hundreds of published studies that
resveratrol protects mice against dozens of diseases, including a variety of
cancers, heart disease, stroke and heart attacks, neurodegeneration,
in ammatory diseases, and wound healing, and general y makes mice healthier
and more resilient.33 And in col aboration with de Cabo, we discovered that
when resveratrol is combined with intermittent fasting, it can greatly extend
both average and maximum lifespan even beyond what fasting alone
accomplishes. Out of fty mice, one lived more than 3 years—in human terms,
that would amount to about 115 years. 34
The rst paper on resveratrol’s e ects on aging went on to be one of the most
highly cited papers of 200635 and was widely circulated in the mainstream
media, too. We were al over TV, and I was starting to be recognized in public. I
ran o to the little German vil age cal ed Burlo, where my wife was born, and the
news had even made it there. Sales of red wine reportedly went up 30 percent. If
you like red wine but needed a good excuse to imbibe, you can thank Rob
Zipkin.36
On our kitchen wal hangs a variety of cartoons from the day. My favorite is
one by Tom Toles. In it, a wife tries to downplay the enthusiasm of her very large
husband, who covers most of a couch.
“The study said that to get the same dose as they gave the mice you’d have to
drink between 750 and 1,000 glasses of red wine every day,” the wife says.
“The news just keeps getting better and better,” the husband replies.
As it turned out, resveratrol wasn’t very potent and wasn’t very soluble in thehuman gut, two attributes that most medicines need to be e ective at treating
diseases. Despite its limitations as a drug, it did serve as an important rst proof
that a molecule can give the bene ts of calorie restriction without the subject
having to go hungry, and it set o a global race to nd other molecules that
might delay aging. Final y, at least in scienti c circles, slowing aging with a drug
was no longer considered bonkers.
By studying resveratrol, we also learned that it is possible to activate sirtuins
with a chemical. This prompted a ood of research into other sirtuin-activating
compounds, cal ed STACs, that are many times more potent than resveratrol at
stimulating the survival circuit and extending healthy lifespans in animals. They
go by names such as SRT1720 and SRT2104, both of which extend the healthy
lifespan of mice when given to them late in life.37 There are, today, hundreds of
chemicals that have been demonstrated to have an e ect on sirtuins that are even
more e ective than resveratrol’s and some that have already been demonstrated
in clinical trials to lower fatty acid and cholesterol levels, and to treat psoriasis in
humans. 38
Another STAC is NAD, sometimes written as NAD+. 39 NAD has an
advantage over other STACs because it boosts the activity of al seven sirtuins.
NAD was discovered in the early twentieth century as an alcoholic
fermentation enhancer. That was fortuitous: if it hadn’t had the potential to
improve the way we make booze, scientists might not have been so enamored by
it. Instead they worked on it for decades, and in 1938 they had a breakthrough:
NAD was able to cure black tongue disease in dogs, the canine equivalent of
pel agra. It turned out that NAD is a product of the vitamin niacin, a severe lack
of which causes in amed skin, diarrhea, dementia, skin sores, and ultimately
death. And because NAD is used by over ve hundred di erent enzymes,
without any NAD, we’d be dead in thirty seconds.
By the 1960s, however, researchers had concluded that al the interesting
research on NAD had been done. For decades to come, NAD was simply a
housekeeping chemical that teenage biology students had to learn about—with
al the enthusiasm of a teenager doing housekeeping. That al changed in the
1990s, when we began to realize that NAD wasn’t just keeping things running;
it was a central regulator of many major biological processes, including aging anddisease. That’s because Shin-ichiro Imai and Lenny Guarente showed that NAD
acts as fuel for sirtuins. Without su cient NAD, the sirtuins don’t work
e ciently: they can’t remove the acetyl groups from histones, they can’t silence
genes, and they can’t extend lifespan. And we sure wouldn’t have seen the
lifespan-extending impact of the activator resveratrol. We and others also noticed
that NAD levels decrease with age throughout the body, in the brain, blood,
muscle, immune cel s, pancreas, skin, and even the endothelial cel s that coat the
inside of microscopic blood vessels.
But because it’s so central to so many fundamental cel ular processes, no
researchers in the twentieth century had any interest in testing the e ects of
boosting levels of NAD. “Bad stu wil happen if you mess with NAD,” they
thought. But not even having tried to manipulate it, they didn’t real y know
what would happen if they did.
The bene t of working with yeast, though, is that the worst-case scenario in
any experiment is a yeast massacre.
There was little risk in looking for ways to boost NAD in yeast. So that’s
what my lab members and I did. The easiest way was to identify the genes that
make NAD in yeast. We rst discovered a gene cal ed PNC1, which turns
vitamin B3 into NAD. That led us to try boosting PNC1 by introducing four
extra copies of it into the yeast cel s, giving them ve copies in total. Those yeast
cel s lived 50 percent longer than normal, but not if we removed the SIR2 gene.
The cel s were making extra NAD, and the sirtuin survival circuit was being
engaged!
Could we do this in humans? Theoretical y, yes. We already have the
technology to do it in my lab, using viruses to deliver the human equivalent of
the PNC1 gene cal ed NAMPT. But turning humans into transgenic organisms
requires more paperwork and considerably more knowledge about safety—for
the stakes are higher than a yeast massacre.
That’s why we once again began searching for safe molecules that would
achieve the same result.
Charles Brenner, who is now the head of biochemistry at the University of
Iowa, discovered in 2004 that a form of vitamin B3 cal ed nicotinamide riboside,
or NR, is a vital precursor of NAD. He later found that NR, which is found intrace levels in milk, can extend the lifespan of yeast cel s by boosting NAD and
increasing the activity of Sir2. Once a rare chemical, NR is now sold by the ton
each month as a nutraceutical.
Meanwhile, on a paral el path, researchers, including us, were homing in on a
chemical cal ed nicotinamide mononucleotide, or NMN, a compound made by
our cel s and found in foods such as avocado, broccoli, and cabbage. In the body,
NR is converted into NMN, which is then converted into NAD. Give an animal
a drink with NR or NMN in it, 40 and the levels of NAD in its body go up about
25 percent over the next couple of hours, about the same as if it had been fasting
or exercising a great deal.
My friend from the Guarente lab Shin-ichiro Imai demonstrated in 2011 that
NMN could treat the symptoms of type 2 diabetes in old mice by restoring
NAD levels. Then researchers in my lab at Harvard showed we could make the
mitochondria in old mice function just like mitochondria in young mice after
just a week of NMN injections.
In 2016, my other lab at the University of New South Wales col aborated
with Margaret Morris to demonstrate that NMN treats a form of type 2 diabetes
in obese female mice and their diabetes-prone o spring. And back at Harvard,
we found that NMN could give old mice the endurance of young mice and then
some, leading to the Great Mouse Treadmil Failure of 2017, when we had to
reset the tracking program on our lab’s miniature exercise machines because no
one had expected that an elderly mouse, or any mouse, could run anywhere near
three kilometers.
This molecule doesn’t just turn old mice into ultramarathoners; we have used
NMN-treated mice in studies that tested their balance, coordination, speed,
strength, and memory, too. The di erence between the mice that were on the
molecule and the mice that were not was astounding. Were they human, those
rodents would long since have been eligible for senior citizen discounts.
Nicotinamide mononucleotide turned them into the equivalent of contenders
on American Ninja Warrior. Other labs have shown that NMN can protect
against kidney damage, neurodegeneration, mitochondrial diseases, and an
inherited disease cal ed Friedreich’s ataxia that lands active 20-year-olds inwheelchairs.
As I write this, a group of mice that were put on NMN late in life are getting
very old. In fact, only seven out of the original forty mice are stil alive, but they
are al healthy and stil moving happily around the cage. The number of mice
alive that didn’t get the NMN?
Zero.
Every day I’m asked by members of the public, “Which is the superior
molecule: NR or NMN?” We nd NMN to be more stable than NR and see
some health bene ts in mouse experiments that aren’t seen when NR is used.
But it’s NR that has been proven to extend the lifespan of mice. NMN is stil
being tested. So there’s no de nitive answer, at least not yet.
Human studies with NAD boosters are ongoing. So far, there has been no
toxicity, not even a hint of it. Studies to test its e ectiveness in muscle and
neurological diseases are in progress or about to begin, fol owed by super-NAD-
boosting molecules that are a couple of years behind them in development.
But a lot of people haven’t been content to wait for these studies, which can
take years to play out. And that has given us some interesting leads about where
these molecules, or ones like them, might take us.
FERTILE GROUND
We know that NAD boosters are an e ective treatment for a wide variety of
ailments in mice and that they extend their lifespan even when given late in life.
We know that emerging research strongly suggests they could have a similar, if
not duplicative, e ect on human health.
We also know that the way it does this, in terms of the epigenetic landscape, is
by creating the right level of stress—just enough to push our longevity genes
into action to suppress epigenetic changes to maintain the youthful program. In
doing so, NMN and other vitality molecules, including metformin and
rapamycin, reduce the buildup of informational noise that causes aging, thus
restoring the program.
How do they do this? We are stil working to understand how epigeneticnoise is dampened at a molecular level, but we know in principle how it works.
When we give silencing proteins such as sirtuins a boost, they can maintain the
youthful epigenome even with DNA damage occurring, like the long-lived yeast
cel s with extra copies of the SIR2 gene. Somehow they can cope with it. Perhaps
they are just supere cient at repairing DNA breaks and head home before they
get lost, or if half the sirtuins head o , the remaining enzymes can hold down
the fort.
Either way, the increased activity of the sirtuins may prevent Waddington’s
marbles from escaping their val eys. And even if they have started to head out of
the val ey, molecules such as NMN may push them back down, like extra gravity.
In essence, this would be age reversal in some parts of the body—a smal step,
but age reversal nonetheless.
One of the rst clues this might be true in an animal larger than a mouse
came when a student who works in my lab at Harvard came into my o ce one
afternoon.
“David,” he said quietly, “do you have a moment? There is something I need
to discuss. It’s about my mother.”
Given the expression on his face and the tone of his voice, I immediately
worried that my student, who came from another country, would tel me his
mother was sick. Having been half a planet away from my mother when she was
dying, I very much knew how that felt.
“Whatever you need,” I blurted out.
The student seemed taken aback—and I realized I hadn’t yet posed the most
pertinent question. “Is your mother al right?” I asked.
“Yes,” he said. “Wel . . . I mean, yes . . . wel . . . mostly.”
He reminded me that his mother had been taking supplemental NMN, as
some of my students and their family members do. “The thing is, wel ”—his
voice lowered to a whisper—“she has started her, um . . . cycle again.”
It took me a few seconds to realize what cycle he was talking about.
As women approach and go through menopause, the menstrual cycle can
become quite irregular, which is why a year with no periods must go by before
most doctors wil con rm that menopause has occurred.
After that, such bleeding can be a cause for concern, as it could be a sign ofcancer, broid tumors, infections, or an adverse reaction to a medication.
“Has she been seen by a doctor?” I asked.
“Yes,” my student said again. “The doctors say there is nothing wrong. They
said this just looks like a normal period.”
I was intrigued. “Okay,” I said. “What we real y need is more information.
Can you give your mom a cal to ask her some more questions?”
I’ve never seen the color so quickly wash away from someone’s face.
“Oh, David,” he pleaded, “please, please, pleeease don’t make me ask my
mother any more questions about that!”
Since that conversation, which took place in the fal of 2017, I have known a
couple of other women and read the accounts of others claiming to have had
similar experiences. These cases could, perhaps, be the result of a placebo e ect.
But a trial in 2018 to test whether an NAD booster could restore the fertility of
old horses was successful, surprising the skeptical supervising veterinarian. As far
as I know, horses don’t experience the placebo e ect.
Stil , these stories and clinical results could be random chance. These matters
wil be studied in much greater detail. If, however, it turns out that mares and
women can become fertile again, it wil completely overturn our understanding
of reproductive biology.
In school, our teachers taught us that women were born with a set number of
eggs (perhaps as many as 2 mil ion). Most of the eggs die o before puberty.
Almost al the rest are either released during menstruation throughout the
course of a woman’s life or just die o along the way, until there are no more.
And then, we were told, a woman is no longer fertile. Period.
These anecdotal reports of restored menstruation and fertile horses are early
but interesting indicators that NAD boosters might restore failing or failed
ovaries. We also see that NMN is able to restore the fertility of old mice that have
had all their eggs kil ed o by chemotherapy or have gone through
“mousopause.” These results, by the way, even though they were done multiple
times and reproduced in two di erent labs by di erent people, are so
controversial that almost no one on the team voted to publish them. I was the
exception. They remain unpublished, for now.
To me it is clear that we biologists are missing something. Something big.In 2004, Jonathan Til y—a highly controversial gure in the reproductive
biology community—claimed that human stem cel s that can give rise to new
eggs, late in life, exist in the ovaries. Controversial though this theory is, it would
explain how it is possible to restore fertility even in mice that are old or have
undergone chemotherapy. 41,42
Whether or not “egg precursor” cel s exist in the ovary, there’s no doubt in
my mind that we are moving with staggering speed toward a world in which
women wil be able to retain fertility for a much longer portion of their lives and
possibly regain it if it is lost.
Al of this, of course, is good for people who wish to have a child but haven’t
been able to for any number of social, economic, or medical reasons. But what
does it have to do with aging?
To answer that question, we need to remember what an ovary is. It’s not just,
as so many of us were taught in school, a slow-release mechanism for human
eggs. It’s an organ—just like our hearts, kidneys, or lungs—that has a day-to-day
function, both holding on to eggs that were created during embryonic
development and potential y being a repository for additional eggs derived from
precursor cel s later in life.
The ovary is also the rst major organ to break down as a result of aging, in
humans and animal models alike. What that means in mice is that, instead of
waiting for two years for a mouse to reach “old age,” we can start to see and
investigate the causes and cures for aging in about 12 months, at the age female
mice typical y lose their ability to reproduce.
We also have to remember what NMN does: it boosts NAD, and this boosts
the activity of the SIRT2 enzyme, a human form of yeast Sir2 found in the
cytoplasm. SIRT2, we’ve found, controls the process by which an immature egg
divides so that only one copy of the mother’s chromosomes remain in the nal
egg in order to make way for the father’s chromosomes. Without NMN or
additional SIRT2 in old mice, their eggs were toast. Pairs of chromosomes were
ripped apart from numerous directions, instead of exactly two. But if the old
female mice were pretreated with NMN for a few weeks, their eggs looked
pristine, identical to those of young mice.43
Al of this is why early indicators of restored ovarian function in humans,anecdotal as they may be, are so fascinating. If true, the mechanisms that work to
prolong, rejuvenate, and reverse aging in ovaries are pathways we can use to do
the same thing in other organs.
One more thing that is important to bear in mind: NMN is hardly the only
longevity molecule showing promise in this area. Metformin is already widely
used to improve ovulation in women with infrequent or prolonged menstrual
periods as a result of polycystic ovary syndrome.44 Meanwhile, emerging
research is demonstrating that the inhibition of mammalian target of rapamycin,
or mTOR, may be able to preserve ovarian function and fertility during
chemotherapy,45 while the same gene pathway plays an important role in male
fertility, as a central player in the production and development of sperm. 46
LIFE WITH FATHER
Most of the time, rodent studies come long before formal human studies. That
was the case for NAD boosters. But the early indicators of the safety and
e ectiveness of the molecules in yeast, worms, and rodents are such that many
people have already begun their own private human experiments.
My father is among them.
Though he trained as a biochemist, my dad’s passion was computing. He was
a computer guy at a pathology company. That meant he spent a lot of his time
sitting in front of a screen and on his behind—another thing experts say is
devastatingly bad for our health. Some researchers have even suggested it could
be as bad for us as smoking.
By the time my mother died in 2014, my father’s health had also begun its
seemingly inexorable decline. He had retired at 67 and was in his mid-70s, stil
fairly active. He liked to travel and garden. But he had passed the type 2 diabetes
threshold, was losing his hearing, and his eyes were starting to go bad. He would
tire fast. He repeated himself. He was grumpy. He was hardly a picture of
exuberant life.
He started taking metformin for his borderline type 2 diabetes. The next yearhe started taking NMN.
My father has always been a skeptic. But he is also insatiably curious and was
fascinated by what he heard from me about what was happening to the mice in
my lab. NMN isn’t a regulated substance; it’s available as a supplement. So he
tried it out, starting with smal doses.
He knew quite wel , though, that there are very big di erences between mice
and humans. At rst he would say to me and to anyone else who asked,
“Nothing has changed. How would I know?”
So the statement that came about six months into his NMN tryout was
tel ing.
“I don’t want to get carried away,” he said, “but something is happening.”
He was feeling less tired, he told me. Less sore. More mental y aware. “I’m
outpacing my friends,” he said. “They’re complaining about feeling old. They
can’t even come for bushwalks with me anymore. I’m no longer feeling that way.
I don’t have aches or pains. I’m beating much younger people at rowing exercises
at the gym.” His doctor, meanwhile, was struck by the fact that his liver enzymes
normalized after twenty years of being abnormal.
Upon his next visit to the United States, I noticed that something else was
di erent, something very subtle. It dawned on me: for the rst time since my
mother’s death, the smile had returned to his face.
These days, he runs around like a teenager. Hiking for six days through wind
and snow to reach the peak of the highest mountain in Tasmania. Riding three-
wheelers through the Aussie bush. Hunting remote waterfal s in the American
West. Zip-line touring through the forest in northern Germany. Whitewater
rafting in Montana. Ice cave exploring in Austria.
He’s “aging in place,” but he’s rarely at his place. 47
And because he missed working, he took on a new career at one of Australia’s
largest universities, where he sits on the ethics committee that approves human
research studies, taking ful advantage of his knowledge about scienti c rigor,
medical practice, and data security.
You might expect this sort of behavior from someone who had lived his
whole life this way, but he is de nitely not a guy who has lived his whole life this
way. Dad used to say he wasn’t looking forward to getting old. He isn’t outgoingor optimistic by nature; he’s more like Eeyore from Winnie-the-Pooh. He
expected to have a decent ten years of retirement, then go into a nursing home.
The future was clear. He had seen what had happened to his mother. He had
watched helplessly as her health had declined in her 70s and 80s and as she had
su ered from pain and dementia in the nal decade of her life.
With al of that fresh in mind, the idea of living much past his 70s wasn’t very
interesting to him. In fact, it was pretty scary. But he’s pretty happy with how
it’s turning out and wakes up every morning with a deep-seated desire to l his
life with new, exciting experiences. To that end, he faithful y takes his metformin
and his NMN each morning and gets nervous when they start to run low. The
turnaround in his energy, enjoyment of life, and perspective on growing old has
been remarkable. It could al be unrelated to the molecules he’s taking. I suppose
his physical and mental transformation may just be how some people age. But it
sure wasn’t that way for any of my other relatives.
My father is also wondering what to think. We are a family of scientists, after
al . “I can’t be sure that the NMN is responsible,” he told me recently. He
thought about his life for a moment, then smiled and shrugged his shoulders,
“but there’s real y no other explanation.”
Recently, after touring much of the East Coast of the United States, Dad was
heading home to Australia. I sheepishly asked him if he could y back to the
United States for an event being held the fol owing month. I had been named an
O cer of the Order of Australia, an honor bestowed “for distinguished service
to medical research into the biology of ageing, to biosecurity initiatives, and as
an advocate for the study of science,” and there was going to be a ceremony at
the Australian Embassy in Washington, DC.
“Sandra says it’s not fair of me to ask for you to come back,” I told him. “It’s
only four weeks from now, and you’re almost eighty, and it’s a long journey
back, and—”
“I would love to come,” he said, “but I’m just not sure I can t it into my
schedule.”
He canceled some meetings and did t the trip into his schedule, and having
him there, along with Sandra and the kids, ensured that it was one of the best
days of my life. As I looked at Dad, standing with my family, I thought, “This iswhat longer life is al about—having your parents there for life’s important
moments.”
And as he stood there, he later told me, he thought, “This is what longer life
is al about—being around for your children’s important moments.”
My father’s story of reinvigoration is, of course, completely anecdotal. I
won’t be publishing it in a scienti c journal anytime soon—a placebo can be a
powerful drug, after al . There’s simply no way to know if the combo of NMN
and metformin is the reason he’s feeling better or is simply what he started
taking at the time he decided, subconsciously, that it was time for a big change in
his approach to life.
Compel ing evidence that the clock of aging is reversible wil come when
wel -planned double-blind human clinical studies are completed. Until then, I
remain very proud of my father, an average guy who grabbed life by the horns in
his late 70s to start his life anew—a shining example of what life can be like if we
don’t accept aging as “just the way it goes.”
Stil , it’s hard for me and anyone else who has seen what has happened to my
father to not suspect that something special might be going on.
It’s also hard to know what I know, to see what I’ve seen—the results of
experiments and other clinical trials around the world years before the rest of the
world learns about them—and not believe that something profound is about to
happen to humanity.
COME WHAT MAY
By engaging our bodies’ survival mechanisms in the absence of real adversity, wil
we push our lifespans far beyond what we can today? And what wil be the best
way to do this? Could it be a souped-up AMPK activator? A TOR inhibitor? A
STAC or NAD booster? Or a combination of them with intermittent fasting
and high-intensity interval training? The potential permutations are virtual y
endless.
Maybe the research under way on any one of these molecular approaches tobattling aging wil provide half a decade of additional good health. Maybe a
combination of these compounds and an optimal lifestyle wil be the elixir that
gets us a couple of extra decades. Or maybe, as time goes by, our enthusiasm for
these molecules wil be dwarfed by what we discover next.
The discovery of the molecules I have described here can be credited to a lot
of serendipity. But imagine what the world wil discover now that we’re actively
and intentional y looking for molecules that engage our in-built defenses.
Armies of chemists are now working to create and analyze natural and synthetic
molecules that have the potential to be even better at suppressing epigenomic
noise and resetting our epigenetic landscape.
There are hundreds of compounds that have already shown potential in this
area and hundreds of thousands more that are waiting to be researched. And it’s
very possible that there is an as-yet-undiscovered chemical out there, hiding in a
microorganism such as S. hygroscopicus or in a ower such as G. officinalis, that is
just waiting to show us another way to help our bodies stay healthier longer.
And that’s just the natural chemicals—which are typical y many times less
e ective than the synthetic drugs they inspire. Indeed, the emerging analogs of
the molecules I’ve already described are demonstrating tremendous potential in
early-stage human clinical trials.
It wil take some time to sort out which of these molecules are best, when,
and for whom. But we’re getting closer every day. There wil come a time in
which signi cantly prolonged vitality is indeed only a few pil s away; there are
too many promising leads, too many talented researchers, and too much
momentum for it to be otherwise.
Wil any of these be a “cure” for aging? No. What’s likely is that researchers
wil continue to identify molecules that are better and better at promoting both
a reduction of epigenetic noise and a rejuvenation of cel ular tissue. As we do,
we’l be buying time for other advances that wil also lead to signi cantly
prolonged vitality.
But let’s say that doesn’t happen. For the sake of argument, not to mention
emphasis, let’s pretend we live in a world in which none of these molecules had
ever been discovered and no one had ever thought to address aging with apharmaceutical.
That would not change the inevitability of longer and healthier lives. Not at
al . For drugs that engage the ancient survival mechanisms within us are just one
of the many ways that scientists, engineers, and entrepreneurs are setting the
stage for the most signi cant shift in the evolution of our species since . . .
. . . wel , since . . .
. . . forever.
SIXBIG STEPS AHEAD
TO THE EXTENT WE THOUGHT about it—and we seldom did—we used to think
aging would be a very complicated thing to change, if we could change it at al .
For most of human history, of course, we simply saw aging like the coming of
the seasons; indeed, the shift from spring to summer to fal to winter was a
common analogy we used to describe the movement from childhood to young
adulthood to middle age to our “golden years.” More recently, we gured that
aging was inexorable but we might be able to deal with some of the diseases that
made it a less appealing process. Later stil , we gured that we might be able to
attack each of the hal marks and perhaps we could treat a few of the symptoms
at a time. Even then, it seemed as though it would be a huge endeavor.
But here’s the thing: it’s real y not.
Once you recognize that there are universal regulators of aging in everything
from yeast to roundworms to mice to humans . . .
. . . and once you understand that those regulators can be changed with a
molecule such as NMN or a few hours of vigorous exercise or a few less meals . . .
. . . and once you realize that it’s al just one disease . . .
. . . it al becomes clear:
Aging is going to be remarkably easy to tackle.
Easier than cancer.
I know how that sounds. It sounds crazy.
But so did the idea of microorganisms before an amateur scientist namedAntonie van Leeuwenhoek rst described the world of the “smal little animals”
he saw under his homemade microscope in 1671; for hundreds of years to come,
doctors rebel ed against the idea that they needed to wash their hands before
surgery. Now infections, one of the chief reasons patients used to die after
surgery, have become the very thing hospital personnel are most fastidiously
attentive to preventing in the operating room. Just by washing up before
surgery, we have profoundly improved the rates at which patients survive. Once
we understood what the problem was, it was an easy problem to solve.
For goodness’ sake, we solved it with soap.
The idea of vaccines would also have sounded crazy to most people before the
English physician Edward Jenner successful y used uid he had gathered from a
cowpox blister to inoculate an eight-year-old boy named James Phipps in what
today would be an egregiously unethical experiment but at the time sparked a
new era in immunological medicine. Indeed, the idea of giving a patient a little
bit of a disease in order to prevent a lot of disease would have been seen as insane
—even potential y homicidal—to many people until Jenner did it in 1796. We
now know that vaccines are the single most e ective medical intervention in
human history in terms of saving and extending lifespans. So again, once we
understood what the problem was, it was an easy one to solve.
The successes of STACs, AMPK activators, and mTOR inhibitors are a
tremendously powerful indicator that we’re working in an area of our biology
that is upstream of every major aging-associated disease. The fact that these
molecules have been shown to extend the lifespan of virtual y every organism
they’ve been tested on is further evidence that we’re engaging with an ancient
and powerful program to prolong life.1
But there is another pharmaceutical target that could increase our longevity,
just a bit downstream from the processes we believe longevity molecules are
impacting but stil upstream of a lot of the symptoms of aging.
You might recal that one of the key hal marks of aging is the accumulation of
senescent cel s. These are cel s that have permanently ceased reproduction.
Young human cel s taken out of the body and grown in a petri dish divide
about forty to sixty times until their telomeres become critical y short, a point
discovered by the anatomist Leonard Hay ick that we now cal the Hay icklimit. Although the enzyme known as telomerase can extend telomeres—the
discovery of which a orded Elizabeth Blackburn, Carol Greider, and Jack
Szostak a Nobel Prize in 2009—it is switched o to protect us from cancer,
except in stem cel s. In 1997, it was a remarkable nding that if you put
telomerase into cultured skin cel s, they don’t ever senesce.
Why short telomeres cause senescence has been mostly worked out. A very
short telomere wil lose its histone packaging, and, like a shoelace that’s lost an
aglet, the DNA at the end of the chromosome becomes exposed. The cel detects
the DNA end and thinks it’s a DNA break. It goes to work to try to repair the
DNA end, sometimes fusing two ends of di erent chromosomes together,
which leads to hypergenome instability as chromosomes are shredded during cel
division and fused again, over and over, potential y becoming a cancer.
The other, safer solution to a short telomere is to shut the cel down. This
happens, I believe, by permanently engaging the survival circuit. The exposed
telomere, seen as a DNA break, causes epigenetic factors such as the sirtuins to
leave their posts permanently in an attempt to repair the damage, but there is no
other DNA end to ligate it to. This shuts cel replication down, similar to the
way that broken DNA in old yeast distracts Sir2 from the mating genes and
shuts down fertility.
Triggering of the DNA damage response and major alterations to the
epigenome are wel known to occur in human senescent cel s—and when we
introduce epigenetic noise into the ICE cel s they go on to senesce earlier than
untreated cel s, so maybe this idea has merit. I suspect that senescence in nerve
and muscle cel s, which don’t divide much or at al , is the result of epigenetic
noise that causes cel s to lose their identity and shut down. This once-bene cial
response, which evolved to help cel s survive DNA damage, has a dark side: the
permanently panicked cel sends out signals to surrounding cel s, causing them
to panic, too.
Senescent cel s are often referred to as “zombie cel s,” because even though
they should be dead, they refuse to die. In the petri dish and in frozen, thinly
sliced tissue sections, we can stain zombie cel s blue because they make a rare
enzyme cal ed beta-galactosidase, and when we do that, they light up clearly. The
older the cel s, the more blue we see. For example, a sample of white fat lookswhite when we are in our 20s, pale blue in middle age, and dark royal blue in old
age. And that’s scary, because when we have lots of these senescent cel s in our
bodies, it’s a clear sign that aging is getting a strong grip on us.
Smal numbers of senescent cel s can cause widespread havoc. Even though
they stop dividing, they continue to release tiny proteins cal ed cytokines that
cause in ammation and attract immune cel s cal ed macrophages that then
attack the tissue. Being chronical y in amed is unhealthy: just ask someone with
multiple sclerosis, in ammatory bowel disease, or psoriasis. Al these diseases are
associated with excess cytokine proteins.2 In ammation is also a driving force in
heart disease, diabetes, and dementia. It is so central to the development of age-
related diseases that scientists often refer to the process as “in ammaging.” And
cytokines don’t just cause in ammation; they also cause other cel s to become
zombies, like a biological apocalypse. When this happens, they can even
stimulate surrounding cel s to become a tumor and spread.
We already know that destroying senescent cel s in mice can give them
substantial y healthier and signi cantly longer lives. It keeps their kidneys
functioning better for longer. It makes their hearts more resistance to stress.
Their lifespans, as a result, are 20 to 30 percent longer, according to research led
by Mayo Clinic molecular biologists Darren Baker and Jan van Deursen.3 In
animal models of disease, kil ing of senescent cel s makes brotic lungs more
pliable, slows the progression of glaucoma and osteoarthritis, and reduces the
size of al sorts of tumors.
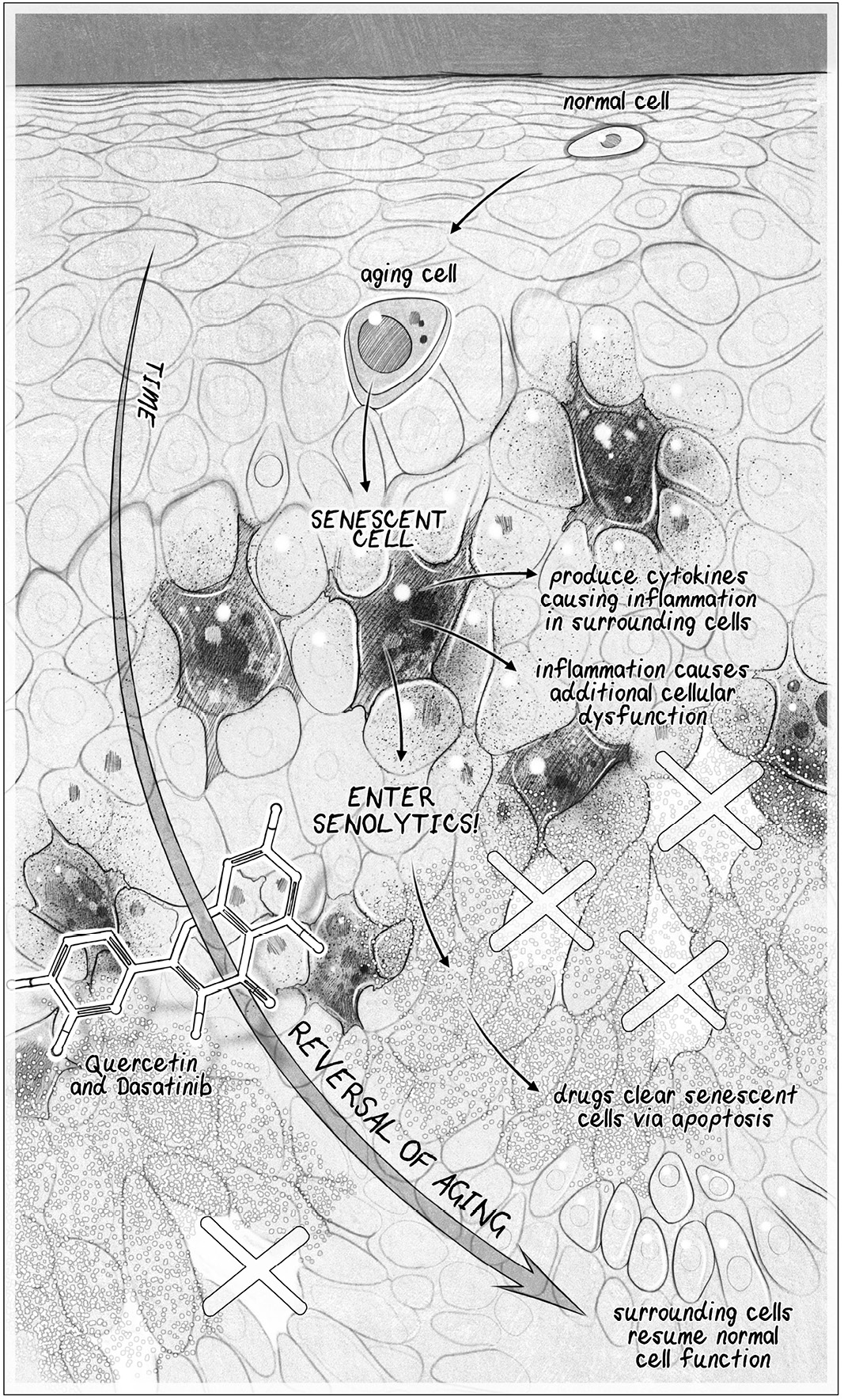
DELETING THE ZOMBIE SENESCENT CELLS IN OLD TISSUES. Thanks to the primordial
survival circuit we’ve inherited from our ancestors, our cel s eventual y lose their identities and
cease to divide, in some cases sitting in our tissues for decades. Zombie cel s secrete factors that
accelerate cancer, in ammation, and help turn other cel s into zombies. Senescent cel s are hard
to reverse aging in, so the best thing to do is to kil them o . Drugs cal ed senolytics are in development to do just that, and they could rapidly rejuvenate us.
Understanding why senescence evolved is not just an academic exercise; it
could help us design better ways to prevent or kil senescent cel s. Cel ular
senescence is a consequence of our inherited primordial survival circuits, which
evolved to stop cel division and reproduction when DNA breaks were detected.
Just as in old yeast cel s, if DNA breaks happen too frequently or they
overwhelm the circuit, human cel s wil stop dividing, then sit there in a panic,
trying to repair the damage, messing up their epigenome, and secreting
cytokines. This is the nal stage of cel ular aging—and it’s not pretty.
If zombie cel s are so bad for our health, why doesn’t our body just kil them
o ? Why are senescent cel s al owed to cause trouble for decades? Back in the
1950s, the evolutionary biologist George Wil iams was already on the case. His
work, built upon by Judith Campisi from the Buck Institute for Research onAging in California, proposes that we evolved senescence as a rather clever trick
to prevent cancer when we are in our 30s and 40s. Senescent cel s, after al , don’t
divide, which means that cel s with mutations aren’t able to spread and form
tumors. But if senescence evolved to prevent cancer, why would it eventual y
promote cancer in adjacent tissue, not to mention a host of other aging-related
symptoms?
This is where “antagonistic pleiotropy” comes into play: the idea that a
survival mechanism that is good for us when we are young is kept through
evolution because this far outweighs any problems it might cause when we get
older. Yes, natural selection is cal ous, but it works.
Consider the 15-mil ion-year history of hominids, the great apes. In the vast
majority of our family’s evolutionary journey, the forces of predation, starvation,
disease, maternal mortality, infection, catastrophic weather events, and
intraspecies violence meant that very few individuals saw more than a decade or
two of life. Even in the relatively recent era of the Homo genus, what we now
think of as “middle age” is an exceptional y new phenomenon.
A life expectancy of 50 and beyond was simply not a reality for most of our
evolutionary history. Therefore, it didn’t matter if a mechanism for slowing the
spread of cancer would eventual y cause more cancer and other diseases, because
it general y worked, as long as it al owed people to breed and rear some children.
The saber-toothed tigers took things from there.
These days, of course, few people have to worry about being picked o by
hungry predators. Hunger and malnutrition are stil far too common, but abject
starvation is increasingly rare. We’re getting better and better at staving o
childhood diseases and have eliminated some of them almost entirely. Childbirth
is an increasingly safe a air (although that, too, is something that can be vastly
improved upon, especial y in the developing world). Modern sanitation has
resulted in tremendous improvements in the rates at which we die of infectious
diseases. Modern technology is helping to warn us of impending catastrophes
such as hurricanes and volcanic eruptions. And although the world often seems
to be a vicious and violent place, the worldwide homicide rate and the numbers
of wars global y have been fal ing for decades.
So we live longer—and evolution hasn’t had a chance to catch up. We’replagued by senescent cel s, which might as wel be radioactive waste. If you put a
tiny dab of these cel s under a young mouse’s skin, it won’t be long before
in ammation spreads and the entire mouse is l ed with zombie cel s that cause
premature signs of aging.
A class of pharmaceuticals cal ed senolytics may be the zombie kil ers we need
to ght the battle against aging on this front. These smal -molecule drugs are
designed to speci cal y kil senescent cel s by inducing the death program that
should have happened in the rst place.
That’s what the Mayo Clinic’s James Kirkland has done. He needed only a
quick course of two senolytic molecules—quercetin, which is found in capers,
kale, and red onions, and a drug cal ed dasatinib, which is a standard
chemotherapy treatment for leukemia—to eliminate the senescent cel s in lab
mice and extend their lifespan by 36 percent. 4 The implications of this work cannot be overstated. If senolytics work, you could take a course of a medicine
for a week, be rejuvenated, and come back ten years later for another course.
Meanwhile, the same medicines could be injected into an osteoarthritic joint or
an eye going blind, or inhaled into lungs made brotic and in exible by
chemotherapy, to give them an age-reversal boost, too. (Rapamycin, the Easter
Island longevity molecule, is what’s known as a “senomorphic” molecule, in that
it doesn’t kil senescent cel s but does prevent them from releasing in ammatory
molecules, which may be almost as good. 5)
The rst human trials of senolytics were started in 2018 to treat osteoarthritis
and glaucoma, conditions in which senescent cel s can accumulate. It wil be a
few more years before we know enough about the e ects and safety of these
drugs to provide them to everyone, but if they work, the potential is vast.
But there is another option, just a bit further upstream, that could be even
better.
THE HITCHHIKER’S GUIDE
The sel sh genes we discussed earlier, cal ed LINE-1 retrotransposons, and theirfossil remnants, make up about half of the human genome, what is often
referred to as “junk DNA.”
It’s a lot of genetic baggage, and they are sneaky buggers. In young cel s, these
ancient “mobile DNA elements,” also known as retrotransposons, are prevented
by chromatin from jumping out of the genome, then breaking DNA to reinsert
themselves elsewhere. We and others have shown that LINE-1 genes are bundled
up and rendered silent by sirtuins. 6 But as mice age, and possibly as we do as
wel , these sirtuins become scattered al over the genome, having been recruited
away to repair DNA breaks elsewhere, and many of them never nd their way
home. This loss is exacerbated by a drop in NAD levels—the same thing we rst
saw in old yeast. Without sirtuins to spool the chromatin and silence the
transposon DNA, cel s start to transcribe these endogenous viruses.
This is bad. And it only gets worse.
Over time, as mice age, the once silent LINE-1 prisoners are turned into
RNA and the RNA is turned into DNA, which is reinserted into the genome at
a di erent place. Besides creating genome instability and epigenomic noise that
causes in ammation, LINE-1 DNA leaks from the nucleus into the cytoplasm,
where it is recognized as a foreign invader. In response, the cel s release even
more immunostimulatory cytokines that cause in ammation throughout the
body.
New work by John Sedivy at Brown University and Vera Gorbunova from
the University of Rochester raises the possibility that one of the main reasons
SIRT6 mutant mice age so rapidly is that these retroviral hel hounds have no
leash, causing numerous DNA breaks and the epigenome to degrade rapidly
instead of slowly. Convincing evidence has come from experiments showing that
antiretrovirals, the same kinds used to ght HIV, extend the lifespan of SIRT6
mutant mice about twofold. It may turn out that, as NAD levels decline with
age, sirtuins are rendered unable to silence retrotransposon DNA. Perhaps one
day, safe antiretroviral drugs or NAD boosters wil be used to keep these
jumping genes silent. 7 We would not have stopped aging completely at its
source, but we would be ghting the battle before total anarchy ensues and the
genie that is aging becomes even harder to put back in the bottle.
VAX TO THE FUTUREIn 2018, scientists at Stanford University reported that they had developed an
inoculation that signi cantly lowered the rates at which mice su ered from
breast, lung, and skin cancer. By injecting the mice with stem cel s inactivated by
radiation and later adding a booster shot like those humans use for tetanus,
hepatitis B, and whooping cough, the stem cel s primed the immune system to
attack cancers that normal y would be invisible to the immune system. 8 Other
immuno-oncological approaches are making even greater strides. Therapies such
as PD-1 and PD-L1 inhibitors, which expose cancer cel s so they can be kil ed,
and chimeric antigen receptors T-cel (CAR-T) therapies, which modify the
patient’s own immune T-cel s and reinject them to go kil cancer cel s, are saving
lives of people who, just a few years before, have been told to go home and make
funeral arrangements. Now, some of these patients are being given a new lease
on life.
If we can use the immune system to kil cancer cel s, it stands to reason that
we can do that for senescent cel s, too. And some scientists are on the case.
Judith Campisi from the Buck Institute for Research on Aging and Manuel
Serrano from Barcelona University believe that senescent cel s, like cancers,
remain invisible to the immune system by waving little protein signs that say,
“No zombie cel s here.”
If Campisi and Serrano are right, we should be able to take away those signs
and give the immune system permission to go kil senescent cel s. Perhaps a few
decades from now a typical vaccine schedule that currently protects babies
against polio, measles, mumps, and rubel a might also include a shot to prevent
senescence when they reach middle age.
When people rst hear that it may be possible to vaccinate against aging,
rather than just treat its symptoms or slow it down, it’s not uncommon for them
to immediately express worries that we are “playing God” or “interfering with
Mother Nature.” Maybe we are, but if so, that’s not unique to people involved
in the ght against aging. We ght diseases of al kinds that God or Mother
Nature gave us. We’ve been doing so for a long time, and we’re going to keep
doing so for a long time to come.
The world rightful y celebrated the eradication of smal pox in 1980. Whenmalaria is likewise eradicated—and I believe it wil be sometime in the coming
decades—our global community wil rejoice once again. And if I could o er the
world a vaccine for HIV, right now, there wouldn’t be many people—no decent
ones, at least—who would say that we should just “let nature run its course.”
These are ailments we’ve long considered diseases, though, and I accept that it
wil take some time to convince people that aging is no di erent.
To this end, I’ve found this thought experiment to be helpful: imagine an
Airbus A380, a double-decker “superjumbo” l ed with six hundred people on
board, on approach to Los Angeles. The plane does not have landing gear, only
parachutes. And al but one of the doors is stuck, so when the passengers
evacuate, one by one, they’l be scattered across the most densely populated area
of the country.
Oh, and one more thing: the passengers are sick. Real y sick. The disease they
carry is highly contagious; it starts with lethargy and sore joints, then develops
into hearing and vision loss, bones as brittle as century-old teacups,
excruciatingly painful heart failure, and brain signals so badly interrupted that
many victims won’t even be able to remember who they are. No one survives
this disease, and death is almost always agonizing.
After a life of faithful service to the United States, you have found yourself
behind the Resolute Desk in the Oval O ce of the White House. The phone
rings. The deputy director for infectious diseases from the Centers for Disease
Control and Prevention tel s you that if even one of the passengers is permitted
to parachute into the greater Los Angeles area, tens of thousands of people wil
catch the disease and die. Each additional parachuter wil increase the projected
death tol exponential y.
The moment you put the receiver down, the phone rings again. The
chairperson of the Joint Chiefs of Sta tel s you that six US Air Force F-22
Raptor ghters are tracking the plane as it circles over the Paci c Ocean. The
pilots have it locked in; their missiles are ready. The plane is running out of gas.
The fate of the passengers, and the entire United States, rests upon your orders.
What do you do?
This, of course, is a “trol ey problem,” an ethical thought experiment, of thetype popularized by the philosopher Philippa Foot, that pits our moral duty not
to in ict harm on others against our social responsibility to save a greater
number of lives. It’s also, however, a handy metaphor, because the highly
contagious disease the passengers are carrying is, as you doubtless have noticed,
nothing more than a faster-acting version of aging.
When presented with the idea of a disease that could infect and kil legions of
people—with horrendous symptoms, no less—very few of us would not make
the horrible but necessary cal to shoot down the plane, taking the lives of
hundreds of people to protect the lives of mil ions.
With that in mind, consider this question: If you would sacri ce hundreds of
human lives to stop a fast-acting version of aging from infecting mil ions, what
would you be wil ing to do to prevent the disease as it actual y occurs in the lives
of everyone on the planet?
Worry not: what I’m about to suggest won’t actual y come at the cost of
human lives. Not hundreds. Not dozens. Not even one. But it would require us
to confront an idea that many people would nd alarming: infecting ourselves
with a virus that would quickly move into every cel in our body, turning us into
genetical y modi ed organisms. The virus wouldn’t kil ; it would do the
opposite.
GET WITH THE REPROGRAM
Vaccines against senescent cel s, CR mimetics, and retrotransposon suppressors
are possible pathways to prolonged vitality, and work is under way already in labs
and clinics around the world. But what if we didn’t need any of that? What if we
could reset the aging clock and prevent cel s from ever losing their identity and
becoming senescent in the rst place?
Yes, the solution to aging could be cel ular reprogramming, a resetting of the
landscape—the way, for instance, that jel y sh have been shown to do by using
smal body fragments to regenerate polyps that spawn a dozen new jel ies.
The DNA blueprint to be young, after al , is always there, even when we areold. So how can we make the cel reread the blueprint? Here it’s helpful to
return to the DVD metaphor. Over time, thanks to use and perhaps misuse, the
digital information encoded as pits in the top layer of aluminum becomes
obscured by some deep and some ne scratches, making it hard for the DVD
player to read the disk. A DVD has thirty miles of data spiraled around the disk
from the edge to the center, so if the disc is scratched, nding the start of a
particular song becomes extremely di cult.
It’s the same situation for old cel s, but far worse. The DNA in our cel s holds
about the same amount of data as a DVD, but in six feet of DNA that’s packed
into a cel a tenth the size of a speck of dust. Together, al the DNA in our body,
if laid end to end, would stretch twice the diameter of the solar system. Unlike a
simple DVD, though, the DNA in our cel s is wet and vibrating in three
dimensions. And there aren’t 50 songs, there are more than 20,000. No wonder
gene reading becomes di cult the older we get; it’s miraculous that any cel nds
the right genes in the rst place.
There are two ways to play an old, scratched DVD with delity. You could
buy a better DVD player, one with a more powerful laser that could reveal the
data under the scratches. Or you could polish the disc to expose the information
again, making the DVD as good as new. I’ve heard that a rag with toothpaste on
it works just ne.
Restoring youth in an organism is never going to be as simple as polishing a
disk with toothpaste, but the rst approach, putting a scratched DVD into a
new player, was. Oxford University professor John Gurdon rst did this in 1958,
when he removed the chromosomes from a frog’s egg and replaced them with
some chromosomes from an adult frog and obtained living tadpoles. Then, in
1996, Ian Wilmut and his col eagues at the University of Edinburgh replaced the
chromosomes of a sheep’s egg with those from an udder cel . The result was
Dol y, whose birth was met with a heated public debate about the purported
dangers of cloning. The debate overshadowed the most important point: that
old DNA retains the information needed to be young again.
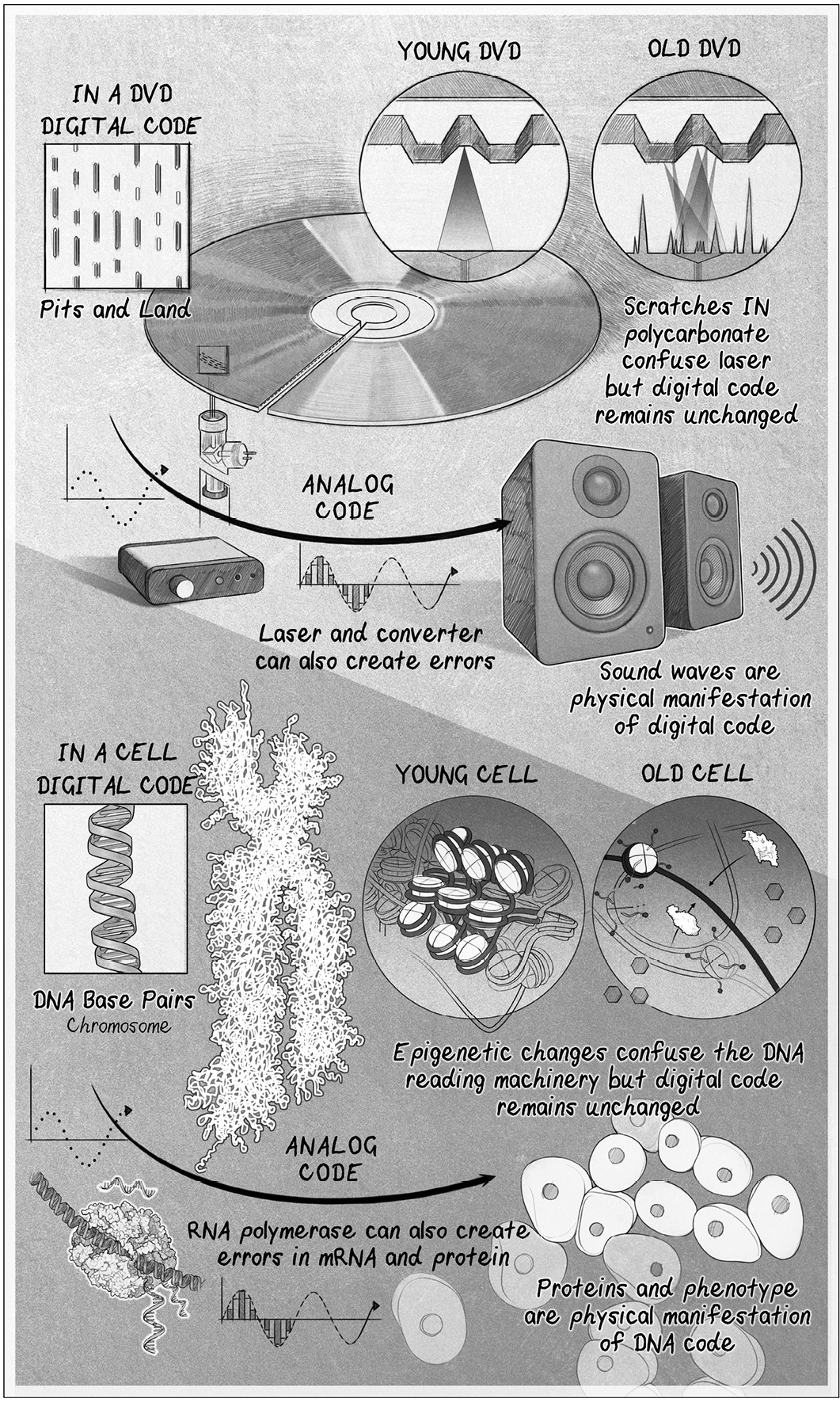
WE ARE ANALOG, THEREFORE WE AGE. According to the Information Theory of Aging, we
become old and susceptible to diseases because our cel s lose youthful information. DNA stores
information digital y, a robust format, whereas the epigenome stores it in analog format, and is
therefore prone to the introduction of epigenetic “noise.” An apt metaphor is a DVD player
from the 1990s. The information is digital; the reader that moves around is analog. Aging is
similar to the accumulation of scratches on the disc so the information can no longer be read
correctly. Where’s the polish?
That debate has since died down; the world today has other concerns.
Cloning is now routinely done to produce farm animals, racehorses, and even
pets. In 2017, you could order up a dog clone for the “bargain” price of $40,000
—or two of them, as Barbra Streisand did to replace her beloved Sammie, a
curly-haired Coton de Tulear. 9 The fact that Sammie was 14 when she died and
donated cel s—that’s somewhere in the range of 75 in dog years—didn’t impact
the clones one bit.
The implications of these experiments are profound. What they show is that
aging can be reset. The scratches on the DVD can be removed, and the original
information can be recovered. Epigenomic noise is not a one-way street.
But how might we reset the body without becoming a clone?In his 1948 publications about the preservation of information during data
transmissions, Claude Shannon provided a valuable clue.10
In an abstract sense, he proposed that information loss is simply an increase
in entropy, or the uncertainty of resolving a message, and provided bril iant
equations to back his ideas up. His work stemmed from the mathematics of
Harry Nyquist and Ralph Hartley, two other engineers at Bel Labs who, in the
1920s, revolutionized our understanding of information transmission. Their
notions of an “ideal code” were important for Shannon’s development of his
communication theory.
In the 1940s, Shannon became obsessed with communications over a noisy
channel, in which information is simply a set of possible messages that needs to
be reconstructed by the recipient of the message—the receiver.
As Shannon bril iantly showed in his “noisy-channel coding theorem,” it is
possible to communicate information nearly error free as long as you don’t
exceed the channel capacity. But if the data exceeds the channel capacity or is
subject to noise, which is often the case with analog data, the best way to ensure
it makes it to the receiver is to store a backup set of data. That way, even if some
primary data are lost, an “observer” can send this “correcting data” to a
“correcting device” to recover the original message. This is how the internet
works. If data packets are lost, they are recovered and resent moments later, al
thanks to Transmission Control Protocol/Internet Protocol (TCP/IP).
As Shannon put it, “This observer notes the errors in the recovered message
and transmits data to the receiving point over a ‘correction channel’ to enable
the receiver to correct the errors.”
Though it may sound like esoteric language from the 1940s, what dawned on
me in 2014 is that Shannon’s “A Mathematical Theory of Communication” is
relevant to the Information Theory of Aging.
In Shannon’s drawing, there are three di erent components that have analogs
in biology:
• The “source” of the information is the egg and sperm, from your parents.
• The “transmitter” is the epigenome, transmitting analog information
through space and time.
• The “receiver” is your body in the future.
When an egg is fertilized, epigenetic information—biological “radio
signals”—is sent out. It travels between dividing cel s and across time. If al goes
wel , the egg develops into a healthy baby and eventual y a healthy teenager. But
with successive cel divisions and the overreaction of the survival circuit to DNA
damage, the signal becomes increasingly noisy. Eventual y, the receiver, your
body when it is 80, has lost a lot of the original information.
We know that cloning a new tadpole or a mammal from an old one is
possible. So even if a lot of the epigenetic information is lost in old age, obscured
by epigenetic noise, there must be information that tel s the cel how to reset.
This fundamental information, laid down early in life, is able to tel the body
how to be young again—the equivalent of a backup of the original data.
CLAUDE SHANNON’S 1948 SOLUTION TO RECOVERING LOST INFORMATION DURING DATA
TRANSMISSIONS LED TO CELL PHONES AND THE INTERNET. It may also be the solution to
reversing aging.
Source: C. E. Shannon, “A Mathematical Theory of Communication,” Bell System Technical
Journal 27, no. 3 (July 1948): 379–423 and 27, no. 4 (October 1948): 623–66.
To end aging as we know it, we need to nd three more things that Shannon
knew were essential for a signal to be restored even if it is obscured by noise:
• An “observer” who records the original data
• The original “correction data”
• And a “correcting device” to restore the original signal
I believe we may have nal y found the biological correcting device.In 2006, the Japanese stem cel researcher Shinya Yamanaka announced to the
world that after testing dozens of combinations of genes, he had discovered that
a set of four—Oct4, Klf4, Sox2, and c-Myc—could induce adult cel s to become
pluripotent stem cel s, or iPSCs, which are immature cel s that can be coaxed
into becoming any other cel type. These four genes code for powerful
transcription factors that each controls entire sets of other genes that move cel s
around on the Waddington landscape during embryonic development. These
genes are found in most multicel ular species, including chimpanzees, monkeys,
dogs, cows, mice, rats, chickens, sh, and frogs. For his discovery, essential y
showing that complete cel ular age reversal was possible in a petri dish, Yamanaka
won the Nobel Prize in Physiology or Medicine along with John Gurdon in
2012. We now cal these four genes Yamanaka factors.
At rst blush, Yamanaka’s experiments might sound like a nifty laboratory
parlor trick. But the implications for aging are profound, and not only because
he paved the way for us to grow entirely new populations of blood cel s, tissues,
and organs in the dish that can be and are being transplanted into patients.
What he identi ed, I believe, is the reset switch responsible for Gurdon’s
tadpoles—the biological correcting device.
I predict, and my students are now showing in the lab, that we can use these
and other switches not just to reset our cel s in petri dishes but to reset an entire
body’s epigenetic landscape—to get the marbles back into the val eys where they
belong—sending sirtuins back to where they came from, for instance. Cel s that
have lost their identity during aging can be led back to their true selves. This is
the DVD polish we’ve been looking for.
We are making progress every week in restoring the youthful epigenome of
mice by delivering reprogramming factors. The pace of discovery is mind
spinning. A ful night of sleep for me and my lab members is increasingly rare.
In the 1990s, there were major concerns about the safety of delivering genes
to humans. But there are a rapidly increasing number of approved gene therapy
products and hundreds of clinical trials under way. Patients with an RPE65
mutation that causes blindness, for example, can now be cured with a simple
injection of a safe virus that infects the retina and delivers, forever, the functionalRPE65 gene.
I predict that cel ular reprogramming in the body wil rst be used to treat
age-related diseases in the eye, such as glaucoma and macular degeneration (the
eye is the organ of choice to trial gene therapies because it is immunological y
isolated). But if the therapy is safe enough to deliver into the entire body—as the
long-term mouse studies in my lab suggest they might one day be—this may be
in our future:
At age 30, you would get a week’s course of three injections that introduce a
special y engineered adeno-associated virus, or AAV, which causes a very mild
immune response, less even than what is commonly caused by a u shot. The
virus, which has been known to scientists since the 1960s, has been modi ed so
it doesn’t spread or cause il ness. What this theoretical version of the virus would
carry would be a smal number of genes—some combination of Yamanaka
factors, perhaps—and a fail-safe switch that could be turned on with a wel -
tolerated molecule such as doxycycline, an antibiotic that can be taken as a
tablet, or, even better, one that’s completely inert.
Nothing, at that point, would change in the way your genes work. But when
you began to see and feel the e ects of aging, likely sometime in your mid-40s,
you would be prescribed a month’s course of doxycycline. With that, the
reprogramming genes would be switched on.
During the process, you’d likely place a drop of blood in a home biotracker or
pay a visit to the doctor to make sure the system was working as expected, but
that’s about it. Over the next month, your body would undergo a rejuvenation
process as Waddington’s marbles were sent back to where they once were when
you were young. Gray hair would disappear. Wounds would heal faster. Wrinkles
would fade. Organs would regenerate. You would think faster, hear higher-
pitched sounds, and no longer need glasses to read a menu. Your body would feel
young again.
Like Benjamin Button, you would feel 35 again. Then 30. Then 25.
But unlike Benjamin Button, that’s where you would stop. The prescription
would be discontinued. The AAV would switch o . The Yamanaka factors
would fal silent. Biological y, physical y, and mental y, you would be a couple ofdecades younger, but you’d retain al your knowledge, wisdom, and memories.
You would be young again, not just looking young but actual y young, free to
spend the next few decades of your life without the aches and pains of middle
age, untroubled by the prospects of cancer and heart disease. Then, a few more
decades down the road, when those gray hairs begin showing up again, you’d
start another cycle of the prescribed trigger.
What’s more, with the pace at which biotech is advancing, and as we learn
how to manipulate the factors that reset our cel s, we may be able to move away
from using viruses and simply take a month’s course of pil s.
Does that sound like science ction? Something that is very far out in the
future? Let me be clear: it’s not.
Manuel Serrano, the leader of the Cel ular Plasticity and Disease laboratory at
the Institute for Research in Biomedicine in Barcelona, and Juan Carlos Izpisua
Belmonte, at the Salk Institute for Biological Studies in San Diego, have already
engineered mice that have al of the Yamanaka factors from birth; these can be
turned on by injecting the mice with doxycycline. In a now-famous study from
2016, when Belmonte triggered the Yamanaka factors for just two days a week
throughout the lifespan of a prematurely aging mouse breed cal ed LMNA, the
mice remained young compared to their untreated siblings and lived 40 percent
longer. 11 He’s shown that the skin and kidneys of regular old mice heal more
quickly, too.
The Yamanaka treatment, however, was highly toxic. If Belmonte overdid it
by giving the mice the antibiotic for a few more days, the mice died. Serrano had
also shown that by pushing the marbles too far up the landscape, the four-gene
combo could induce teratomas, which are particularly disgusting tumors made
up of several types of tissue, such as hair, muscle, or bone. Clearly, this tech is
not ready for prime time. At least not yet. But we’re getting closer every day to
being able to control the Waddington marbles safely, making sure they land back
precisely in their original val eys and not at the top of the mountain, where they
could cause cancer.
While al this was going on, guided by the success of the ICE mouse
experiments, my lab had been looking for ways to delay and reverse epigenetic
aging. We’d tried many di erent approaches: the Notch gene, Wnt, the fourYamanaka factors. Some had worked a little, but most were turning into tumor
cel s.One day in 2016, after failing consistently for two years to get old cels to age
in reverse without turning into tumor cel s, a bril iant graduate student named
Yuancheng Lu came into my o ce to say he was close to quitting. As a nal
e ort, he suggested he try leaving out the c-Myc gene that was the likely cause of
the teratomas, and I encouraged him to do so.
He delivered a viral package to mice, but this time with only three of the
Yamanaka factors, then turned them on using doxycycline and waited for al the
mice to get sick or die. But none of them did. They were total y ne. And after
months of monitoring, no tumors arose, either. It was a surprise to both of us—
a great surprise.
Instead of waiting for another year to see if the mice lived longer, Yuancheng
suggested he use a mouse’s optic nerve as a way to test age reversal and
rejuvenation. I was skeptical.
“I’m not superoptimistic this wil work,” I told him. “Optic nerves just don’t
regenerate, unless you are a newborn.”
The intricate network of cel s and bers that transmit nervous signals across
our bodies is divided into two parts: the peripheral system and the central
system. We’ve known for a long time that peripheral nerves, like those in our
arms and legs, can grow back, albeit very, very slowly. The nerves of the central
system, though—optic nerves and the nerves of the spinal cord—never grow
back. Even those scientists who bucked convention, proposing novel therapies
that could regenerate some aspect of the central system, have general y been
circumspect about the potential for signi cant regrowth. Decades of work
aimed at reversing glaucoma in the eye and spinal cord injury has had almost no
positive momentum.
“You’ve picked the hardest problem in biology to solve,” I told Yuancheng.
“But,” he replied, “if we could solve that problem . . .”
There might have been a thousand ways to measure the impact of age reversal
in mice, but buoyed by his recent successes, he decided to “go big or go home.” I
liked that.
“No one changes the world by not taking risks,” I told him. “Go test it.”The images that came to me in a text message a few months later took my
breath away—so much so that I needed to make sure that what I was seeing was
real.I caled Yuancheng immediately. “Am I seeing what I think I am seeing?”
“Maybe,” he said. “What are you seeing?”
“The future,” I said.
Yuancheng let out a tremendous sigh of relief. “David,” he said, “an hour ago
I thought I was going to fail.”
For researchers, doubt is no vice. Doubt is the very normal and very human
consequence of pushing yourself to do audacious things without knowing how
those things are going to work out.
But on that day, things sure did seem to be working out. The image
Yuancheng rst texted to me looked like an orange, glowing jel y sh; its head
was at the top, where the eye of the mouse sits, with long tentacles owing down
toward the brain. Two weeks earlier, Yuancheng and our col aborators had
squeezed the optic nerve a few mil imeters from the back of the eye with a set of
tweezers, causing almost al the nerve cel axons, the tentacles, to die back toward
the brain. They injected an orange uorescent dye into the eye that is taken up
by living neurons. So when Yuancheng took a microscope and looked below the
crush site, there were no glowing nerves, just a mass of dead cel remnants.
The next picture he sent was an example of one where the reprogramming
virus had been turned on after the crush. Instead of dead cel s, a network of
long, healthy spindly tentacles was making its way to connect up with the brain.
It was the greatest example of nerve generation in history, and Yuancheng was
only just getting started.
No one had really expected the reprogramming to work so wel . One-month-
old mice were initial y chosen for these experiments to give us the greatest chance
of success and because that’s what everyone else does. But Yuancheng and our
skil ed col aborators in Professor Zhigang He’s lab at Children’s Hospital at
Harvard Medical School have now tested our reprogramming regimen on the
damaged optic nerves of middle-aged mice aged twelve months. Their nerves also
regenerate.
As I write this, we have restored vision in regular old mice.Vision declines dramatical y in a mouse by 12 months of age. Bruce Ksander
and Meredith Gregory-Ksander, from Massachusetts Eye and Ear at Harvard,
know this wel . There is a loss of the nerve impulses in the retina, and old mice
don’t move their heads as often when moving lines are displayed in front of
them, because they simply don’t see them.
“David, I must admit,” Bruce said, “I never expected this reprogramming
stu to work on normal aged eyes. I was only testing your virus because you were
so excited to try it.”
The result he had seen the morning before had been the most exciting day in
his research life: our OSK reprogramming virus had restored vision.
A few weeks later, Meredith showed that reprogramming also works to
reverse vision loss caused by internal eye pressure known as glaucoma.
“Do you know what we’ve discovered?” Bruce remarked. “Everyone else has
been working to slow the progression of glaucoma. This treatment restores
vision!”
If adult cel s in the body, even old nerves, can be reprogrammed to regain a
youthful epigenome, the information to be young cannot all be lost. There
must be a repository of correction data, a backup set of data or molecular
beacons, that is retained through adulthood and can be accessed by the
Yamanaka factors to reset the epigenome using the cel ular equivalent of
TCP/IP.
What those youth markers are, we’re stil not sure. They are likely to involve
methyl tags on DNA, which are used to estimate an organism’s age, the so-cal ed
Horvath clock. They likely also involve something else: a protein, an RNA, or
even a novel chemical attached to DNA that we haven’t yet discovered. But
whatever they are made of, they are important, for they would be the
fundamental correcting data that cel s retain over a lifetime that somehow direct
a reboot.
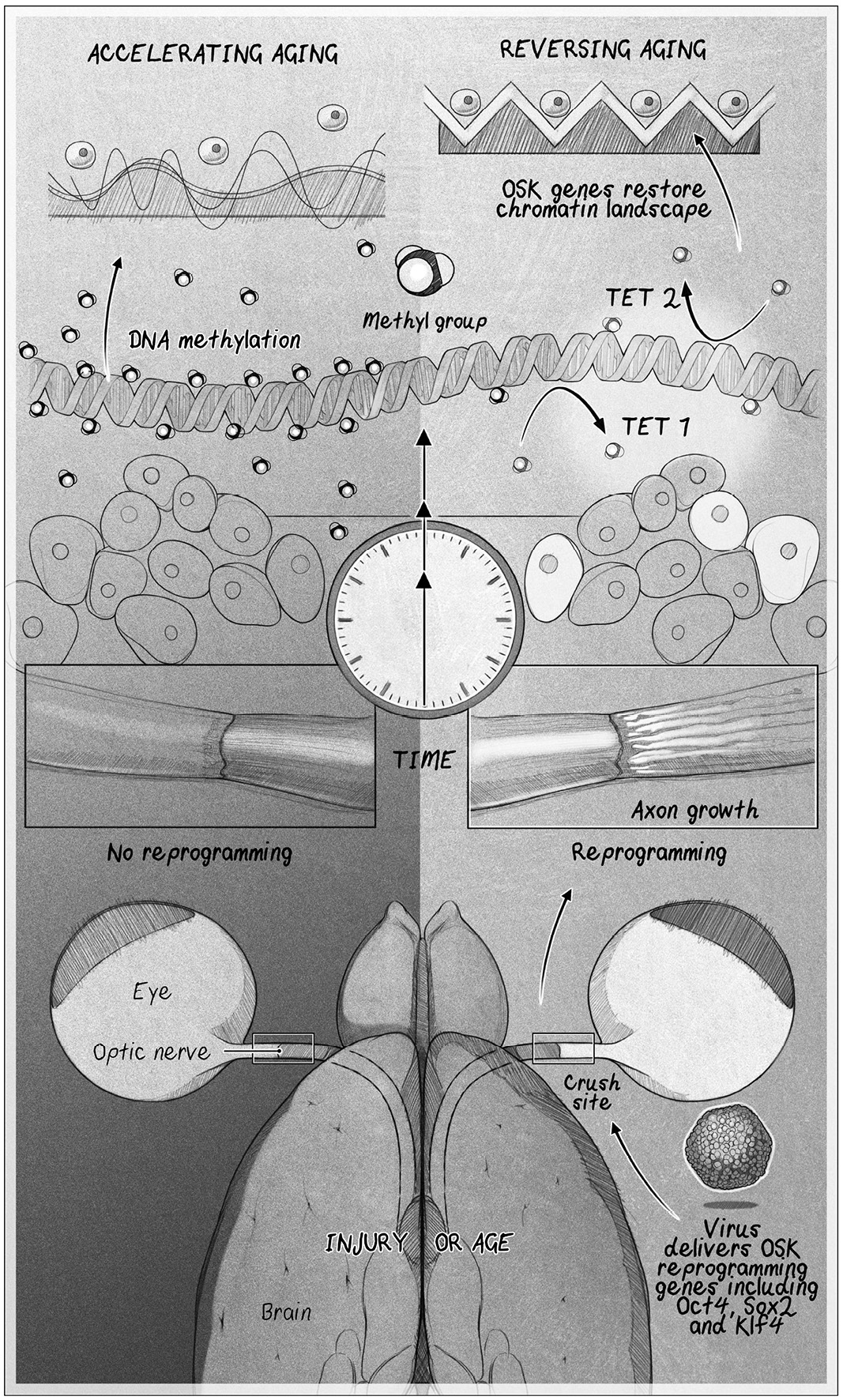
EPIGENETIC REPROGRAMMING REGROWS OPTIC NERVES AND RESTORES EYESIGHT IN OLD
MICE. The Information Theory of Aging predicts that it is a loss of epigenetic rather than
genetic information in the form of mutations. By infecting mice with reprogramming genes
cal ed Oct4, Sox2, and Klf4, the age of cel s is reversed by the TET enzymes, which remove just
the right methyl tags on DNA, reversing the clock of aging and al owing the cel s to survive and
grow like a newborn’s. How the enzymes know which tags are the youthful ones is a mystery.
Solving that mystery would be the equivalent of nding Claude Shannon’s “observer,” the
person who holds the the original data.
We also need to nd the observer, the one who records what the original
signal is when we are young. It can’t just be DNA methylation, because that
doesn’t explain how the reprogrammed cel s know to focus on some of the
youthful methyl marks and strip o the ones that accumulated during aging, the
cel equivalent of the scratches on the DVD. Perhaps it is a specialized histone, or
a transcription factor, or a protein that latches onto methylated DNA when we
are developing in utero and stays there for eighty years waiting until a signal
comes from the correcting device to restore the original information.
In Claude Shannon’s parlance, when the correcting device is switched on by
infecting cel s with OSK genes, the cel somehow knows how to contact the
observer and use the correction data to restore the original signal to that of a young cel .Growing new nerves and restoring eyesight wasn’t enough for Yuancheng.
When the DNA of the damaged neurons was examined, they seemed to be going
through a very rapid aging program, one that was countered by the
reprogramming factors. The neurons that received the reprogramming factors
didn’t age, and they didn’t die. This is a radical idea but one that makes a lot of
sense: severe cel ular injury overwhelms the survival circuit and accelerates aging
of the cel , leading to death, unless the clock is somehow reversed.
With these discoveries, we may be on the verge of understanding what makes
biological time tick and how to wind it back. We know from our experiments
that the biological information correcting device requires enzymes cal ed ten-
eleven translocation enzymes, or TETs, which clip o methyl tags from DNA,
the same chemical tags that mark the passage of the Horvath aging clock. This is
no coincidence, and points to the DNA methylation clock as not just an
indicator of age but a control er of it. It’s the di erence between a wristwatch
and physical time.
In their role as a component of the correcting device, the TETs cannot just
strip o al the methyls from the genome, for that would turn a cel into a
primordial stem cel . We would not have old mice that can see better: we would
have blind mice with tumors. How the TETs know to remove only the more
recent methyls while preserving the original ones is a complete mystery.
It wil likely take another decade and many other labs’ work to know precisely
what the biological equivalent of the TCP/IP information recovery system is.
But whatever it is, eyesight that should not be able to be restored is being
restored and cel s that should not be able to regrow are regrowing.
Compared to the decades of research into how to slow down aging and age-
related disease by a few percent, the reprogramming work has been relatively
quick and easy. Al it took was an intrepid idea and the courage to buck
convention.
The future looks interesting, to say the least. If we can x the toughest-to- x
and regenerate the toughest-to-regenerate cel s in our body, there’s real y no
reason to suspect we cannot regrow any type of cel s our bodies need. Yes, that
could mean xing fresh spinal cord injuries, but it also means regrowing anyother kind of tissue in our body that has been damaged by age: from the liver to
the kidney, from the heart to the brain. Nothing is o the table.
So far, the three-Yamanaka-gene combination seems safe in mice even when
turned on for a year, but there is stil plenty of work to be done. There are a lot
of unanswered questions: Can we deliver the combination to al cel s? Wil it
eventual y cause cancer? Should we keep the genes on or turn them o to let the
cel s rest? Wil this work in some tissues better than others? Can it be given to
middle-aged people, before they become sick, the same way we take statins to
keep cholesterol in check to prevent heart disease?
I have little doubt that cel ular reprogramming is the next frontier in aging
research. One day it might be possible to reprogram cel s via pil s that stimulate
the activity of the OSK factors or the TETs. This may be simpler than it sounds.
Natural molecules stimulate the TET enzymes, including vitamin C and alpha-
ketoglutarate, a molecule made in mitochondria that is boosted by CR and,
when given to nematode worms, extends their lifespan, too.
For now, though, the best bet is gene therapy.
Because it could be so impactful, we should start debating the ethics of this
technology now, before it arrives on our doorstep. The rst question is who
should be al owed to use this technology. A select few? The rich? The very sick?
Should doctors let people who have terminal il nesses try it for so-cal ed
compassionate use? How about people over 100? Or 80? Or 60? When does the
reward outweigh the risk?
There is an army of people wil ing to “boldly go,” sound-minded volunteers
in their 90s and 100s whose bodies have been broken by the disease of aging. I
can assure you that there is no shortage of those who, having peered up the road
at perhaps a few more years of life that is de ned by ever-increasing frailty and
pain, are ready to take a chance at a few more good years, if not for that, then for
the chance to give their children, grandchildren, and great-grandchildren a
longer, healthier life. What do they have to lose, after al ?
The ethics of the technology become more di cult, though, if
reprogramming becomes safe enough to use in a way that is preventive. At what
age should it be given? Does a disease have to appear before an antibiotic
activator of reprogramming is prescribed? If mainstream doctors refuse to help,wil people head overseas? If the technology could signi cantly cut health care
costs, should it be mandated?
And if we can help children live longer, healthier lives, do we have a moral
obligation to do so? If reprogramming technology can help a child repair an eye
or recover from a spinal injury, should the genes be delivered before an accident
happens so they are ready to be switched on at a moment’s notice, starting
perhaps with an antibiotic drip in the ambulance?
If smal pox were to return to our planet, after al , parents who refused to
vaccinate their children would be pariahs of the lowest order. When safe and
e ective treatments are available for a common childhood disease, parents who
refuse to save their children’s lives can have their guardianship overridden by the
doctrine of parens patriae.
Should every human have a choice to su er from aging? Or should that
choice be made, as vaccine decisions are in most cases, for the good of both
individuals and humankind? Wil those who elect to be rejuvenated stil have to
pay for those who have decided not to? Is it moral y wrong not to do so,
knowing you wil prematurely become a burden on family members?
These are theoretical questions today, but they probably won’t remain
theoretical for long.
In late 2018, a Chinese researcher, He Jiankui, reported that he had helped
create the world’s rst genetical y altered children—twin girls whose births
sparked a debate in scienti c circles about the ethics of using gene editing to
make “designer babies.”
The side e ects of inducing DNA damage in embryos and the accuracy of
gene editing are not wel understood yet, which is why the scienti c community
has had such a violent negative reaction. There is also a tacit reason: scientists are
fearful that gene-editing technologies, if abused, wil go the way of GMOs and
be outlawed for political or irrational reasons before their true potential can be
realized.
These fears may be unfounded. If news of the rst genetical y modi ed
children had broken in the 2000s, it would have sparked global debate and
dominated the news for months. Protesters would have stormed labs, and
presidents would have banned this use of the technology on embryos. But howtimes have changed. With a news cycle of hours and politics dished out over the
internet, the story lasted a few days; then the world moved on to other, more
interesting topics.
He’s stated intention was to give the twins the ability to resist HIV. That may
sound admirable, but if I do the numbers, the risk wasn’t worth it. The chance
of contracting HIV in China is less than one in a thousand. If He was going to
maximize health bene ts to o set the risks of the procedure, why not edit a gene
that causes heart disease, which has an almost one-in-two chance of kil ing
them? 12 Or aging, which has a 90 percent chance of kil ing them? HIV
immunity was just the simplest edit, not the most impactful.
As these technologies become commonplace and parents ponder how to get
the biggest bang for the buck, how long wil it be before another rogue scientist
teams up with the world’s most driven helicopter parent to create a genetical y
modi ed family with the capacity to resist the e ects of aging?
It may not be long at al .
SEVENTHE AGE OF INNOVATION
THE FOUR PRESCRIPTION MEDICINES KUHN Lawan was taking were precisely right for
the cancer with which she had been diagnosed. But the drugs weren’t working.
Not even a bit. The elderly Thai woman’s lung cancer persisted. And with it, it
seemed, the end of her life was growing ever nearer.
Her children were understandably distraught. The doctors had told them
that Lawan’s cancer was likely treatable. They seemed to have caught it early,
after al . The fear and uncertainty they’d felt when their mother was rst
diagnosed had been replaced with hope, only to give way, once again, to fear and
uncertainty.
Dr. Mark Boguski has spent a long time thinking about people such as Lawan
and about how modern medicine has long failed so many people like her,
especial y later in life.
“In the most common manner of medical thinking, Lawan was getting the
right care,” he told me one day. “Her doctors in Thailand were top notch. But
that’s the thing about how we do medicine.”
Most doctors, he said, stil rely on early-twentieth-century technology to
diagnose and treat life-threatening diseases. Take a swab and grow it in a petri
dish. Bang the knee and wait for a kick. Breathe in, breathe out. Look to the left
and cough.
When it comes to cancer, doctors note where a tumor is growing and cut outa tissue sample. Then they send it to a lab, where it is put into wax, cut into thin
slices, stained with red and blue dyes, and looked at under the microscope. That
works—sometimes. Sometimes the correct medicine is given.
But sometimes it isn’t. That’s because, the way I see it, looking at a tumor in
this way is the equivalent of a mechanic trying to diagnose a car’s faulty engine
without plugging into the vehicle’s computer. It’s an educated guess. Most of us
accept this sort of approach when it comes to potential y life-and-death
decisions. Yet in the United States alone, with one of the better health care
systems in the world, about 5 percent of cancer patients, or 86,500 people, are
misdiagnosed every year. 1
From the time he began studying computational biology in the early 1980s,
Boguski has been driven by the idea of making medical care more exacting. He is
a luminary in the eld of genomics—and one of the rst scientists engaged in
the Human Genome Project.
“What we cal ‘good medicine’ is doing what works for most of the people
most of the time,” Boguski told me. “But not everyone is most people.”
And so there was a chance, and not a smal one, that Kuhn Lawan was getting
the wrong care. And that might have actual y been making her worse.
But Boguski believes there is hope in a new way of doing medicine. A better
way. A way that uses new technologies, many that are already here but simply
not being utilized to their ful est potential, to refocus our medical system on
individuals—upending centuries of deeply entrenched medical culture and
philosophy. He coined the term precision medicine to describe the promise of
next-generation health monitoring, genome sequencing, and analytics for
treating patients based on personal data, not diagnostic manuals.
Thanks to the plummeting prices of DNA sequencing, wearable devices,
massive computing power, and arti cial intel igence, we’re moving into a world
in which treatment decisions no longer have to be based on what is best for most
people most of the time. These technologies are available to some patients now
and wil be available to most people on the planet in the next couple of decades.
That’s going to save mil ions of lives—and it’s going to extend average healthy
lifespans irrespective of whether we extend maximum lifespans.
But for mil ions of people like Lawan, these advances cannot come soonenough. When her family sought a second opinion in the form of precision
DNA sequencing of her lung tumor biopsy, the totality of the danger she was in
became crystal clear. Lawan did have an aggressive cancer but not the kind of
cancer for which she was being treated. She didn’t have lung cancer; she had a
solid form of leukemia growing in her lung.
In the vast majority of cases in which cancer is found where it was found in
Lawan’s body, it is indeed lung cancer. But now that we can detect the genetic
signature of speci c forms of cancer, using the place where you nd the cancer as
the only guide for what treatment to use is as ridiculous as categorizing an
animal species based on where you’ve located it. It is like saying a whale is a sh
because they both live in water.
Once we have a better idea of what kind of cancer we’re dealing with, we can
better apply emerging techniques for dealing with it. We can even design a
therapy tailored to a patient’s speci c tumor—kil ing it before it has a chance to
grow or spread to another place in the body.
That’s the idea behind one of the cancer- ghting innovations we discussed
earlier, CAR T-cel therapy, in which doctors remove immune system cel s from
a patient’s blood and add a gene that al ows the cel s to bind to proteins on the
patient’s tumor. Grown en masse in a lab and then reinfused into the patient’s
body, the CAR T-cel s go to work, hunting down cancer cel s and kil ing them
by using the body’s own defenses.
Another immuno-oncology approach we discussed earlier, checkpoint
blockade therapy, quashes the ability of cancerous cel s to evade detection by our
immune systems. Much of the early work on this technique was completed by
Arlene Sharpe, whose lab is located on the oor above mine at Harvard Medical
School. In this approach, drugs are used to block the ability of cancer cel s to
present themselves as regular cel s, essential y con scating their fake passports
and thus making it easier for T-cel s to discriminate between friend and foe. This
is the approach that was used, along with radiation therapy, by former president
Jimmy Carter’s doctors to help his immune system ght o the melanoma in his
brain and liver. Prior to this innovation, a diagnosis like his was, without
exception, fatal.
CAR-T therapy and checkpoint inhibition are less than a decade old. Andthere are hundreds of other immuno-oncology clinical trials under way. The
results thus far are promising, with remission rates of greater than 80 percent in
some studies. Doctors who have spent their entire careers ghting cancer say this
is the revolution they’ve been waiting for.
DNA-sequencing technology has also o ered us an opportunity to
understand the evolution of a speci c patient’s cancer. We can take single cel s
from a slice of a tumor, read every letter of the DNA in those cel s, and look at
the cel s’ three-dimensional chromatin architecture. In doing so, we can see the
ages of di erent parts of the tumor. We can see how it has grown, how it has
continued to mutate, and how it has lost its identity over time. That’s
important, because if you look at only one part of a tumor—an older part, for
instance—you could be missing the most aggressive part. Accordingly, you
might treat it with a less e ective therapy.
Through sequencing, we can even see what kinds of bacteria have managed to
make their way into a tumor. Bacteria, it turns out, can protect tumors from
anticancer drugs. Using genomics, we can identify which bacteria are present
and predict which antibiotics wil work against those single-cel ed tumor
protectors.
We can do al of this. Right now. Yet in many hospitals around the world the
if-it-is-here-it-must-be-this and if-the-symptoms-are-this-it-must-be-that modes
of diagnosis are stil practiced. And so, procedural y speaking, the doctors who
treated Lawan had done nothing wrong. They had simply done what doctors al
over the world do, fol owing an empirical process of diagnosis and intervention
that leads to positive outcomes in most people most of the time.
If you accept that this is simply the way we care for people—and that it
usual y produces the right results—you could cal this an understandable
medical approach. But if you picture your own mother accidental y receiving a
cancer treatment she doesn’t need while the medicine that wil save her life sits
on a shelf nearby, you’l probably come to a di erent conclusion about what is,
in fact, “understandable.”
The hardworking, ethical doctors, nurses, and medical professionals who go
to war with death every day, while navigating the overarching standard-of-care
stipulations of governmental programs and insurance companies, should neverbe expected to be perfect. But we can prevent a lot of unnecessary deaths by
giving medical sta more information, just as Lawan’s doctors were able to get
her onto a new treatment regimen once they better understood what they were
dealing with.
Indeed, it wasn’t long after Lawan’s DNA-based diagnosis that she was on a
new treatment regimen—one that was speci c to the actual cancer in her body.
Months later she was doing much better. Hope had returned.
There is hope for al of us. We know that humans, both male and female, are
capable of living past the age of 115. It has been done, and it can be done again.
Even for those who reach only their 100th year, their 80s and 90s could be
among their best.
Helping more people reach that potential is a matter of bringing costs down
and using emerging treatments, therapies, and technologies in a way that truly
puts individuals at the center of their own care. And that’s not just about
diagnosing people right when something does go wrong—it’s also about
knowing what to do for us, as individuals, even before a diagnosis has been
made.
KNOWING THYSELF
Since the new mil ennium, we’ve been told that “knowing our genes” wil help
us understand what diseases we are most susceptible to later in life and give us
the information we need to take preventive actions to live longer. That is true,
but it is only a smal part of the DNA-sequencing revolution that is under way.
There are 3.234 bil ion base pairs, or letters, in the human genome. In 1990,
when the Human Genome Project was launched, it cost about $10 to read just
one letter in the genome, an A, G, C, or T. The entire project took ten years,
thousands of scientists, and cost a few bil ion dol ars. And that was for one
genome.
Today, I can read an entire human genome of 25,000 genes in a few days for
less than a hundred dol ars on a candy bar–sized DNA sequencer cal ed a
MinION that I plug into my laptop. And that’s for a fairly complete readout ofa human genome, plus the DNA methyl marks that tel you your biological age. 2
Targeted sequencing aimed at answering a speci c question—such as “What
kind of cancer is this?” or “What infection do I have?”—can now be done in less
than twenty-four hours. Within ten years, it wil be done in a few minutes, and
the most expensive part wil be the lancet that pricks your nger. 3
But those aren’t the only questions that our DNA can answer. Increasingly, it
can also tel you what foods to eat, what microbiomes to cultivate in your gut
and on your skin, and what therapies wil work best to ensure that you reach
your maximum potential lifespan. And it can give you guidance for how to treat
your body as the unique machine it is.
It’s common knowledge that we don’t al respond to drugs in the same way.
Sometimes these aren’t smal di erences in smal numbers of people. G6PD
genetic de ciency, for instance, a ects 300 mil ion people of primarily Asian and
African descent. It’s the most common genetic disease of humanity. After
ingesting recommended doses of medicines for headaches and malaria and
certain antibiotics, G6PD carriers can be caught unawares by hemolysis—what
amounts to red blood cel mass suicide.4
Some mutations sensitize people to particular foods. If you’re a G6PD
carrier, for example, fava beans can kil you. And while gluten is usual y a
harmless protein that comes in foods rich in the ber, vitamins, and minerals we
need, for those with celiac disease, it’s a poison.
The same is true of medical interventions: our genes can tel us which are
better for us and which could do more harm than good. That’s changing the
game for many breast cancer patients. Those who score in a certain range on a
genetic test cal ed Oncotype DX, it has been discovered, respond every bit as wel
to hormone treatments as they do to chemotherapy, the latter of which has far
more side e ects. 5 The tragedy of this discovery is that it didn’t come until 2015.
The Oncotype DX test has been in use since 2004, but it wasn’t until a team of
researchers decided to take another look at possible treatment options and
outcomes that it became clear that the medical community had been subjecting
tens of thousands of women to treatments that were more harmful and no more
e ective.
What Lawan’s case and this study demonstrate is that we can’t simply rest on“this is how we do it” as a strategy for treating patients. We need to be constantly
chal enging the assumptions upon which medical manuals are based.
One of these assumptions is that males and females are essential y the same.
We’re al too slowly coming around to the shameful recognition that, for most
of medical history, our treatments and therapies have been based on what was
best for males,6 thus hindering healthy clinical outcomes for females. Males
don’t just di er from females at a few sites in the genome; they have a whole
other chromosome.
The bias begins early in the drug development process. Until recently, it was
perfectly ne to study male mice only. Scientists general y aren’t rodent sexists,
but they are always trying to reduce statistical noise and save precious grant
money. Ever since female mice have been regularly included in lifespan
experiments, thanks largely to NIH stipulations, large gender di erences in the
e ects of longevity genes and molecules have been seen. 7 Treatments that work
through insulin or mTOR signaling typical y favor females, whereas chemical
therapies typical y favor males, and no one real y knows why.8
If females and males are in the same environment, in general, females wil live
longer. It’s a common theme throughout the animal kingdom. Scientists have
tested whether it is the X chromosome or the ovary that is important. Using a
genetic trick, they created mice with one or two Xs, with either ovaries or testes.9
Those with a double dose of the X lived longer, even if they had testes and
especial y if they didn’t, thus proving once and for al that female is the stronger
sex.Besides the X, there are dozens of other genetic factors at play. One of the
most promising uses of genomics is predicting how drugs wil be metabolized.
That’s why an increasing number of drugs now have pharmacogenetic labels—
information about how the medication is known to act di erently among
people of di erent genotypes. 10 Examples include the blood-thinning drugs
Coumadin and Plavix, the chemotherapy drugs Erbitux and Vecitibix, and the
depression drug Celexa. In the future, a patient’s epigenetic age wil also be
determined and used to predict drug responses, a new eld cal ed
pharmacoepigenetics. It’s a rapidly advancing technology but somepharmacogenetic tests can’t come soon enough.
For more than two hundred years, the drug digoxin from the digitalis family
of plants has been used in smal doses by doctors to treat failing hearts (and in
larger doses by murderers). 11 Even under a doctor’s supervision, your chance of
death if you are on digoxin increases by 29 percent, according to one study.12
To help reduce uid buildup owing to her weakening heart, my mother was
prescribed digoxin. I had no idea of the risk and I suspect neither did my mother,
who was sensitive to the drug. She steadily declined from living a reasonably
normal life to being barely able to walk. Fortunately, my father, a biochemist and
a pretty smart guy, diagnosed the problem: the amount of the drug prescribed
was superlow, but it had been accumulating in my mom’s heart. He told the
doctor to test for drug levels, which she reluctantly agreed to do, and the test
came back positive for an overdose.
The drug was immediately discontinued, and my mom recovered to her
original self in a matter of weeks. Yes, the doctor should have done regular blood
tests for drug levels, but if a test for sensitivity to digoxin prior to prescription
existed, the doctor could have been on high alert. How close are we to a test?
Not close enough. A few studies have identi ed genetic variants that predict
digoxin blood levels and risk of death, but they haven’t been repeated.13
Hopeful y, there wil be a pharmacogenetic test for this drug soon, as wel as for
many more. They are badly needed. We cannot keep prescribing medicines as
though we al respond to them the same way, because we don’t.
Drug developers have gured this out. They are using genomic information
to nd new and revive failed drugs that work for people with speci c genetic
variations. One of these drugs is Bayer’s Vitrakvi, known generical y as
larotrectinib, which is the rst of many drugs to be designed from the beginning
to treat cancers with a speci c genetic mutation, not where in the body the
cancer came from. A similar story is being written about the failed blood
pressure drug Gencaro. It worked wel on a subset of the population and, if
revived by the FDA, would become the rst heart drug to require a genetic test.
This is the future. Eventual y, every drug wil be included in a huge and ever-
expanding database of pharmacogenetic e ects. It won’t be long before
prescribing a drug without rst knowing a patient’s genome wil seem medieval.And vital y, with genomic information aiding in our doctors’ decisions, we
won’t have to wait to become sick to know what treatments wil work best to
prevent those diseases from developing in the rst place.
As Julie Johnson, the director of the University of Florida’s Personalized
Medicine Program, has pointed out, we are about to enter a world in which our
genomes wil be sequenced, stored, and already red-lighted for treatments that
have been demonstrated to have adverse e ects on people with similar gene types
and combinations as we have.14 Likewise, we’l be green-lighted for treatments
that are known to work for people with similar genes, even if those treatments
don’t work for most other people most of the time. This wil be particularly
important in developing countries, where the local genetics and gut ora are
wildly di erent from the population the drug was tested on.15 These di erences
are rarely talked about in medical circles, but they can have a marked e ect on
drug e cacy and patient survival, including the e cacy of what are thought of
as wel -understood cancer chemotherapies.16
We are also learning to read the entire human proteome—al of the proteins
that can be expressed by every type of cel . Researchers in my lab and others have
discovered hundreds of new proteins in human blood, and each protein can tel
a story about the kind of cel from which it came, a story we can use to
understand what diseases are in our bodies long before they are detectable any
other way. That wil o er a faster, better view of the problems we’re facing,
giving doctors the ability to target those problems with far greater precision.
Right now, when people fal il , especial y older people, they often wait to see
if things just “work themselves out” before making an appointment to see a
doctor. Only when symptoms persist do they make the cal . Then they have to
wait—nearly a month, according to one 2017 study—before they are able to see
a physician. That wait time has been growing in recent years, owing to the
combination of a doctor shortage and an increase in baby boomer patients. And
in some places, it’s much worse. In the city in which I live, Boston—home to
twenty-four of the best hospitals in the world—the wait is fty-two days.17
That’s atrocious.
Long wait times aren’t just in the United States, which has a largely privatemedical system; Canada’s socialized system has notoriously long wait times, too.
The problem isn’t how we pay for care; the problem is that we’ve set up doctors
as the only conduits to diagnosis and often, in the case of primary care
physicians, as the only people who can refer a patient to a specialist.
The backlog could clear soon, thanks to technologies that give doctors the
ability to conduct video home visits. Within a decade, using a device the size of a
package of gum and possibly even disposable, it wil be technical y feasible to
col ect the samples your doctor needs at home, plug the device into your
computer, and look together at a readout of your metabolites and your genes.
There are more than a hundred companies just in the United States pursuing
lightning-fast, superfocused DNA testing that can o er us early and accurate
diagnoses of a vast range of ailments and even estimate our rate of biological
aging. 18 A few are aimed at detecting the genetic signature of cancer and other
il nesses years before they can normal y be detected. Soon, we wil no longer have
to wait for tumors to grow so big and so heterogeneously mutated that their
spread is no longer control able. With a simple blood test, doctors wil be able to
scan for circulating cel -free DNA, or cfDNA, and diagnose cancers that would
be impossible to spot without the aid of computer algorithms optimized by
machine learning processes trained on thousands of cancer patient samples.
These circulating genetic clues wil tel you not just if you have cancer but what
kind of cancer you have and how to kil it. They wil even tel you where in your
body an otherwise undetectable tumor is growing, since the genetic (and
epigenetic) signatures of tumors in one part of the body can be vastly di erent
from those from other parts.19
Al of this means we’re on the way to a fundamental shift in the way we
search for, diagnose, and treat disease. Our awed, symptom- rst approach to
medicine is about to change. We’re going to get ahead of symptoms. Way ahead.
We’re even going to get ahead of “feeling bad.” Many diseases, after al , are
genetical y detectable long before they are symptomatic. In the very near future,
proactive personal DNA scanning is going to be as routine as brushing our
teeth. Doctors wil nd themselves saying the words “I just wish we’d caught this
earlier” less and less—and eventual y not at al .
But the coming age of genomics is just the start.THE RIGHT TRACK
The dashboard on a car equipped with intel igent vehicle technologies is a
marvelous thing. It can tel you how fast you’re going, of course, and how many
miles the vehicle has remaining before it needs a re l —adjusted second by
second based on the conditions of the road and the way in which you are
driving. It can tel you the temperature outside, inside, and under the hood. It
can tel you what cars, bicycles, and pedestrians are around you and warn you if
they’re getting a little close for comfort. When something is wrong—a tire with
too little air or a transmission that isn’t shifting perfectly—it can let you know
that, too. And if you get a bit distracted and begin to veer over the line, it wil
take control of the wheel and pul you back on course or drive autonomously
down the highway, with no more than a bit of resistance from a hand on the
steering wheel to tel it there’s a human there, just in case.
Back in the 1980s, there were very few sensors in cars. But by 2017, there
were nearly 100 sensors in each new vehicle—a number that had doubled in the
prior couple of years. 20 Car buyers increasingly expect features such as tire sensors, passenger sensors, climate sensors, nighttime pedestrian warning
sensors, steering angle guides, proximity alerts, ambient light sensors, washer
uid sensors, automatic high-beam, rain sensors, blind spot detection sensors,
automatic suspension lift, voice recognition, automatic reverse parking, active
cruise control, auto emergency braking, and autopilot.
Perhaps there are people out there who’d be happy to drive without any
dashboard at al , relying solely on their intuition and experience to tel them how
fast they are going, when their car needs fuel or recharging, and what to x when
something goes wrong. The vast majority of us, however, would never drive a car
that wasn’t giving us at least some quantitative feedback, and, through our
purchasing decisions, we have made it clear to car companies that we want more
and more intel igent cars.
Of course we do. We want them to protect us, and we want them to last.
Surprisingly, we’ve never demanded the same for our own bodies. Indeed, weknow more about the health of our cars than we know about our own health.
That’s farcical. And it’s about to change.
We’ve already taken some pretty big steps into the age of personal biosensors.
Our watches monitor our heart rate, measure our sleep cycles, and can even
provide suggestions for food intake and activity. Athletes and health conscious
individuals are increasingly wearing sensors twenty-four hours a day that
monitor the ways in which their vital signs and major chemicals are rising and
fal ing in response to diet, stress, training, and competition.
As just about anyone with diabetes or HIV can attest, blood sugar and blood
cel monitoring are exceptional y easy and increasingly painless a airs these days,
with noninvasive and minimal y invasive monitoring technologies ever more
available, a ordable, and accurate.
In 2017, the US Food and Drug Administration approved a glucose sensor,
rst launched in Europe in 2014, that you stick on your skin to provide a
constant readout of blood sugar levels on your phone or watch. In thirty
countries, a nger prick for diabetics is becoming a distant memory.
Rhonda Patrick, a longevity scientist turned health and tness expert, has
been using a continual blood glucose–sensing device to see what foods give her
body a major sugar spike, something many of us believe is to be avoided if we are
to give ourselves the greatest chance of a long life. She’s seen that, at least for her,
white rice is bad and potatoes aren’t so bad. When I asked her what food had
been the most surprising, she didn’t hesitate.
“Grapes!” she exclaimed. “Avoid grapes.”
Researchers at MIT are working on scanners, straight out of Star Trek, that
can give readouts of thousands of biomarkers. Meanwhile, researchers at the
University of Cincinnati have been working with the US military to develop
sensors that can identify diseases, diet changes, injuries, and stress through
sweat. 21 A few companies are developing handheld breath analyzers that can
diagnose cancer, infectious diseases, and in ammatory diseases. Their mission:
to save 100,000 lives and $1.5 bil ion in health care costs.22 Numerous other companies are working on designing clothing with sensors that can track
biomarkers, and automotive engineers are exploring putting biosensors in car
seats that would send an alert to your dashboard or doctor if there’s somethingamiss in your heart rate or breathing pattern.
As I write this, I am wearing a regular-sized ring that is monitoring my heart
rate, body temperature, and movements. It tel s me each morning if I slept wel ,
how much I dreamed, and how alert I wil be during the day. Technology like
this has been around for some time, I suppose, for people such as Bruce Wayne
and James Bond. Now it costs a few hundred dol ars and can be ordered by
anyone online. 23
Recently, my wife and eldest child came home with matching ear piercings,
which got me thinking: there’s real y no reason that an even smal er piece of
body jewelry—particularly one that pierces the skin—couldn’t be used to track
thousands of biomarkers. Every member of the family could be measured:
grandparents, parents, and children. Even infants and four-legged family
members wil have monitors on them, because they are the ones who are least
able to tel us what they are feeling.
Eventual y, I suspect, very few people wil want to live without tech like this.
We won’t leave home without it, the same way we feel about our smartphones.
The next iteration wil be innocuous skin patches, eventual y giving way to
under-skin implants. Future generations of sensors wil measure and track not
only a person’s glucose but his or her basic vital signs, the level of oxygen in the
blood, vitamin balance, and thousands of chemicals and hormones.
Combined with technologies that coalesce data from your day-to-day
movements and even the tone of your voice,24 your biometric vitals wil be the
bel wether for your body. If you are a man who has been spending more time in
the bathroom than usual, your AI guardian wil check for prostate-speci c
antigens and prostate DNA in your blood, then book you an appointment for a
prostate exam. Changes in how you move your hands while speaking, and even
the manner in which your strike the keys on your computer,25 wil be used to
diagnose neurodegenerative diseases years before symptoms would be noticed by
you or your doctor.
One biotechnological advancement at a time, this world is coming, and fast.
Real-time monitoring of our bodies, the likes of which we could hardly have
imagined a generation ago, wil be as inherent to the experience of living as
dashboards are to the experience of driving. And for the rst time in history, thatwil permit us to make data-driven day-to-day health decisions.26
The most critical daily decisions that a ect how long we live are centered
around the foods we eat. If your blood sugar is high at breakfast, you’l know to
avoid sugar in your morning co ee. If your body is low on iron at lunch, you’l
know it and can order a spinach salad to compensate. When you get home from
work, if you’ve failed to go outside for your daily dose of vitamin D from the
sun, you’l know that, too, and you’l be able to mix up a smoothie that wil
address the de ciency. If you’re on the road and you need X vitamin or Y
mineral, you’l know not only what you need but where to get it. Your personal
virtual assistant—the same AI-driven being who answers your internet search
queries and reminds you about your next meeting—wil point you to the nearest
restaurant that has what you need or o er to have it delivered by a drone to
wherever you are. It could, quite literal y, be dropped into your hands from the
sky.Biometrics and analytics already tel us when and how much to exercise, but
increasingly they wil also help us monitor the e ects of our exercise—or lack
thereof. And our levels of stress. And even how the uids we drink and the air
we’re breathing are impacting our body’s chemistry and functionality.
Increasingly, our devices wil o er recommendations on what to do to mitigate
suboptimal blood biomarkers: to take a walk, meditate, drink a green tea, or
change the lter on the air conditioner. This wil help us make better decisions
about our bodies and our lifestyles.
Al of this is coming soon. There are companies that are crunching data from
hundreds of thousands of blood tests, comparing them to customers’ genomes
and providing feedback to them on what to eat and how to truly optimize their
particular bodies, and looking to rol out new generations of these technologies
every year.
I am fortunate to have been one of the rst people to get an early look at what
this sort of technology can o er us. I am a scienti c adviser to a local company,
spun out of MIT, cal ed InsideTracker. 27 By signing up for regular tests, I have
been able to fol ow a few dozen blood biomarkers over the past seven years,
including vitamins D and B12, hemoglobin, zinc, glucose, testosterone,
in ammatory markers, liver function, muscle health markers, cholesterol, andtriglycerides. My tests are taken every few months instead of every few seconds—
as they wil soon be in our future—but the reports, adjusted to my speci c age,
sex, race, and DNA, have been instrumental in helping me choose what to order
when I sit down at a restaurant and what to pick up when I stop at the market
on my way home. I can even have daily text messages, based on my most recent
results, that remind me what my body needs.
Along the way, I am creating data speci c to my body. And over time, that
data is helping me identify negative and positive trends that may be subtly
di erent for me than for other people. We know, of course, that our genetic
heritage can have a signi cant impact on the sorts of food our bodies need,
tolerate, or reject, but everyone’s genetic heritage is di erent. What you need,
what your partner needs, and what your children need can likely be found in the
meals you put on your table, but the particulars may be quite di erent.
Biotracking wil also help us stop acute and traumatic preventable deaths—
by the mil ions. In 2018, a peer-reviewed study published by the team at
InsideTracker and me, showed that biotracking and computer-generated food
recommendations reduce blood sugar levels as e ciently as the leading diabetes
drug, while optimizing other health biomarkers, too.
The signs of an increasingly blocked carotid artery might be hard to notice in
our day-to-day lives, or even in periodic visits to the doctor, but they wil be
almost impossible to miss when our bodies are being measured and monitored
al the time. Same, too, for heartbeat irregularities, minor strokes, venous
blockages during air medical transport, and many other medical problems that
currently are almost always treated in critical care conditions—when it is too
late. Before, if you suspected your heart was malfunctioning, and even if you
didn’t, it would take a visit to a couple of doctors to get an electrocardiogram.
Now mil ions of people can conduct their own accurate ECG in 30 seconds,
wherever they are, just by pressing their nger to the dial on their watch.

TECHNOLOGIES TO EXTEND OUR LIVES. In the near future, families wil be monitored by
biosensing wearables, smal devices at home, and implants that wil optimize our health and save
lives by suggesting meals and detecting fal s, infections, and diseases. When an anomaly is found,
an AI-assisted, videoconferenced doctor wil send an ambulance, a nurse, or medicines to your
door.
Of course, I use the term watch loosely, given that today’s wrist devices don’t
just tel you the time and date. They are also calendars, audiobooks, tness
trackers, email and text programs, newsstands, timers, alarms, weather stations,
heart rate and body temperature monitors, voice recorders, photo albums, music
players, personal assistants, and phones. If these devices can do al of that, there’s
no reason we should not expect them to help us avoid traumatic health
incidents, too.
In the future, if you are experiencing a heart attack—even if it’s perceptible
only as a slight pain in your arm—or a ministroke, which so often goes
undiagnosed until it’s identi ed on a brain scan years later, you’l be alerted, and
so wil those around you who need to know. In an emergency, a trusted
neighbor, a best friend, or whatever doctor happens to be closest to you can also
be alerted. An ambulance wil be dispatched to your door. This time, the doctorsat the nearest hospital wil know exactly why you are coming in before you even
arrive.
Do you know an emergency room doctor? Ask her about the value of a single
minute of additional treatment time. Or a single blood test’s worth of additional
information. Or a recent electrocardiogram. Or a patient who is stil conscious,
not in pain, and not su ering from a loss of blood to their brain when they
arrive—a person who is able to help in the process of making appropriate
emergency health care choices. It may not be long before medics routinely ask
for a download of your most recent biotracking data to aid them in making what
could be life-and-death decisions.
Biotracking is already helping us identify diseases faster than ever before.
That is what happened in the summer of 2017 to a woman named Suzanne.
After a time of subtle shifts in her menstrual cycle, changes her doctor very
reasonably attributed to a shift into menopause, the 52-year-old woman
downloaded an app that helped her track her periods. Three months later, the
app sent her an email alerting her to the possibility that her data might be
“outside the norm” for women of her age. Armed with that data, Suzanne
returned to her doctor. She was immediately sent for blood tests and an
ultrasound that revealed mixed Mül erian tumors, a malignant form of cancer
found predominantly in postmenopausal women over the age of 65. It took a
radical hysterectomy to remove the cancer before it could spread further, but
Suzanne’s life was spared. 28
The app she used was relatively simple compared to those that are on the way.
It required proactive data entry and tracked only a few metrics. Yet it saved her
life. Imagine, then, what “hands-o ” trackers that col ect mil ions of daily data
points can o er us. Now imagine coupling that data with what we learn from
routine DNA sequencing.
And don’t stop imagining there. Because biotracking won’t just tel you
when your heart rate is up, your vitamin levels are low, or your cortisol level is
spiking, it wil also tel us when our bodies are under attack—and that could save
everyone on this planet.
READY FOR THE WORSTIn 1918—long before our modern, superfast, hyperconnected global
transportation network took shape—a worldwide in uenza pandemic that some
historians believe originated in the United States kil ed more people in absolute
numbers than any other disease outbreak in history.29 It was a violent death, with hemorrhage from mucous membranes, especial y from the nose, stomach,
eyes, ears, skin, and intestines. 30 At a time in which the era of human ight was
in its infancy and most people had never ridden in a car, the H1N1 virus found
its way to some of the furthest reaches of our globe. It kil ed people on remote
islands and in arctic vil ages. It kil ed without regard to race or national
boundaries. It kil ed like a new Black Death. Average life expectancy in the
United States plummeted from 55 to 40 years. It recovered, but not until more
than 100 mil ion people of al ages global y had had their lives cut short.
This could happen again. And given how much more humans and animals
are in contact and how much more interconnected our planet is now than it was
a century ago, it could happen quite easily.
The gains in life expectancy we’ve witnessed over the past 120 years, and
those to come, could be wiped out for a generation unless we address the greatest
threat to our lives: other life-forms that seek to prey on us. It doesn’t matter if we
live decades upon decades longer if a pandemic quickly snu s out hundreds of
mil ions of lives—negating and even rol ing back the gains in average lifespan we
wil have achieved. Global warming is a long-term, critical issue to deal with, but
one could also argue that, at least within our lifetimes, infections are our greatest
threat.
Ensuring the next big outbreak never happens could be the greatest gift of
the biotracking revolution. Individual y, of course, real-time monitoring of vitals
and body chemicals o ers incredible bene ts for optimizing health and
preventing emergencies. Col ectively, though, it could help us get ahead of a
global pandemic.
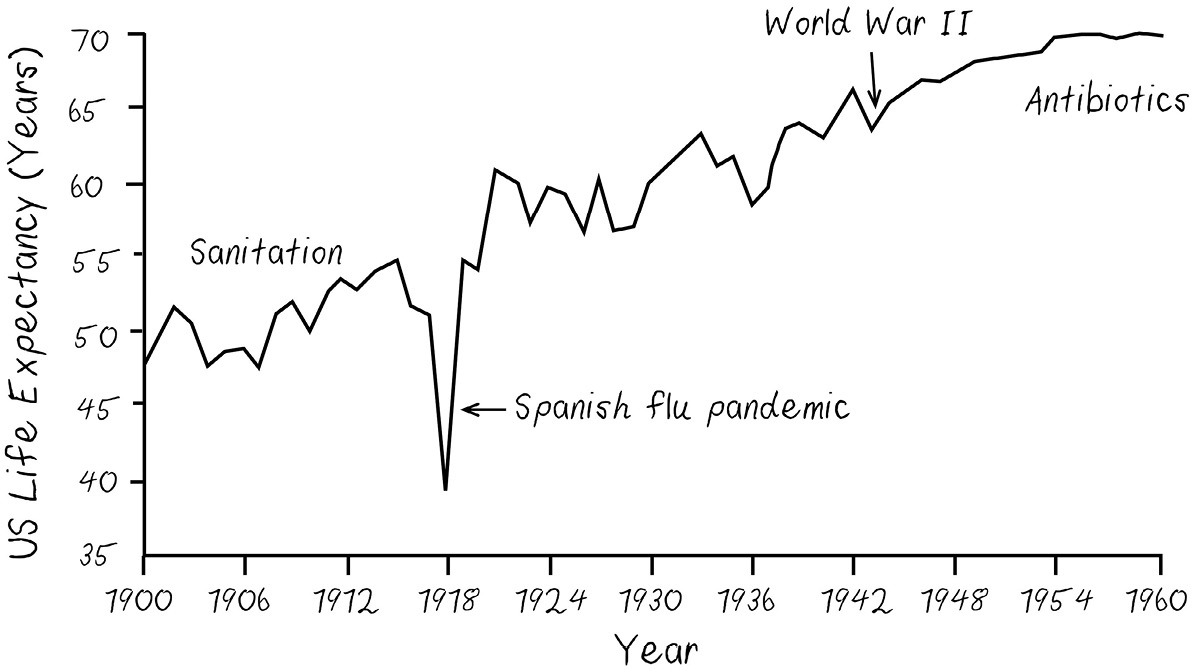
CHANGE IN LIFE EXPECTANCY DURING THE 1918 FLU EPIDEMIC.
Source: S. L. Knobler, A. Mack, A. Mahmoud, and S. M. Lemon, eds., The Threat of Pandemic
In uenza: Are We Ready? Workshop Summary, Institute of Medicine (Washington, DC:
National Academies Press, 2005), https://doi.org/10.17226/11150, PMID: 20669448.
Thanks to wearables, we already have the technology in place to monitor the
body temperature, pulse, and other biometric reactions of more than a hundred
mil ion people in real time. The only things separating us from doing so are a
recognized need and a cultural response.
The need is already here. It has been for quite some time. It took about
twenty years for the deadly mosquito-transmitted Zika virus to spread from
Central Africa, where it was rst documented, to South Asia and about forty-
ve more years to reach French Polynesia in the Central Paci c in 2013. In the
span of those sixty- ve years, it a ected just a smal part of the world. In the next
four years, though—four years, that’s al —the virus spread like wild re across
South America, through Central America, into North America, and back across
the Atlantic Ocean to Europe.
Zika, at least, is somewhat limited in the way it can be spread—mostly
through mosquito bites but also from mother to child and from sexual partner
to sexual partner. It cannot, as far as we know, be transmitted by doorknobs, via
food, or in the air-recirculating climate control systems on airplanes.
But in uenza can, as can other, potential y deadlier viruses.
On March 23, 2014, the World Health Organization reported cases of Ebola
virus disease in the forested rural region of southeastern Guinea, and from there
it spread rapidly to three neighboring countries, causing widespread panic. Even
the richest country in the world, where eleven people were treated for Ebola, was
caught without a uni ed plan.
That October, people in hazmat suits boarded American Airlines ight 45when it landed in New Jersey to shine infrared heat detectors on people’s
foreheads in an attempt to detect a fever. Kaci Hickox, who worked for Doctors
Without Borders, later won a lawsuit that led to a “quarantine bil of rights,”
after she was placed in Governor Chris Christie’s “private prison.” On that
occasion, and a few since then, the deadly virus has been contained, but
humanity may not always be so lucky.
“Whether it occurs by a quirk of nature or at the hand of a terrorist,
epidemiologists say a fast-moving airborne pathogen could kil more than 30
mil ion people in less than a year,” Bil Gates told a crowd at the Munich
Security Conference in 2017, “and they say there is a reasonable probability the
world wil experience such an outbreak in the next 10–15 years. ”31
If that happens, 30 mil ion could be a very conservative estimate.
As our transportation networks continue to expand in reach and speed, as
more people travel to more corners of our world faster than our ancestors could
possibly have imagined, pathogens of al sorts are traveling faster than ever, too.
But with the right data in the right hands, we can move faster, especial y if we
combine mass “biocloud” data with superfast DNA sequencing to detect
pathogens as they spread through cities and along transportation corridors. In
doing so, we can get ahead of a kil er pathogen with emergency travel restrictions
and medical resources. In this ght, every minute wil matter. And every minute
that passes without action wil be measured in human lives.
Not everyone is ready for the biotracking world. That makes sense. To many,
clearly, it wil feel like a step too far. Maybe several steps too far.
In order to get to a world in which hundreds of mil ions of humans—al
being tracked in real time for hormone levels, chemicals, body temperature, and
heart rate—are standing as sentinels to warn us of public health crises as they
happen, someone is going to have to have the data. Who wil that be? One
government? A coalition of governments? Any and every government?
Maybe a computer company. Or maybe a pharmaceutical manufacturer. Or
an internet shopping company. Or an insurer. Or a pharmacy. Or a supplement
company. Or a hospital network.
Most likely, it wil be a combination of these companies, al under one roof.Consolidation has already started and wil continue as these companies set their
sights on the largest and fastest-growing sector of the global economy, health
care, which now exceeds 10 percent of global GNP and is increasing at an annual
rate of 4.1 percent.
Whom do you trust to know your every move? To listen to your every
heartbeat? To see you when you’re sleeping and know when you’re awake, like a
certain benevolent mythical being of wintertime lore? To be able to identify,
through the data, when you are feeling sad, driving too fast, having sex, or had
too much to drink?
There’s no sense in trying to convince people that there is nothing to worry
about. Of course there are things to worry about. Think having your credit card
data stolen is bad? That’s nothing. You can always cal the bank and get a new
credit card, but your medical records are permanent—and far more personal.
More than 110 mil ion medical records were breached in the United States
between 2010 and 2018.32 Jean-Frédéric Karcher, the head of security at
Maintel, a UK communications provider, predicts that attacks wil become far
more common.
“Medical information can be worth ten times more than credit card numbers
on the deep web. Fraudsters can use this data to create fake IDs to buy medical
equipment or drugs,” he has warned. 33
We already trade a tremendous amount of privacy for technological services.
We do it al the time. We do it every time we start a bank account or sign up for a
credit card. We do it often when we point our internet browsers to a new web
page. We do it when we sign up for school. We do it when we get onto an
airplane. And we do it—a lot—when we use our mobile phones. Have these
been good trade-o s for everyone? That’s a matter of personal opinion, of
course. But when most people imagine not being able to use a credit card, surf
the web, sign up for school, travel by air, or use their phones and smart watches,
they quickly conclude that the trade is tolerable.
Wil people trade a little more privacy to stop a global disease pandemic?
Sadly, probably not. The tragedy of the commons is that humans are not very
good at taking personal action to solve col ective problems. The trick to
revolutionary change is nding ways to make self-interest align with thecommon good. For people to accept widespread biometric tracking in a way that
could help us get ahead of fast-moving deadly viruses, they’l need to be o ered
something they have a hard time seeing themselves without.
How to get ready for this world is a conversation that needs to be had. And
soon.
I’m there already. Before I began having my biomarkers checked on a regular
basis, I did worry about what the ever-changing chemical signals in my body
could disclose about me to someone with access to my data. Al the data are held
on health care– or HIPAA-compliant servers, and the data are encrypted. But
there’s always the fear that the data wil be hacked. There’s always a way.
But after I began, the information I received was worth far more than the
concerns I carry. It’s a personal choice, no question. Now, having seen the
changes on my dashboard, I cannot imagine living without it. Just as I now
wonder how I ever managed to drive without a GPS, I wonder how I ever made
decisions about what I should be eating and how much I should be exercising
before I received regular updates from my biosensor ring and blood biomarker
reports. Indeed, I am eager for the day in which the data about my health are
processed in real time. And if that can help protect others, al the better.
MOVING FASTER
While I was doing my PhD, I had a night job. For about eight dol ars an hour, I
tested body uids—urine, feces, spinal uid, blood, and genital swabs horribly
twisted in hair—for the presence of deadly bacteria, parasites, and fungi. It was
glamorous work.
At my disposal, I had al the trappings of nineteenth-century technology:
microscopes, petri dishes, sterile water. A lab technician transported from 1895
to that 1980s microbiology lab would have felt right at home. Today, this is stil
how many microbiology labs operate.
Making life-and-death cal s this way was frustrating. In every other branch of
medicine, we’ve made enormous strides technological y with robotics,
nanotechnology, scanners, and spectrometers.These days, though, I’m no longer frustrated. I’m furious.
Antibiotic-resistant strains of bacteria continue to spread, and new studies
implicate bacteria as causal agents in cancer, heart disease, and Alzheimer’s
disease. 34
But I wasn’t working to solve this problem, until recently. A brush with
Lyme disease has a way of intensifying a person’s feelings about these sorts of
things.
Our daughter Natalie was 11 years old when it happened. In New England,
where we live, there is an epidemic of ticks that carry the bacterial spirochete
Borrelia burgdorferi, which causes Lyme disease. Recent estimates suggest that
approximately 300,000 people in the United States may contract the disease each
year. Left untreated, Borrelia hides out in skin cel s and lymph nodes, causing
facial paralysis, heart problems, nerve pain, memory loss, and arthritis. It hides in
a protective bio lm, making it extremely di cult to kil .
Natalie never had a red ring of skin around a tick bite—a sure sign you’ve
contracted the parasite. She had been complaining about a headache and sore
back, typical signs of u. But quickly it became clear that this wasn’t u—it was
something much worse.
She was unable to turn her head. She was losing her eyesight. She was
terri ed. My wife and I were, too—we’d never felt so helpless in our lives. We
began searching online for answers. Potential diseases included leukemia and a
viral infection of the brain.
Doctors at Boston’s Children’s Hospital began poring over her. The rst test
lit up Lyme disease proteins, but the insurance company needed con rmation
because the rst test occasional y gives a false positive. The second test failed,
putting the course of treatment into limbo pending more lab results.
I asked for a microliter sample of Natalie’s spinal uid to test. My lab was
across the street, and I could sequence the DNA of the pathogen. The hospital
refused.
Given the state of her symptoms at that point, I’ve since learned, she had a 50
percent chance of survival. Her life came down to a coin ip. At a time when
every second counted, doctors were waiting on lab results.
It took three days to con rm that it was a Lyme disease infection, and nal ythe doctors gave Natalie intravenous antibiotics directly into the large vein next
to her heart. She received that treatment every day for nearly a month.
She is okay now, but it was clear to everyone involved, especial y Natalie, that
we desperately need to be applying twenty- rst-century technologies to
diagnosing infectious diseases. In Cambridge, Massachusetts, and Menlo Park,
California, I’ve helped gather a group of very smart folks—infectious disease
doctors, microbiologists, geneticists, mathematicians, and software engineers—
to develop tests that can rapidly and unambiguously tel physicians what an
infection is and how best to kil it, using “high-throughput sequencing.”
The rst step in this process is the extraction of nucleic acids from blood
samples, saliva, feces, or spinal uid. Because it adds cost and reduces sensitivity,
the patient’s DNA is removed using innovative methods honed by the same
scientists who extract ancient DNA from mummies—one of countless cases of
one eld of science bene ting another. Next, the samples are processed through
agnostic DNA-sequencing technologies, meaning that the system is not looking
for any one speci c infectious agent but rather reading the genomes in the entire
sample. That list is then scanned against a database of al known human
pathogens at the strain level. The computer spits out a highly detailed report
about what invaders are present and how best to kil them. The tests are as
accurate as the standard ones, but they provide strain-level information and are
pathogen agnostic. In other words, soon doctors won’t have to guess what to
look for when ordering a test or what treatment wil work best. They wil know.
Just a few years ago, this wouldn’t just have been a slow process, it wouldn’t
even have been possible. Now it can be done in days. Soon it wil be able to be
done in hours and eventual y minutes.
But there’s another way to deal with such diseases: we could prevent them
altogether.
THE NEW AGE OF INOCULATION
There is no rational debate over the immensely positive impact of vaccines onlife expectancy and healthy lifespans over the past century. Childhood mortality
around the world has plummeted, in no smal part because we’ve wiped out
diseases such as smal pox. The number of healthy children in the world has risen
because we’ve destroyed polio. The number of healthy adults has, too. Within
fty years, postpolio syndrome, which causes fatigue, muscle weakness,
abnormal spinal curvature, and speech defects in adults, wil be extinct.
And, of course, the more diseases we can vaccinate for, especial y those that
claim elderly people’s lives, such as u and pneumonia, the more life expectancy
wil rise in the coming years.
When we inoculate the herd, it doesn’t just protect us individual y, it protects
the weakest among us: the young and the old. Chickenpox once claimed
thousands of lives each year around the world—mostly among the very young
and the very old—and accounted for hundreds of thousands of hospitalizations
and mil ions of days of missed work. Those days are over.
A shining example of the power of vaccines to extend lifespan came in the
years after the introduction of vaccines for Streptococcus pneumoniae, a major
source of il ness in older persons and the most common cause of death by
respiratory infection. After the Prevnar vaccine for infants was launched in
2000, hospitalization and deaths from pneumonia fel across the board,
according to a study published in the New England Journal of Medicine.
“The protective e ect we saw in older adults, who do not receive the vaccine
but bene t from vaccination of infants, is quite remarkable,” the study’s rst
author, Marie Gri n, explained. “It is one of the most dramatic examples of
indirect protection, or herd immunity, we have seen in recent years. ”35
In the rst three years alone, deaths from pneumonia were halved, averting
more than 30,000 cases and 3,000 deaths in the United States alone, according
to another study. 36
We can snu out a lot of kil ers with vaccines like this.
Yet for several decades, the promise of vaccines to improve the lives of bil ions
of people around the world has been slowed, not only by a distrust of vaccines
promulgated by debunked science, but by plain old market forces. The golden
era of vaccine research was in the mid–twentieth century, a time that saw the
quick development of a succession of exceptional y e ective inoculations againstwhooping cough, polio, mumps, measles, rubel a, and meningitis.
But by the latter part of the century, the business model that long sustained
vaccine research and development was badly broken. The cost of testing new
vaccines had risen exponential y, thanks in large part to increasing public
concerns about safety and risk-averse regulatory bodies. The “low-hanging fruit”
of the inoculation world had already been picked. Now a simple vaccine can take
more than a decade to produce and cost more than half a bil ion dol ars, and
there is stil the chance it won’t be approved for sale. Even some vaccines that
have worked wel and been critical for the prevention of epidemics, such as
GlaxoSmithKline’s Lyme disease vaccine, have been taken o the market because
the unfounded backlash against vaccines made continuation of the product “just
not worth it.” 37
Governments don’t make vaccines; companies do. So when the market forces
are not conducive, we don’t get the medicines we so badly need. Funding gaps
are sometimes made up by charitable organizations, but not nearly enough. And
economic downturns such as the global recession of the late 2000s and early
2010s left foundations—many of which base their giving on market-tied
endowment earnings—unable or unwil ing to invest as much in these lifesaving
interventions.38
The good news is that we are experiencing a minirenaissance in vaccine
research and development, which has tripled between 2005 and 2015, now
accounting for about a quarter of al biotechnology products being developed. 39
The big one is malaria, infecting 219 mil ion people and claiming 435,000
people in 2017. 40 Thanks to Bil and Melinda Gates, GlaxoSmithKline, and
Program for Appropriate Technology in Health (PATH), a partial y e ective
vaccine against malaria known as Mosquirix was deployed for the rst time in
2017, giving hope that the malaria parasite wil one day be pushed to
extinction. 41
We are also learning how to quickly grow vaccines in human cel s, mosquito
cel s, and bacteria, avoiding the time and expense of infecting the mil ions of
fertilized chicken eggs we currently use, a remarkably antiquated process. One
Boston-based research consortium was able to get a vaccine for Lassa fever, a
disease similar to Ebola, al the way to the animal-testing stage in just fourmonths and for about $1 mil ion, cutting many years and many mil ions of
dol ars from the usual process. 42 That’s nothing short of astounding.
At this moment, researchers are starting the nal sprint toward the end of a
very long race to develop vaccines that wil inoculate us against diseases that are
so ubiquitous that we simply accept them as part of life. Many thought leaders
predict, though with some trepidation, that it won’t be long before we’re no
longer throwing Hail Mary passes such as the annual in uenza vaccine, which in
some years protects less than a third of its recipients, which is stil far better than
nothing. (If you don’t get u vaccines or vaccinate your kids, please do. We are
privileged to live in an age in which we can protect ourselves and our children
from potential y deadly diseases.)
The ability to quickly detect, diagnose, treat, and even prevent diseases that
aren’t related to aging but that claim many mil ions of lives each year wil al ow
us to continue to push our average life expectancy higher and higher, closing the
gap between the mean and the maximum.
Even then, organs wil fail and body parts wil wear out. What wil we do
when al other technologies fail? There’s a revolution happening there, too.
THE ORGAN GRIND
The Great Ocean Road, which runs along the Australian coast west of
Melbourne, is among the most beautiful stretches of highway in the world. But
whenever I’m on it, I can’t help but remember one of the most frightening days
of my life—the day I got a cal tel ing me that my brother, Nick, had been in a
motorcycle accident.
He was 23 years old at the time and touring the country by motorbike. He
was an expert rider, but he hit an oil patch, ew from his bike, and slid under a
metal barrier that crushed his ribs and ruptured his spleen.
Fortunately, he pul ed through, but to save his life, the emergency room
doctors had to remove his spleen, which is involved in the production of blood
cel s and is an important part of the immune system. For the rest of his life, he
has to be careful not to get a major infection, and he certainly seems to get sickermore often and take longer to get better. People without a spleen are also at
higher risk of dying of pneumonia later in life.
It doesn’t take age or disease to do a number on our organs. Sometimes life
does that to us in other ways, and we’re lucky if it’s just our spleen that we lose.
Hearts, livers, kidneys, and lungs are a lot harder to live without.
The same kind of cel ular reprogramming we can use to restore optic nerves
and eyesight may one day o er us the ability to restore function in damaged
organs. But what can we do for organs that have completely failed or need to be
removed because of a tumor?
Right now, there’s only one way to e ectively replace damaged and diseased
organs. It’s a morbid truth, but it’s a truth nonetheless: when people pray for an
organ to become available for a loved one who needs one, part of what they’re
praying for is a deadly car accident.
There’s a lot of irony, or some would say logic, in the fact that the
Department of Motor Vehicles is the organization that asks people whether they
want to be organ donors: each year in the United States alone, more than 35,000
people are kil ed in motor vehicle accidents, making this mode of death one of
the most reliable sources of tissues and organs. If you haven’t signed up to be an
organ donor, I hope you consider it. Between 1988 and 2006, the number of
people waiting for a new organ grew sixfold. As I write this sentence, there are
114,271 people on the US online registry waiting for organ transplants, and
every ten minutes, someone new is added to the transplant waitlist.43
It’s even worse for patients in Japan, where the ability to get an organ
transplant remains far below those of Western countries. The reasons are both
cultural and legal. In 1968, the Buddhist belief that the body should not be
divided after death fueled an emotion-laden restorm in the media about
whether the rst Japanese heart donor had truly been “brain dead” when the
heart was removed by Dr. Juro Wada. A strict law was immediately enacted that
banned the removal of organs from a cadaver until the heart had stopped
beating. The law was relaxed thirty years later, but the Japanese remain divided
on the issue and good organs remain hard to come by.
My brother also su ers from an eye disease cal ed keratoconus, which causedthe corneas covering his lenses to wrinkle like a nger pushed into plastic wrap.
To treat this, he had two separate corneal transplant surgeries, one in his
twenties, the other in his thirties, that swapped two other people’s corneas for
his. Both times, he su ered through six months of corneal stitches that felt like
“branches” in his eyes, but his vision was saved. The fact that Nick now literal y
sees the world through others’ eyes is an amusing topic for dinner conversation
that belies the true depth of our family’s gratefulness to his deceased donors.
Now, as we rapidly approach the era of self-driving cars—a technological and
social paradigm shift that almost every expert expects wil rapidly reduce car
crashes—we need to confront an important question: Where wil the organs
come from?
The geneticist Luhan Yang and her former mentor Professor George Church
in my department at Harvard Medical School had just discovered how to gene
edit mammalian cel s when they began working to edit out genes in pigs. To
what end? They envisioned a world in which pig farmers raise animals
speci cal y designed to produce organs for the mil ions of people who are on
transplant waiting lists. Though scientists have had dreams of widespread
“xenotransplantation” for many decades, Yang took one of the biggest steps
toward that goal when she and her col eagues demonstrated that they could use
gene editing to eliminate dozens of retroviral genes from pigs that currently
prevent them from donating organs. That’s not the only obstacle to
xenotransplantation, but it’s a big one—and one that Yang gured out how to
overcome before her 32nd birthday.
That’s not the only way we’l be getting organs in the future. Ever since
researchers discovered in the early 2000s that they could modify inkjet printers
to lay down 3D layers of living cel s, scientists around the world have been
working toward the goal of printing living tissue. Today scientists have
implanted printed ovaries into mice and spliced printed arteries into monkeys.
Others are working on printing skeletal tissue to x broken bones. And printed
skin is likely to start being used for grafts in the next few years, with livers and
kidneys coming soon after that and hearts—which are a bit more complicated—
a few years behind.
Soon it won’t matter if the morbid pipeline for human organ transplantationends. That pipeline never met the demand anyway. In the future, when we need
body parts, we might very wel print them, perhaps by using our own stem cel s,
which wil be harvested and stored for just such an occasion, or even using
reprogrammed cel s taken from blood or a mouth swab. And because there
won’t be competition for these organs, we won’t have to wait for things to go
catastrophical y wrong for someone else to get one—we’l only have to wait for
the printer to do its job.
JUST IMAGINE
Is al this hard to imagine? That’s understandable. We’ve spent a long time
building up our expectations of what medical care should look like—and indeed
what human life should look like. For a lot of people, it’s simply easier to say, “I
just don’t believe that wil happen,” and leave things at that.
But we’re actual y quite a bit better at changing our minds about what we
expect out of life, and what age actual y means, than many of us think we are.
Consider Tom Cruise. As the Top Gun actor entered his late 50s, with
bulging muscles and a straight line of dark hair sprouting from a minimal y
wrinkled forehead, he was stil at work. Not just acting, but doing the sort of
acting that has long been the purview of much younger actors. He was stil
doing many of his own dangerous stunts, too: riding motorcycles at high speed
through al eys, being strapped to the outside of a plane as it takes o , hanging o
the top of the world’s tal est building, skydiving from the upper reaches of the
atmosphere.
How easily do the words “Fifty is the new thirty” slide from our lips these
days? We forget what we used to expect life past 50 to look like, not hundreds of
years in the past but just a few decades ago.
It didn’t look like Tom Cruise jumping out of airplanes. It looked like
Wilford Brimley. In the 1980s, Brimley was one of Cruise’s costars in the movie
The Firm. Cruise was 39 and Brimley 58, already a gray-haired old man with a
walrus mustache.
A few years earlier, Brimley had starred in Cocoon, a movie about a group ofsenior citizens who stumble upon an alien “fountain of youth” that gives them
the energy—although not the looks—of their youth. The image of old folks
running around like teenagers was played to great comedic e ect.
It was audacious to think of someone that age acting so youthful. At the time
the movie was released, though, Brimley was ve or six years younger than
Cruise is now. According to Ian Crouch of the New Yorker, Cruise has easily
blasted through what he cal s “the Brimley Barrier.” 44
Barriers fal . And they wil fal again. In another generation, we’l be wel
accustomed to seeing movie stars in their 60s and 70s riding motorcycles at high
speeds, jumping from great heights, and delivering kung fu kicks high into the
air. Because 60 wil be the new 40. Then 70 wil be the new 40. And on it wil
go.When wil this happen? It’s already happening. It is not fanciful to say that if
you are reading these words, you are likely to bene t from this revolution; you
wil look younger, act younger, and be younger—both physical y and mental y.
You wil live longer, and those extra years wil be better.
Yes, it is true that any one technology might lead to a dead end. But there is
simply no way that al of them wil fail. Taken separately, any of these
innovations in pharmaceuticals, precision medicine, emergency care, and public
health would save lives, providing extra years that would otherwise have been
lost. When we take them together, though, we are staring up the road at decades
of longer, healthier life.
Each new discovery creates new potential. Each minute saved in the quest for
faster and more accurate gene sequencing can help save lives. Even if it doesn’t
move the needle much on the maximum number of years we live, this age of
innovation wil ensure that we stay much healthier much longer.
Not many of us much of the time, but al of us.
PART IIIWHERE WE’RE GOING
(THE FUTURE)
EIGHTTHE SHAPE OF THINGS TO COME
LET’S DO A LITTLE MATH.
And let’s make it conservative math. Let’s assume that each of these vastly
di erent technologies emerging over the next fty years independently
contributes to a longer, healthier lifespan.
DNA monitoring wil soon be alerting doctors to diseases long before they
become acute. We wil identify and begin to ght cancer years earlier. If you have
an infection, it wil be diagnosed within minutes. If your heartbeat is irregular,
your car seat wil let you know. A breath analyzer wil detect an immune disease
beginning to develop. Keystrokes on the keyboard wil signal early Parkinson’s
disease or multiple sclerosis. Doctors wil have far more information about their
patients—and they wil have access to it long before patients arrive at a clinic or
hospital. Medical errors and misdiagnoses wil be slashed. The result of any one
of these innovations could be decades of prolonged healthy life.
Let’s say, though, that al of these developments together wil give us a decade.
Once people begin to accept that aging is not an inevitable part of life, wil
they take better care of themselves? I certainly have. So, too, it seems, have most
of my friends and family members. Even as we have al stepped forward to be
early adopters of biomedical and technological interventions that reduce the
noise in our epigenomes and keep watch over the biochemical systems that keep
us alive and healthy, I’ve noticed a de nite tendency to eat fewer calories, reduce
animal-based aminos, engage in more exercise, and stoke the development ofbrown fat by embracing a life outside the thermoneutral zone.
These are remedies available to most people regardless of socioeconomic
status, and the impact on vitality has been exceptional y wel studied. Ten
additional healthy years is not an unreasonable expectation for people who eat
wel and stay active. But let’s cut that by half. Let’s cal it ve.
That’s fteen years.
Molecules that bolster our survival circuit, putting our longevity genes to
work, have o ered between 10 and 40 percent more healthy years in animal
studies. But let’s go with 10 percent, which gives us another eight years.
That’s twenty-three years total.
How long wil it be before we are able to reset our epigenome, either with
molecules we ingest or by genetical y modifying our bodies, as my student now
does in mice? How long until we can destroy senescent cel s, either by drugs or
outright vaccination? How long until we can replace parts of organs, grow entire
ones in genetical y altered farm animals, or create them in a 3D printer? A
couple of decades, perhaps. Maybe three. One or al of those innovations is
coming wel within the ever-increasing lifespans of most of us, though. And
when that happens, how many more years wil we get? The maximum potential
could be centuries, but let’s say it’s only ten years.
That’s thirty-three years.
At the moment, life expectancy in the developed world is a tad over 80 years.
Add 33 to that.
That’s 113 years, a conservative estimate of life expectancy in the future, as
long as most people come along for the ride. And recal that this number means
that over half the population wil exceed that number. It’s true that not al of
these advances wil be additive, and not everyone wil eat wel and exercise. But
also consider that the longer we live, the greater chance we have of bene ting
from radical medical advances that we cannot foresee. And the advances we’ve
already made are not going away.
That’s why, as we move faster and faster toward a Star Trek world, for every
month you manage to stay alive, you gain another week of life. Forty years from
now, it could be another two weeks. Eighty years from now, another three.
Things could get real y interesting around the end of the century if, for everymonth you are alive, you live another four weeks.
This is why I say that Jeanne Calment, who may have had the longest lifespan
of any person on our planet, wil eventual y fal o the list of the top ten oldest
humans in history. And it won’t be more than a few decades after that that she
wil leave the top 100. After that she wil leave the top mil ion. Imagine if people
who have lived beyond 110 had had access to al these technologies. Could they
have made it to 120 or 130? Perhaps.
Fel ow scientists often warn me not to be so publicly optimistic. “It’s not a
good look,” one wel -meaning col eague recently told me.
“Why?” I asked.
“Because the public isn’t ready for these numbers.”
I disagree.
Ten years ago, I was a pariah to many of my col eagues for even talking about
making medicines to help patients. One scientist told me that our job as
researchers is to “just show a molecule extends lifespan in mice, and the public
wil take it from there.” Sadly, I wish that were true.
Today, many of my col eagues are just as optimistic as I am, even if they don’t
admit it publicly. I’d wager that about a third of them take metformin or an
NAD booster. A few of them even take low doses of rapamycin intermittently.
International conferences speci cal y about longevity interventions are now held
every few weeks, the participants not charlatans but renowned scientists from
the world’s most prestigious universities and research centers. In these gatherings
it is no longer unusual to hear chatter about how raising the average human
lifespan by a decade, if not more, wil change our world. Mind you, the debate is
not about whether this wil happen; it is about what we should do when it
happens.
The same is increasingly true among the political, business, and religious
leaders with whom I spend more and more of my time these days, talking not
just about new technologies but about their implications. Slowly but surely,
these individuals—legislators, heads of state, CEOs, and thought leaders—are
coming to recognize the world-changing potential of the work being done in the
eld of aging, and they want to be ready.
Al these people might be wrong. I might be wrong. But I expect to bearound long enough to know one way or the other.
If I am wrong, it might be that I was too conservative in my predictions.
Though there are many examples of false predictions—who can forget nuclear-
powered vacuum cleaners and ying cars?—it is far more common for people
not to see something coming. Al of us are guilty of it. We extrapolate linearly.
More people, more horses, more horse manure. More cars, more air pol ution,
always more climate change. But that’s not how it works.
When technologies go exponential, even experts can be blindsided. The
American physicist Albert Michelson, who won a Nobel Prize for measuring the
speed of light, gave a speech at the University of Chicago in 1894, declaring that
there was probably little else to discover in physics besides additional decimal
places.1 He died in 1931, as quantum mechanics was in ful swing. And in his
1995 book, The Road Ahead, Bil Gates made no mention of the internet,
though he substantial y revised it about a year later, humbly admitting that he
had “vastly underestimated how important and how quickly” the internet
would come to prominence. 2
Kevin Kel y, the founding editor of Wired magazine, who has a better track
record than most at predicting the future, has a golden rule: “Embrace things
rather than try and ght them. Work with things rather than try and run from
them or prohibit them. ”3
We often fail to acknowledge that knowledge is multiplicative and
technologies are synergistic. Humankind is far more innovative than we give it
credit for. Over the past two centuries, generation after generation has witnessed
the sudden appearance of new and strange technologies: steam engines, metal
ships, horseless carriages, skyscrapers, planes, personal computers, the internet,
at-screen TVs, mobile devices, and gene-edited babies. At rst we are shocked;
then we barely notice. When the human brain was evolving, the only things to
change in a lifetime were the seasons. It should come as no surprise that we nd
it hard to predict what wil happen when mil ions of people work on complex
technologies that suddenly merge.
No matter if I’m right or wrong about the pace of change, barring a war or an
epidemic, our lifespan wil continue to rise. And the more thought leaders I
speak to around the globe, the more I realize how vast the implications are. Andyes, some of those people have al owed me to think and plan for events wel
beyond the initial scope of my research. But the people who push me to think
even harder are the younger people I teach at Harvard and other universities, and
the often even younger people I hear from via email and social media nearly
every day. They push me to think about how my work wil impact the future
workforce, global health care, and the very fabric of our moral universe—and to
better understand the changes that must take place if we are to meet a world of
signi cantly prolonged human healthspans and lifespans with equity, equality,
and human decency.
If the medical revolution happens and we continue on the linear path we’re
already on, some estimates suggest half of al children born in Japan today wil
live past 107. 4 In the United States the age is 104. Many researchers believe that
those estimates are overly generous, but I don’t. They might be conservative. I
have long said that if even a few of the therapies and treatments that are most
promising come to fruition, it is not an unreasonable expectation for anyone
who is alive and healthy today to reach 100 in good health—active and engaged
at levels we’d expect of healthy 50-year-olds today. One hundred twenty is our
known potential, but there is no reason to think that it needs to be for the
outliers. And I am on record as saying, in part to make a statement and in part
because I have a front-row seat on what’s around the corner, that we could be
living with the world’s rst sesquicentenarian. If cel ular reprogramming reaches
its potential, by century’s end 150 may not be out of reach.
At the moment I write this, there is no one on our planet—no one whose age
can be veri ed, at least—who is over the age of 120. So it wil be several decades,
at least, before we know if I’m right about this, and it could take 150 years before
someone steps over that threshold.
But as for the next century? And the next? It is not at al extravagant to expect
that someday living to 150 wil be standard. And if the Information Theory of
Aging is sound, there may be no upward limit; we could potential y reset the
epigenome in perpetuity.
This is terrifying to a lot of people—and understandably so. We’re on the
cusp of upending nearly every idea we’ve ever had about what it means to be
human. And that has a lot of people saying not just that it can’t be done but thatit shouldn’t be done—for it wil surely lead to our doom.
The critics of my life’s work aren’t nameless, faceless social media trol s.
Sometimes they are my col eagues. Sometimes they are close personal friends.
And sometimes they are my own esh and blood.
Our oldest child, Alex, who at 16 hopes for a career in politics and social
justice, has often struggled to see the future with the same optimism I do.
Especial y when you’re young, it is hard to see much of an arc to the moral
universe, let alone one that bends toward justice.5
Alex grew up, after al , in a world that is quickly and disastrously warming; in
a nation that has been at war for the better part of two decades; and in a city that
su ered a terrorist attack on the people participating in one of its most cherished
traditions, the Boston Marathon. And like so many other young people, Alex
lives in a hyperconnected universe where news of one humanitarian crisis after
the next, from Syria to South Sudan, is never far from the screen of a
smartphone.
So I understand. Or I try to, at least. But it was disappointing to learn, one
recent night, that Alex didn’t share the optimism I’ve always had about the
future. Of course I’m proud that our kid has such a strong moral compass, but it
was saddening to realize this more pessimistic view of the world casts a
signi cant shadow over the way Alex sees my life’s work.
“Your generation, just like al the ones that came before, didn’t do anything
about the destruction that is being done to this planet,” Alex told me that
evening. “And now you want to help people live longer? So they can do even
more damage to the world?”
I went to bed that night troubled. Not by our rstborn’s denouncement of
me; of that, I admit, I was a little proud. We’l never destroy the global patriarchy
if our children don’t rst practice on their fathers. No, what I was troubled by—
what kept me up that night and has done so many since—were the questions
that I simply could not answer.
Most people, upon coming to the realization that longer human lives are
imminent, also quickly recognize that such a transition cannot possibly occur
without signi cant social, political, and economic change. And they are right;
there can be no evolution without disruption. So what if the way I see the futureisn’t at al what we’re headed toward? What if giving bil ions of people longer
and healthier lives enables our species to do greater harm to this planet and to
one another? Greater longevity is inevitable; I’m sure of it. What if it inevitably
leads to our self-destruction?
What if what I do makes the world worse?
There are plenty of people out there—some of them very smart and very
informed—who think that’s the case. But I’m stil optimistic about our shared
future. I don’t agree with the naysayers. But that doesn’t mean I do not listen to
them. I do. And we al should. That’s why, in this chapter, I’m going to explain
some of their concerns—indeed, concerns I share in many cases—but I’l also
present a di erent way of thinking about our future.
You can take it from there.
THE HUNDRED YEARS’ WARNING
The number of Homo sapiens grew slowly over the rst few hundred thousand
years of our history, and at least on one occasion, we almost went extinct. While
there are many young skeletons from the Late Archaic and Paleolithic periods,
there is only a handful of skeletons of individuals over the age of 40. It was rare
for individuals to make it to the point we now have the luxury of cal ing middle
age. 6 Recal , this was a time when teenage girls were mothers and teenage boys
were warriors. Generations turned over quickly. Only the fastest, smartest,
strongest, and most resilient tended to survive. We rapidly evolved superior
bipedal and analytical skil s but at the expense of mil ions of brutal lives and
early deaths.
Our ancestors bred as fast as biology al owed, which was only slightly faster
than the death rate. But that was enough. Humanity endured and scattered to al
ends of the planet. It wasn’t until right around the time Christopher Columbus
rediscovered the New World that we reached the 500-mil ion-individuals mark,
but it took just three hundred more years for that population to double. And
today, with each new human life, our planet becomes more crowded, hurtling ustoward, or perhaps further beyond, what it can sustain.
How many is too many? One report, which examined sixty- ve di erent
scienti c projections, concluded that the most common estimated “carrying
capacity” of our planet is 8 bil ion.7 That’s just about where we’re at right now.
And barring a nuclear holocaust or a global pandemic of historical y deadly
proportions—nothing anyone in his or her right mind would ever wish for—
that’s not where our population is going to peak.
When the Pew Research Center pol ed members of the largest association of
scientists in the world, 82 percent said that there isn’t enough food and other
resources on this planet for its fast-growing human population.8 Among those
who held that opinion was Frank Fenner, an eminent Australian scientist who
helped bring an end to one of the world’s deadliest diseases as the chairman of
the Global Commission for the Certi cation of Smal pox Eradication. It was
Fenner, in fact, who had the distinct honor of announcing the eradication of the
disease to the World Health Organization’s governing body in 1980. Having
helped mil ions of people avoid a virus that kil ed nearly a third of those who
contracted the disease it caused, Fenner would have been justi ed in indulging in
a little exuberant optimism about the ways in which people can come together
to save themselves.
He had planned a quiet retirement. 9 But his mind wouldn’t stop working.
He couldn’t stop trying to identify and solve big problems. He spent the next
twenty years of his life writing about other threats to humankind, many of
which had been virtual y ignored by the same world health leaders who had
banded together to stop smal pox.
His nal act of forewarning came just a few months before his death in 2010,
when he told the Australian newspaper that the human population explosion
and “unbridled consumption” had already sealed our species’ fate. Humanity
would be gone in the next hundred years, he said. “There are too many people
here already.” 10
We’ve heard this song before, of course. At the turn of the nineteenth
century, as the global human population was screaming past the 1 bil ion mark,
the English scholar Thomas Malthus warned that advances in food production
inevitably led to population growth, placing increasing numbers of poor peopleat greater risk of starvation and disease. Viewed from the developed world, it
often looks as though a Malthusian catastrophe has largely been avoided;
agricultural advances have kept us one step ahead of disaster. Viewed global y,
though, Malthus’s warnings were little short of prophetic. About the same
number of people who lived on the planet in Malthus’s time go hungry in our
time. 11
In 1968, as the global population approached 3.5 bil ion, Stanford University
professor Paul Ehrlich and his wife, associate director of Stanford’s Center for
Conservation Biology Anne Ehrlich, sounded the Malthusian alarm once again
in a best-sel ing book cal ed The Population Bomb. When I was young, that book
had a rather prominent place on my father’s bookshelf—right at eye level for a
young boy. The cover was disturbing: a plump, smiling baby sitting inside a
bomb with a lit fuse. I had nightmares about that.
What was inside the cover was worse, though. In the book, Ehrlich described
his “awakening” to the horrors to come, a revelation he had during a cab ride in
New Delhi. “The streets seemed alive with people,” he wrote. “People eating,
people washing, people sleeping. People visiting, arguing, and screaming. People
thrusting their hands through the taxi window, begging. People defecating and
urinating. People clinging to buses. People herding animals. People, people,
people, people. ”12
With every new year, the Ehrlichs wrote, global food production “fal s a bit
further behind burgeoning population growth, and people go to bed a little
hungrier. While there are temporary or local reversals of this trend, it now seems
inevitable that it wil continue to its logical conclusion: mass starvation.” 13 It’s
horrifyingly clear, of course, that mil ions of people have indeed died of
starvation in the decades that have passed since The Population Bomb was rst
published, but not nearly at the levels the Ehrlichs predicted and not typical y
because of a lack of food production but rather as a result of political crises and
military con icts. When a child starves, though, it doesn’t much matter to them
or their family how it came to happen.
Though the direst of their predictions did not come to pass, in focusing so
intently on the food production–population relationship Malthus and the
Ehrlichs may actual y have underestimated the greater and longer-term risk—not mass starvation that might claim hundreds of mil ions of lives but a
planetary rebel ion that wil kil us al .
In November 2016, the late physicist Stephen Hawking predicted that
humanity had less than 1,000 years left on “our fragile planet.” A few months of
contemplation later, he revised his estimate downward by 90 percent. Echoing
Fenner’s warnings, Hawking believed that humanity would have 100 years to
nd a new place to live. “We are running out of space on Earth,” he said. A lot of
good that wil do; the Earth-like planet that is nearest to our solar system is 4.2
light-years away. Barring major advances in warp speed or wormhole-transit
technology, it would take us ten thousand years to get there.
The problem is not just population, it’s consumption. And it’s not just
consumption, it’s waste. In comes the food; out goes the e uent. In come the
fossil fuels; out go the carbon emissions. In come the petrochemicals; out goes
the plastic. On average, Americans consume more than three times the amount
of food they need to survive and about 250 times as much water.14 In return,
they produce 4.4 pounds of trash each day, recycling or composting only about
of a third of it.15 Thanks to things such as cars, planes, big homes, and power-
hungry clothes dryers,16 the annual carbon dioxide emissions of an average
American are ve times as high as the global average. Even the “ oor”—below
which even monks living in American monasteries typical y do not go—is twice
the global average. 17
It isn’t just that Americans consume and waste so much, it’s that hundreds of
mil ions of other people consume and waste as much and in some cases more,18
and bil ions of other people are moving in that same direction. If everyone in the
world consumed as Americans do for one year, the nonpro t Global Footprint
Network estimates it would take the Earth four years to regenerate what has
been used and absorb what has been wasted. 19 This is textbook unsustainability;
we use and use and use, and return little of value to our natural world.
The increasing number of scientists making hundred-year warnings has
formed around a terrifying environmental reality: even with “very stringent and
unrealistical y ambitious abatement strategies,” 20 we likely wil not be able to prevent global temperature changes that wil be greater than 2°C, a “tipping
point” that many scientists believe wil be catastrophic for humanity.21 Indeed, as Fenner said, it might truly be “too late.”We are not yet at that two-degree tipping point, and nonetheless the
consequences are already quite staggering. Human-caused climate change is
destroying food webs around the globe, and by some estimates, one in six species
is now at risk of extinction. Warming temperatures have “cooked the life out of
the corals” of our oceans, 22 including the Great Barrier Reef, which is roughly
the size of California and the most diverse ecosystem on our planet. More than
90 percent of that Australian natural wonder has su ered from bleaching,
meaning it is being starved of the algae it needs to survive. In 2018, the
Australian government released a report acknowledging what scientists had been
saying for many years: that the reef is headed toward “col apse.” 23 And in the
same year, Australian researchers said that global warming had claimed its rst
mammalian victim, a long-tailed marsupial mouse cal ed the Bramble Cay
melomys, which was sent into extinction when its island ecosystem was
destroyed by surging seawater.
There can also be no debate, at this point, that the melting of the Antarctic
and Greenland ice caps is driving a rise in sea levels, which the National Oceanic
and Atmospheric Association and others have warned wil worsen coastal
ooding in the coming years, threatening cities such as New York, Miami,
Philadelphia, Houston, Fort Lauderdale, Galveston, Boston, Rio de Janeiro,
Amsterdam, Mumbai, Osaka, Guangzhou, and Shanghai. A bil ion people or
more live in areas likely to be a ected by rising sea levels.24 Meanwhile, we’re facing more—and more severe—hurricanes, oods, and droughts; the World
Health Organization estimates that 150,000 people are already dying each year
as a direct result of climate change, and that number is likely to at least double in
coming years. 25
Al of these dire warnings are predicated upon a world in which humans live
for an average of about 75 or 80 years. Thus even the most pessimistic of
assertions about the future of our environment are actual y underestimating the
extent of the problem. There is simply no model in which more years of life does
not equate to more people and in which that does not lead to more crowding,
more environmental degradation, more consumption, and more waste. As welive longer, these environmental crises wil be exacerbated.
And that could be only part of our woes.
THE HUNDRED-YEAR POLITICIAN
If there has been a consistent driving force that has made our world a kinder,
more tolerant, more inclusive, and more just place, it is that humans don’t last
long. Social, legal, and scienti c revolutions, after al , are waged, as the
economist Paul Samuelson often noted, “one funeral at a time.”
The quantum physicist Max Planck also knew this to be true.
“A new scienti c truth does not triumph by convincing its opponents and
making them see the light,” Planck wrote shortly before his death in 1947, “but
rather because its opponents eventual y die, and a new generation grows up that
is familiar with it.” 26
Having witnessed a few di erent sorts of revolutions during my life—from
the fal of the Berlin Wal in Europe to the rise of LGBTQ rights in the United
States to the strengthening of national gun laws in Australia and New Zealand
—I can vouch for these insights. People can change their minds about things.
Compassion and common sense can move nations. And yes, the market of ideas
has certainly had an impact on the way we vote when it comes to issues such as
civil rights, animal rights, the ways we treat the sick and people with special
needs, and death with dignity. But it is the mortal attrition of those who
steadfastly hold on to old views that most permits new values to ourish in a
democratic world.
Death by death, the world sheds ideas that need to be shed. Ipso facto, birth
by birth, the world is o ered an opportunity to do things better. Alas, we don’t
always get it right. And it’s often a slow and uneven sort of progress. With a
generation time of twenty minutes, bacteria evolve rapidly to survive a new
chal enge. With a generation time of twenty years, human culture and ideas can
take decades to evolve. Sometimes they devolve.
In recent years, nationalism has moved from being the purview of angryfringe groups to being the force behind powerful political movements around
the world. There is no one single factor that can explain al of these movements,
but the economist Harun Onder is among those who have made a demographic
observation: nationalist arguments tend to resonate with older people.27
Therefore, it is likely that the antiglobalist wave wil be with us for some time to
come. “Virtual y every country in the world,” the United Nations reported in
2015, “is experiencing growth in the number and proportion of older persons in
their population.” Europe and North America already have the largest per capita
share of older persons; by 2030, according to the report, those over the age of 60
wil account for more than a quarter of the population on both of these
continents, and that proportion wil continue to grow for decades to come.
Once again, these are estimates based on ridiculously low projections for
lengthened lifespans. 28
Older constituencies support older politicians. As it is now, politicians seem
steadfastly opposed to stepping down in their 70s and 80s. More than half of the
US senators running for reelection in 2018 were 65 or older. Democratic leader
Nancy Pelosi was 78 that year. Dianne Feinstein and Chuck Grassley, two
powerful senators, were 85. On average, members of the US Congress are 20
years older than their constituents.
At the time of his death in 2003, Strom Thurmond was 100 years old and
had served 48 years as a US senator. That Thurmond was a centenarian in
Congress is no vice—we want our leaders to have experience and wisdom, as
long as they aren’t stuck in the past. The travesty was that Thurmond somehow
managed to keep his seat in spite of a long record of supporting segregation and
opposing civil rights, including basic voting rights. At the age of 99, he voted to
use military force in Iraq, opposed legislation to make pharmaceuticals more
a ordable, and helped kil a bil that would have added sexual orientation,
gender, and disability to a list of categories covered by hate crimes legislation.29
After his death, the “family values” politician was revealed to have had a
daughter with his family’s teenage African American housekeeper when he was
22, which was almost certainly an act of statutory rape under South Carolina
law. Though he knew about the child, he never publicly acknowledged her.30
Thurmond lived in retirement only six months; those who were too young tovote then wil have to live with the consequences of his votes for the rest of their
lives.We tend to tolerate a bit of bigotry among older people as a condition of the
“age in which they grew up,” but perhaps also because we know we won’t have
to live with it for long. Consider, though, a world in which people in their 60s
wil be voting not for another twenty or thirty years but for another sixty or
seventy. Imagine a man like Thurmond serving in Congress not for half a
century but for an entire century. Or, if it makes it easier to envision from your
place on the political spectrum, picture the politician you despise more than any
other holding power longer than any other leader in history. Now consider how
long despots in far less democratic nations wil cling to power—and what they
wil do with that power.
What wil this mean for our world political y? If a steadfast driving force for
kindness, tolerance, inclusivity, and justice suddenly ceases to exist, what wil our
world look like?
And the potential problems don’t stop there.
SOCIAL INSECURITY
Few people were spared the trauma in icted by the worldwide Great Depression
during the 1930s. But the impact was particularly felt by those in the last decades
of their lives. Stock market crashes and bank failures claimed the life savings of
mil ions of older Americans. With so many people out of work, the few
employers who were o ering jobs were reluctant to hire older workers.
Destitution was rampant. About half of the elderly were poor. 31
Those people had been deacons in churches, pil ars of communities, teachers
and farmers and factory workers. They were grandmothers and grandfathers,
and their desperation shook the nation to its core, prompting the United States
in 1935 to join about twenty other countries that had already instituted a social
insurance program.
Social Security made moral sense. It made mathematical sense, too. At thattime, just over half of men who reached their 21st birthday would also reach
their 65th, the year at which most could begin to col ect a supplemental income.
Those who reached age 65 could count on about thirteen more years of life.32
And there were a lot of younger workers paying into the system to support that
short retirement; at that time only about 7 percent of Americans were over the
age of 65. As the economy began to boom again in the wake of World War II,
there were forty-one workers paying into the system for every bene ciary. Those
are the numbers that supported the system when its rst bene ciary, a legal
secretary from Vermont named Ida May Ful er, began col ecting her checks.
Ful er had worked for three years under Social Security and paid $24.75 into the
system. She lived to the age of 100 and by the time of her death in 1975 had
col ected $22,888.92. At that point, the poverty rate among seniors had fal en to
15 percent, and it has continued to fal ever since, owing largely to social
insurance.33
Now about three-quarters of Americans who reach the age of 21 also see 65.
And changes to the laws that govern the US social insurance safety net have
prompted many to retire—and begin col ecting—earlier than that. New bene ts
have been added over the years. Of course, people are living longer, too;
individuals who make it to the age of 65 can count on about twenty more years
of life. 34 And as just about every social insurance doomsdayer can tel you, the
ratio of workers to bene ciaries is an unsustainable three to one.
That is not to say that Social Security is necessarily doomed. There are
reasonable adjustments that can be made to keep it solvent for decades to come.
But al of the most commonly recommended adjustments, as you might by now
suspect, are predicated on the assumption that we wil enjoy only modest gains
in lifespan in coming years. There are very few policy makers in the United States
—let alone the 170 other countries that now have some form of social insurance
program—who have so much as considered a world in which, at the age of 65,
many people wil be reaching the midpoint of their lives.
Even upon considering this, it can be assured that many politicians, if not the
overwhelming majority of them, wil choose to bury their heads in the sand.
Lyndon Johnson’s landslide victory over Barry Goldwater in the 1964 US
presidential race can largely be attributed to Goldwater’s perceived hostility tosocial insurance. But by the 1980s, politicians on both sides of the political aisle
had taken to cal ing Social Security the “third rail” of American politics: “Touch
it, you’re dead. ”35 At that time 15 percent of Americans were col ecting Social
Security. Today about 20 percent are.36 Today, people over the age of 65 make
up 20 percent of the voting population and wil grow by 60 percent by 2060,37
in addition to which they are about twice as likely as 18- to 29-year-olds to go to
the pol s. 38
There is a very rational argument for the resistance of the AARP (formerly
the American Association of Retired Persons) to any change to social insurance.
A few more years of waiting for retirement might not seem so bad to people who
work in occupations with low physical impact or in a job they love, but what of
those who have spent 45 years doing heavy manual labor, working on an
assembly line, or toiling in a meatpacking plant? Is it fair to expect them to work
even longer? Longevity drugs and healthspan therapies are very likely to help
those people feel better and stay healthier for longer, but that wouldn’t justify
forcing people who have worked arduously for most of their lives back to the
mines.
There are no easy answers, but if past is prologue—and it so often is with
human behavior—politicians wil watch this slow-moving disaster until it
becomes a fast-moving disaster; then they wil sit and watch some more. In many
nations, and particularly those of western Europe, social insurance programs are
relatively generous to bene ciaries and have been embraced by the political Left
and Right alike. These programs have become strained in recent years under the
weight of government de cits and the inability to meet long-held promises to
aging workers,39 prompting ghts over which entitlements are most sacred,
pitting education against health care and health care against pensions and
pensions against disability compensation. These ghts wil only increase as the
systems become further strained. And that strain is inevitable without
revolutionary reforms that account for the fact that the ranks of retirees wil
soon be brimming with those who, when the systems were designed in the mid-
1900s, were aged outliers.
At least every couple of months, I get a cal from a politician for an update onthe latest developments in biology, medicine, or defense. Almost always we end
up discussing what wil happen to the economy as people live longer and longer.
I tel him or her that there is simply no economic model for a world in which
people live forty years or more past the time of traditional retirement. We
literal y have no data whatsoever on the work patterns, retirement arrangements,
spending habits, health care needs, savings, and investments of large groups of
people who live, quite healthily, wel into their 100s.
Working with the world-renowned economists Andrew Scott at the
University of London and Martin El ison at Oxford University, we are
developing a model to predict what the future looks like. There are quite a few
variables, not al of them positive. Wil people continue to work? What jobs wil
they be able to get in a world in which the labor market wil already be being
upended by automation? Wil they spend a half century or more in retirement?
Some economists believe that economic growth is slowed when a country ages,
in part because people spend less in retirement. What wil happen if people
spend half of their very long lives out of work, spending only enough to get by?
Wil they save more? Invest more? Get bored soon after retirement and start a
new career? Take long sabbaticals from work, only to return decades later when
their money runs out? Spend less on health care because they are so much
healthier? Spend more on health care because they are living so much longer?
Invest more years and money into their educations early on?
Anyone who claims to know the answer to any of these questions is a
charlatan. Anyone who says these questions aren’t important is a fool. We have
absolutely no idea what’s going to happen. We are ying blind into one of the
most economical y destabilizing events in the history of the world.
Yet that is not the worst of it.
WHAT DIVIDES US GROWS GREATER
If you were a member of the American upper middle class in the 1970s, you
weren’t just enjoying a more a uent life, you had a longer one, too. Those in the
top half of the economy were living an average of 1.2 more years than those inthe bottom half.
By the early 2000s, the di erence had increased dramatical y. Those in the
upper half of the income spectrum could expect nearly six additional years of
life, and by 2018, the divide had widened, with the richest 10 percent of
Americans living thirteen more years of life than the poorest 10 percent. 40
The impact of this disparity cannot be overstated. Just by living longer, the
rich are getting richer. And of course, by getting richer, they are living longer.
Extra years o er more time to preside over family businesses, and more time for
family investments to multiply exponential y.
Riches are not just invested into companies; they provide rich people with
access to the world’s leading doctors (there are about ve in the United States
that they al seem to use), nutritionists, personal trainers, yoga instructors, and
the latest medical therapies—stem cel injections, hormones, longevity drugs—
which mean they stay healthier and live longer, which al ows them to
accumulate even more wealth during their lifetimes. The accumulation of
wealth has been a virtuous cycle for families lucky enough to get onto it.
And the rich don’t invest only in their health; they also invest in politics,
which is no smal part of the reason why a series of revisions to the US tax code
has resulted in a dramatic reduction in taxes on the wealthy.
Most countries tax people when they die as a way to limit wealth
accumulation over generations, but it’s a little-known fact that, in the United
States, estate taxes weren’t initial y designed to limit multigenerational wealth;
they were imposed to nance wars.41 In 1797, a federal tax was imposed to build
a navy to fend o a possible French invasion; in 1862, an inheritance tax was
instituted to nance the Civil War. The 1916 estate tax, which was similar to
present-day estate taxes, helped pay for World War I.
In recent times, the burden of paying for wars has shifted to the rest of the
population. Thanks to tax loopholes, the percentage of rich American families
who pay what were cleverly branded as “death taxes” decreased vefold,
providing the lowest cost for “dying rich” in modern times. 42
Al this means that the children of the wealthy are faring extremely wel .
Unless there is an upward revision to the tax code, they wil continue to do
better, both in how much money they inherit and in how much longer they willive than others do.
Remember, too, that aging is not yet considered a disease by any nation.
Insurance companies don’t cover pharmaceuticals to treat diseases that aren’t
recognized by government regulators, even if it would bene t humanity and the
nation’s bottom line. Without such a designation, unless you are already
su ering from a speci c disease, such as diabetes in the case of metformin,
longevity drugs wil have to be paid for out of pocket, for they wil be elective
luxuries. Unless aging is designated a medical condition, initial y only the
wealthy wil be able to a ord many of these advances. The same wil be true for
the most advanced biotracking, DNA sequencing, and epigenome analyses to
permit truly personalized health care. Eventual y prices wil come down, but
unless governments act soon, there wil be a period of major disparity between
the very rich and the rest of the world.
Imagine a world of haves and have-nots unlike anything we have experienced
since the dark ages: a world in which those born into a certain station in life can,
by virtue of nothing more than exceptional fortune, live thirty years longer than
those who were born without the means to literal y buy into therapies that
provide longer healthspans and enable more productive working years and
greater investment returns.
We have already taken the rst tenuous steps into a world that was predicted
by the 1997 lm Gattaca, a society in which technologies original y intended to
assist in human reproduction are used to eliminate “prejudicial conditions,” but
only for those who can a ord them. In the coming decades, barring a safety issue
or a global backlash against the unknown, we’l likely see the increased ability
and acceptance of gene editing global y, providing would-be parents with the
option to limit disease susceptibility, choose physical traits, and even select
intel ectual and athletic abilities. Those of means who wish to give their children
“the best possible start,” as a doctor tel s two prospective parents in Gattaca, wil
be able to do so, and with longevity genes identi ed, they could be given the best
possible nish, too. Whatever advantages genetical y enhanced people wil
already have, they could be multiplied by virtue of economic access to longevity
drugs, organ replacements, and therapies we haven’t even yet dreamed of.
Indeed, unless we act to ensure equality, we stand at the precipice of a worldin which the über-rich could ensure that their children, and even their
companion animals, live far longer than some poor people’s children do.
That would be a world in which the rich and poor wil be separated not
simply by di ering economic experiences but by the very ways in which human
life is de ned—a world in which the rich wil be permitted to evolve and the
poor are left behind.
Yet . . .
Notwithstanding the potential that extending human longevity has to
exacerbate some of the direst problems of our world—and indeed to give us new
troubles in the decades to come—I remain optimistic about the potential of this
revolution to change the world for the better.
We’ve been here before, after al .
TO WEND OUR WAY
To understand the future, it is often helpful to travel into the past. So if we want
to better understand the desperate world we are about to enter, a good place to
go is to another desperate time.
In a city brimming with iconic landmarks, from the Tower of London to
Trafalgar Square, from Buckingham Palace to Big Ben, it is perfectly reasonable
that many people, and indeed even many Londoners, have never dedicated so
much as a thought to the Cannon Street Railway Bridge.
There are no songs about it; not to my knowledge, at least. I know of no
authors who have set their stories upon its rusted rails. When it appears in
cityscape paintings, it is almost always an incidental character.
Granted, it is a rather unsightly thing, an uninvolving and utterly utilitarian
structure of green-painted steel and concrete. And if you were to look easterly
upon the River Thames from the far more charming, lamp-lined sidewalks of
Southwark Bridge, you could indeed be forgiven for missing it altogether,
although it is right before you, for just beyond on the right is architect Renzo
Piano’s famous Shard building, and just beyond that, spanning the river, is theeven more famous London Bridge, among other grand sights downstream.
In 1866, the year the Cannon Street Railway bridge was opened, there were
nearly 3 mil ion people in London. More arrived in the years to come, often
arriving from abroad by boat to Cannon Street Station, London’s equivalent of
El is Island, and dispersing from there by rail, across the humble bridge, to the
other parts of the city as it grew more and more crowded by the day. I can
scarcely imagine what someone looking upon the throngs of out-of-town
arrivals must have thought in the years in which London seemed so clearly
unable to sustain any more people, let alone the masses coming from other parts
of the world and the many more being born into the already overcrowded city.
Even the exodus to colonies in the Americas and Australia did nothing to
stem the population explosion. By 1800, approximately a mil ion people were
living in London, and by the 1860s that number had tripled, unleashing dire
consequences on the capital of the British Empire.
Central London was a particularly hel ish place. The mud and horse manure
were often ankle deep in streets further littered by newspapers, broken glass,
cigar ends, and rotting food. Dockworkers, factory workers, laundresses, and
their families were packed into tiny hovels with dirt oors. The air was thick
with soot in the summer and soot-drenched fog in the winter. With every
breath, Londoners l ed their lungs with mutagenic, acid-coated particles of
sulfur, wood, metals, soil, and dust.
A sewer system intended to take human waste away from the richer
neighborhoods of central London did just that—sending it into the River
Thames, where it owed east past the Isle of Dogs toward the poorer quarters,
where people drew the water to wash and drink. 43, 44
In those squalid conditions, it should come as little surprise that cholera
could spread with devastating speed. And it had, with three large outbreaks so
far that century, in 1831, 1848, and 1853, claiming more than 30,000 lives, with
thousands more lost to smal er outbreaks during the intermediate years.
The Final Catastrophe, as it came to be known, was focused almost
exclusively on the inhabitants of Soho in the West End, where a contaminated
wel provided water to more than a thousand people. Today, the Broad Street
pump is preserved on what is now Broadwick Street, surrounded by pubs,restaurants, and high-end clothing stores. The pump’s granite base is often used
as a seat by unsuspecting tourists. Save for the keystone plaque on the building
nearby, there are no clues about the misery this site wrought.
Twenty people died in the rst week of the cholera outbreak, July 7 to 14 of
1866, fal ing to diarrhea, nausea, vomiting, and dehydration. Doctors had only
just realized that they were dealing with another outbreak when the second wave
began. More than three hundred additional people had died by July 21. From
there it only got worse. On no day between July 21 and August 6 did fewer than
a hundred people perish, and the death tol continued to mount through
November.
That was the hel scape in which a former domestic servant named Sarah Neal
gave birth to her fourth child on September 21, 1866, just six miles south of the
epicenter of the outbreak. She cal ed her son “Bertie.” So did her husband,
Joseph Wel s. But the boy would ultimately choose to go by the initials of his
given name, Herbert George.
In the center of despair and squalor, in a city breaking under the weight of a
population boom, in the heart of hopelessness, was born the father of utopian
futurism, H. G. Wel s.
Wel s is most famous today for his dystopian ction The Time Machine, but
in stories such as The Shape of Things to Come, he audaciously predicted a
“future history” that included genetic engineering, lasers, airplanes, audiobooks,
and television. 45 He also predicted that scientists and engineers would lead us
away from ghting war after war toward a world devoid of violence, poverty,
hunger, and disease. 46 It was, in many ways, a blueprint for Star Trek creator Gene Roddenberry’s vision of a future Earth that would be a utopian base for
exploration of the “ nal frontier.” 47
How did we go from a world of such misery to one in which such dreams
were even possible?
Wel , as it turned out, the disease was the cure.
The Cannon Street Bridge, completed the same year that cursed London
with the Final Catastrophe and blessed the world with the genius of H. G. Wel s,
stands as a testament to the ways in which the London of yesterday came to be
the London of today, of how population and progress are intrinsical yconnected, and, indeed, of utopian dreams realized. For London’s nineteenth-
century population boom forced the city to confront its most horri c
chal enges. There was simply no other option. The choice was clear: adapt or
perish. 48
And so it was that the late nineteenth century brought to London some of
the world’s rst public housing projects, replacing dirt- oored shanties with
plumbed tenements that would, upon the passage of the Housing of the
Working Classes Act of 1900, also have access to electric power. The same time
period saw a tremendous rise in the number and quality of public institutions of
education, including mandated schooling for children between the ages of 5 and
12, imperfectly but increasingly drawing legions of children away from the
dangerous and exploitative conditions of life on London’s streets.
Perhaps the most important of the reforms, however, was in the eld of
public health, beginning in 1854 with the physician John Snow’s rebel ion
against the entrenched medical view that cholera was caused by miasma, or “bad
air.” By talking to residents and triangulating the problem, Snow had the Broad
Street pump’s handle removed. The epidemic soon ended. Government o cials
were quick to replace the pump handle, in part because the fecal-oral route of
infection was too horri c to contemplate. Final y, in the eventful year of 1866,
Snow’s chief opponent, Wil iam Farr, was investigating another cholera
outbreak and came to the realization that Snow was right. The resolution of that
public health skirmish led to improved water delivery and sewage systems in the
capital of the world’s largest empire.
Those innovations were soon copied around the globe—one of the greatest
global health achievements in human history. Far more than any other lifestyle
change or medical intervention, clean water and working sanitation systems have
led to longer and healthier lives the world over. And London, where this al
began, is Exhibit A. Lifespans in the United Kingdom have more than doubled
in the past 150 years, in no smal part because of innovations that were made in
direct response to the overcrowding in it that the early-nineteenth-century
parliamentarian Wil iam Cobbett derisively cal ed the Great Wen, a nickname
that compared the city to a swel ing, pus- l ed, sebaceous cyst.
The movement from miasmatic theory to germ theory, meanwhile,fundamental y shifted ideas about how to combat al sorts of other diseases,
setting the stage for Louis Pasteur’s breakthroughs in fermentation,
pasteurization, and vaccination. The ripples are manifold and can be measured,
without the slightest hint of hyperbole, in hundreds of mil ions of human lives.
If it hadn’t been for the advances that came out of that period of our history,
bil ions upon bil ions of people would not be alive today. You might be here. I
might be here. But the chances that we would both be here would be very slim.
It turned out that the population of London wasn’t the problem after al .
The problem wasn’t how many people lived in the city but how they lived in
the city.
At 9 mil ion residents and stil growing, London today has three times as
many people as it did in 1866 but far less death, disease, and despair.
Indeed, if you were to describe the London of today to Londoners of the
1860s, I submit that you would be hard-pressed to nd a single soul who would
not agree that their city, in the twenty- rst century, would have far surpassed
their most sanguine utopian dreams.
Do not get me wrong: the limitless and legitimate concerns people express
about a world in which humans live twice as long as they do now—or longer—
cannot be dismissed with a story about old London. The city is by no means
perfect. Anyone who has ever priced a one-bedroom at in the city knows this to
be true.
But today, we can plainly see that the city is ourishing not in spite of its
population but because of it, such that today the capital of and most populous
city in the United Kingdom is home to a myriad of museums, restaurants, clubs,
and culture. It is home to several Premier League footbal clubs, the world’s
most prestigious tennis tournament, and two of the best cricket teams on the
globe. It is home to one of the world’s largest stock exchanges, a booming tech
sector, and many of the world’s biggest and most powerful law rms. It is home
to dozens of institutions of higher education and hundreds of thousands of
university students.
And it is home to what is arguably the most prestigious national scienti c
association in the world, the Royal Society.
Founded in the 1600s during the Age of Enlightenment and formerly headedby Australia’s catalyst, the botanist Sir Joseph Banks, as wel as such legendary
minds as Sir Isaac Newton and Thomas Henry Huxley, the society’s cheeky
motto is a pretty good one to live by: “Nullius in Verba,” it says underneath the
society’s coat of arms. That’s Latin for “Take nobody’s word for it.”
So far in this chapter I have presented a case—one agreed upon by many great
scientists—that even at current and very conservative population growth
projections, based on lives that are extended only slightly in the coming decades,
our planet is already past its carrying capacity and we, as a species, are only
exacerbating that problem with the ways in which we are increasingly choosing
to live. And yes, advances in healthspans and lifespans could greatly exacerbate
some of the problems we already face as a society.
But there is another way of seeing our future—one in which prolonged
vitality and increasing populations are every bit as inevitable but not damning to
our world. In this future, the coming changes are our salvation.
But, please: don’t just take my word for it.
A SPECIES WITH NO LIMITS
When he is remembered at al , the Dutch amateur scientist Antonie van
Leeuwenhoek is almost always thought of as the father of microbiology. But
Leeuwenhoek dabbled in great questions of al sorts, including one that may
impact the world every bit as grandly. In 1679, by way of trying to convey to the
Royal Society just how multitudinous the unseen microscopic world was, he
embarked upon an e ort to calculate—“but very roughly,” he hastened to add—
the number of human beings who could survive on the Earth. 49 Using the
population of Hol and at the time, which was roughly 1 mil ion people, and
some very round estimates for the globe’s size and total land surface, he came to
the conclusion that the planet could carry about 13.4 bil ion people.
That wasn’t a bad guess for someone using what we might today cal “back-
of-the-napkin” math. Albeit high, it’s in the bal park of the estimates of many
more contemporary scientists who have explored the same question with farmore data to work with.
A United Nations Environment Programme report detailing sixty- ve
scienti c estimates of global carrying capacity found that the majority—thirty-
three—had pegged the maximum sustainable human population at 8 bil ion or
fewer people. And yes, by these estimates, we have either already met or wil soon
meet the maximum number of human beings our planet can sustain.50
But an almost equal number of estimates—thirty-two of them—concluded
that the number is somewhere above 8 bil ion. Eighteen of those estimates
suggested that the carrying capacity is at least 16 bil ion. And a few estimates
suggested that our planet has the potential to sustain more than 100 bil ion
people.
Clearly, someone’s numbers must be way o .
As you might imagine, these varying estimates are largely dependent on
di erences in the ways in which the constraining limits of population are
de ned. Some researchers consider only the most basic factors; not unlike
Leeuwenhoek, they speculate as to a maximum population per square mile,
multiply that by the roughly 25 mil ion square miles of habitable land on Earth,
and that’s that.
More robust estimates have included basic constraining factors such as food
and water. After al , it does not matter if we can t tens of thousands of people
into a square mile—as is the case in exceptional y dense cities such as Manil a,
Mumbai, and Montrouge—if those people starve or die of thirst.
Detailed estimates of the entire globe’s carrying capacity include the
interaction of constraining factors and the impact of human exploitation of the
global environment. Having enough land and water doesn’t matter, either, if
continued population growth aggravates the already dire consequences of
climate change, further destroying the forests and biological diversity that
sustain our existence.
But whatever the method and whatever the resulting number, the very act of
engaging in the process of trying to derive a carrying capacity acknowledges that
there is, in fact, a de nitive uppermost limit. Indeed, my col eague at Harvard,
the Pulitzer Prize–winning biologist Edward O. Wilson, wrote in The Future of
Life, “it should be obvious to anyone not in a euphoric delirium that whateverhumanity does or does not do, Earth’s capacity to support our species is
approaching the limit. ”51 That was in 2002, when the Earth’s population was a
paltry 6.3 bil ion. In the next fteen years, another 1.5 bil ion people were added.
Scientists general y pride themselves on rejecting the notion that anything
“should be obvious.” Evidence, not obviousness, drives our work. So at the very
least, the overwhelming certainty that a limit exists deserves to be debated, as any
scienti c idea does.
It needs to be pointed out that very few of the global carrying capacity
models account for human ingenuity. As we have discussed, it is easier not to see
things coming than to see them, so we tend to extrapolate into the future
directly from the way things are now. That’s unfortunate and, in my view,
scienti cal y wrong, for it eliminates an important factor from the equation.
Positive views about the future aren’t as popular as negative ones. In rejecting
wel -meaning but imperfect estimates and arguing that there is no scienti cal y
foreseeable limit to the number of people the planet can sustain, the
environmental scientist Erle C. El is at the University of Maryland has taken a
lot of heat. That, of course, is what happens when scientists chal enge
entrenched ideas. But El is has stood rm, even penning an op-ed for the New
York Times in which he cal ed the very notion that we might be able to identify a
global carrying capacity “nonsense. ”52
“The idea that humans must live within the natural environmental limits of
our planet denies the realities of our entire history, and most likely the future,”
he wrote. “. . . Our planet’s human-carrying capacity emerges from the
capabilities of our social systems and our technologies more than from any
environmental limits. ”53
If there were anything like a “natural” limit, El is has argued, the human
population probably exceeded it tens of thousands of years ago, when our
hunter-gatherer ancestors began to rely upon increasingly sophisticated water
control systems and agricultural technologies to sustain and grow their numbers.
From that point on, our species has grown only by the combined grace of the
natural world and our ability to adapt to it technological y.
“Humans are niche creators,” El is stated. “We transform ecosystems tosustain ourselves. This is what we do and have always done.”
In this way of thinking, few of the adaptations that sustain our lives are
“natural.” Water delivery systems are not natural. Agriculture is not natural.
Electricity is not natural. Schools and hospitals and roads and clothes are not
natural. We have long since crossed al of those gurative and literal bridges.
On a plane from Boston to Tokyo recently, I introduced myself to a man
sitting next to me and we chatted about our work. When I told him that I was
endeavoring to extend human lives, he curled his upper lip.
“I don’t know about that,” he said. “It sounds unnatural.”
I gestured for him to look around. “We are in reclinable chairs, ying at six
hundred miles an hour seven miles above the North Pole, at night, breathing
pressurized air, drinking gin and tonics, texting our partners, and watching on-
demand movies,” I said. “What about any of this is natural?”
You don’t have to be in an airplane to be removed from the natural world.
Look around. What about your current situation is “natural”?
We long ago left a world in which the vast majority of humans could expect a
life of “no arts; no letters; no society,” as Thomas Hobbes wrote in 1651, “and
which is worst of al , continual fear, and danger of violent death.”
If that is indeed what is natural, I have no interest in living a natural life, and I
would wager that you do not wish for that, either.
So what is natural? Certainly we can agree that the impulses that compel us
to live better lives—to strive for existences with less fear, danger, and violence—
are natural. And it is true that most of the adaptations that enable survival on
this planet, including our wonderful survival circuit and the longevity genes it
has created, are the products of natural selection, weeding out over bil ions of
years those who failed to hunker down when times were tough, but a great many
are skil s we’ve accumulated over the past 500,000. When chimps use sticks to
probe termite nests, birds drop rocks on mol usks to break their shel s, or
monkeys bathe in warm volcanic pools in Japan, it’s al natural.
Humans just happen to be a species that excels at acquiring and passing on
learned skil s. In the past two hundred years, we have invented and utilized a
process cal ed the scienti c method, which has accelerated the advancement of
learning. In this way of thinking, then, culture and technology are both“natural.” Innovations that permit us to feed more people, to reduce disease,
and, yes, to extend our healthy lives are natural. Cars and planes. Laptop
computers and mobile phones. The dogs and cats who share our homes. The
beds on which we sleep. The hospitals in which we care for one another in times
of sickness. Al of this is natural for creatures who long ago exceeded the
numbers that could be sustained in conditions Hobbes famously described as
“solitary, poor, nasty, brutish, and short.”
To me, the only thing that seems unnatural—in that it has never happened in
the history of our species—is to accept limitations on what we can and cannot
do to improve our lives. We have always pushed against perceived boundaries; in
fact, biology compels us to.
Prolonging vitality is a mere extension of this process. And yes, it comes with
consequences, chal enges, and risks, one of which is increased population. But
possibility is not inevitability, for as a species we are naturally compel ed to
innovate in response. The question, then, is not whether the natural and
unnatural bounties of our Earth can sustain 8 bil ion, 16 bil ion, or 20 bil ion
people. That’s a moot point. The question is whether humans can continue to
develop the technologies that wil permit us to stay ahead of the curve in the face
of population growth, and indeed make the planet a better place for al creatures.
So can we?
Absolutely. And the past century is proof.
PEOPLE, PEOPLE, GLORIOUS PEOPLE
After our species was almost driven to extinction 74,000 years ago, up until
1900, the human population grew at a rate amounting to a fraction of a percent
each year as we expanded to al habitable regions on the planet, breeding with at
least two other human species or subspecies. By 1930, thanks to sanitation and
decreases in child-mother mortality, our species was increasing its numbers at 1
percent each year. And by 1970, due to immunization and improvements in
food production global y, the rate was 2 percent each year.
Two percent might not seem like a lot, but it added up fast. It took more than120 years for our population to move from 1 bil ion to 2 bil ion, but after
reaching that mark in 1927, it took just thirty-three more years to add another
bil ion and then fourteen years to add another.
This is how, at the end of the second decade of the twenty- rst century, we
came to have more than 7.7 bil ion people on our planet, and every year one
additional person per square kilometer.54 Stepping back, if you graph human
population size over the last 10,000 years, the transition from humans being very
rare creatures to being the dominant species on Earth looks like a vertical step
up. That baby inside the bomb would, on the face of it, seem justi ed.
Over the past few decades, however, the rate of human population growth
has been fal ing steadily—principal y as women who have better economic and
social opportunities, not to mention basic human rights, choose to have fewer
children. Until the late 1960s, each woman on the planet had an average of more
than ve children. Since then that average has fal en fast, and with it the rate at
which our population is increasing has fal en, too.
The annual population growth rate has plummeted, from 2 percent around
1970 to about 1 percent today. By 2100, some researchers believe, the growth
rate could fal as low as one-tenth of 1 percent. As this happens, United Nations
demographers anticipate that our total global population wil plateau, reaching
about 11 bil ion people by the year 2100, then stop and drop from there. 55
This assumes, as we have discussed, that most people wil continue to live
longer on average but wil stil die in their 80s. That’s not likely going to be the
case. In my experience, most people tend to signi cantly overestimate the impact
of death on population growth. Of course death keeps the human population in
check, but not by much.
Bil Gates made a convincing argument for why improving human health is
money wel spent, and won’t lead to overpopulation, in his 2018 video “Does
Saving More Lives Lead to Overpopulation?” 56 The short answer is: No.
If we were to stop all deaths—every single one around the globe—right now,
we would add about 150,000 people to our planet each day. That would be 55
mil ion people each year. That might sound like a lot, but it would be less than a
single percentage point. At that rate, we would add a bil ion people to our ranks
every eighteen years, which is stil considerably slower than the rate at which thelast few bil ion people have come along and easily countered by the global
decline in family sizes.
It’s stil an increase, but it’s not the sort of exponential growth many people
fret about when they rst encounter the idea of slowing aging.
Recal , these calculations are what we’d face if we ended all deaths right away.
And although I’m very optimistic about the prospects for prolonged vitality, I’m
not that optimistic. I don’t know any reputable scientist who is. One hundred
years is a reasonable expectation for most people alive today. One hundred
twenty is our known potential and one that many people could reach—again, in
good health if technologies in development come to fruition. If epigenetic
reprogramming reaches its potential or someone comes up with another way to
convince cel s to be young again, 150 might even be possible for someone living
on this planet with us right now. And ultimately there is no upward biological
limit, no law that says we must die at a certain age.
But these milestones wil come one at a time, and slowly. Death wil remain a
part of our lives for a very long time to come, even as the time of it is pushed out
in the coming decades.
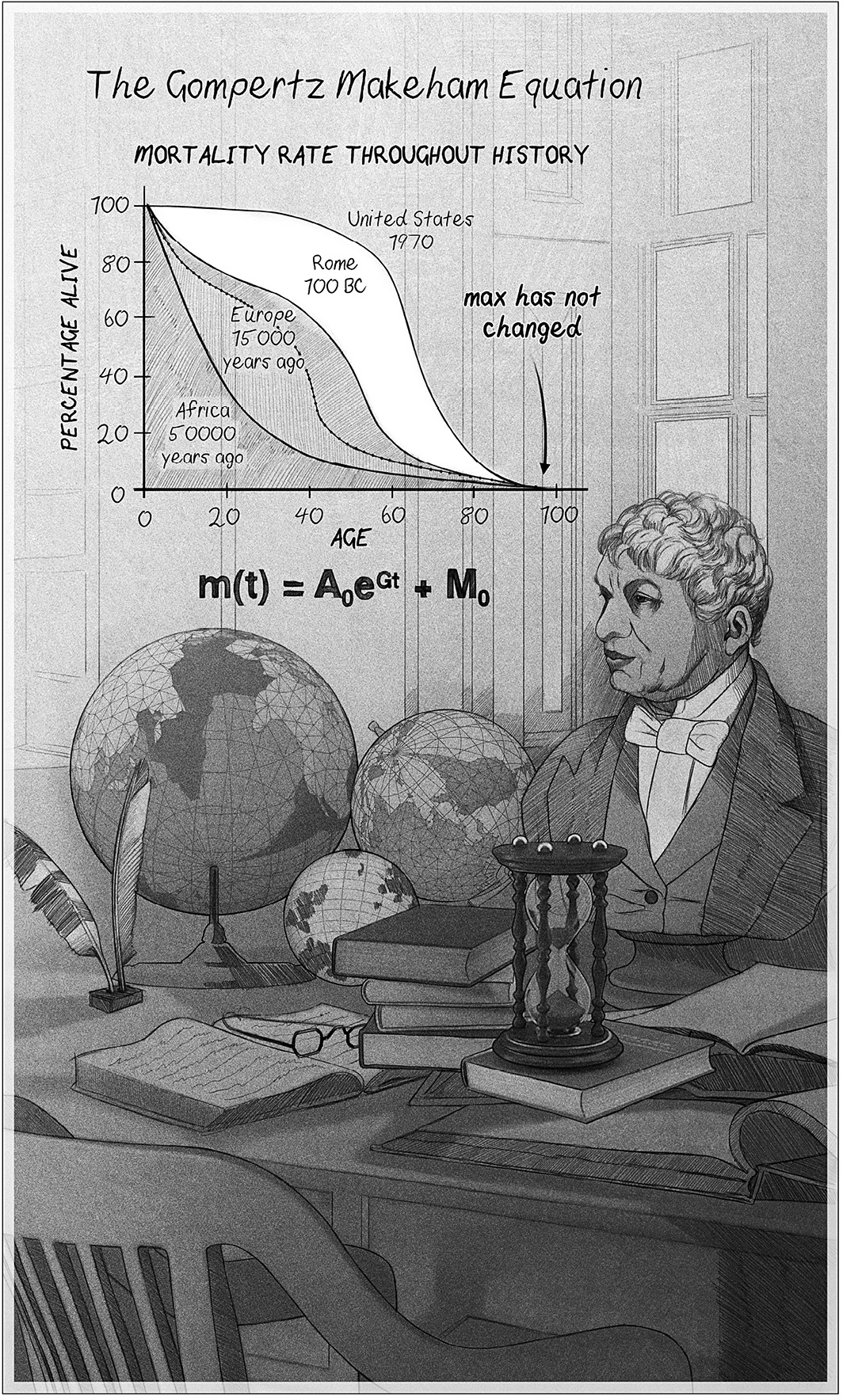
THE LAW OF HUMAN MORTALITY. Benjamin Gompertz, a self-taught mathematical genius, was
barred from attending university in nineteenth-century London for being a Jew yet was elected
to the Royal Society in 1819. His brother-in-law, Sir Moses Monte ore, in partnership with
Nathan Rothschild, founded Al iance Assurance Company in 1824, and Gompertz was
appointed actuary. His tidy equation, which replaced mortality tables, tracks the exponential
increase in the chance of death with age. As important as this “law” is to insurance companies, it
does not mean that aging is a fact of life.
That change, though, wil be set against an ongoing fal in birth rates that has
been under way for decades. So overal , our population might continue to grow
but more slowly and not at al in the explosive ways we experienced in the past
century. Rather than fearing the more moderate population increase we are
likely to see, we should welcome it. Let us not forget what happened during the
past century: our species not only survived in the midst of exponential
population growth, it thrived.
Yes: thrived. No one can ignore the vast devastation we have unleashed upon
our planet, not to mention the evils we have in icted upon one another. We
should rightful y focus our attention on these failures; that’s the only way to
learn from them. But the continual focus on the negative impacts the way wethink about the state of our world today and in the future, which is likely why,
when the global pol ing company YouGov asked people in nine developed
nations, “Al things considered, do you think the world is getting better or worse,
or neither getting better nor worse?” only 18 percent of people believed that
things were getting better.
Oh, wait. That was 18 percent of people in Australia—which was the most
optimistic of the Western nations included in the survey. In the United States,
only 6 percent of people were similarly con dent that things were getting better
in our world.
It’s important to note that the pol sters didn’t ask about whether
respondents’ individual lives were getting better or worse. They asked about the
world. And they asked people in some of the richest nations in the world. 57 And
sure, these are people who might have reason to think that their individual
standards of living—supported until recently by economic bene ts rooted in
slavery and colonialism—have been fal ing a bit in recent years. These are also
people, however, who have tremendous access to information about the world,
and thus, quite frankly, they should know better.
In much of the rest of the world, however, the future is not viewed in nearly
such a dismal way. Not at al .
In China, which holds about a fth of the global population, some 80
percent of people pol ed in 2014 by Ipsos MORI, a UK research company,
believed the lives of younger people wil be better than their own. The same
survey identi ed similarly signi cant levels of optimism in Brazil, Russia, India,
and Turkey—al places where standards of living have been on the rise. 58 And
yes, this includes habits of increasing consumption, but it also includes lowering
birth rates, fal ing rates of poverty, greater access to clean water and electricity,
more stable access to food and shelter, and greater availability of medical care.
Pessimism, it turns out, is often indicative of exceptional privilege. When
viewed global y, however, it gets a lot harder to make the case that the world is an
increasingly miserable place. It’s simply not.
In the past two hundred years—an era that saw the most explosive
population growth in human history—we transformed from a world in which
nearly everyone but monarchs and their viceroys was living in poverty to a globalsociety in which the rate of extreme poverty is now below 10 percent and rapidly
fal ing. Meanwhile, in a century in which we added bil ions of people to our
planet’s population, we also improved educational access for people around the
world. In 1800, the global literacy rate was 12 percent, by 1900 it was 21 percent,
and today it’s 85 percent. We now live in a world where more than four out of
ve people can read, the majority of whom have instant access to essential y al
the world’s knowledge.
One signi cant reason our population grew so fast in the past century was
that child mortality fel from more than 36 percent in 1900 to less than 8
percent in 2000.59 No decent person could possibly believe that our world
would be better if a third of al children were stil dying before their fth
birthday.
Did these improvements to the human condition occur in spite of our
population boom or because of it? I contend it is the latter, but it actual y
doesn’t matter. They happened simultaneously. As yet, there is real y no
evidence in modern times that population levels correlate with, let alone cause,
increases in human misery. Much to the contrary, in fact, our world is more
populated today than it ever has been—and it’s a better place for more people,
too.The Harvard psychologist Steven Pinker put it this way in his book
Enlightenment Now: The Case for Reason, Science, Humanism, and Progress:
“Most people agree that life is better than death. Health is better than sickness.
Sustenance is better than hunger. Abundance is better than poverty. Peace is
better than war. Safety is better than danger. Freedom is better than tyranny.
Equal rights are better than bigotry and discrimination. Literacy is better than
il iteracy. ”60 We have al of those things in greater plenitude today than we did a
hundred years ago, when our planet was far less populated and we lived far
shorter lives.
So, when I consider the prospect of a more populated planet, it is far easier to
envision one in which a greater share of the global population is living better
than it ever has. The science simply compels me to dream this way.
But why? Why do we live better even though there are more of us and moreof us are living longer lives?
There are a great many factors, including the good that comes from networks
of human capital of al ages. But if I had to explain it in just one word, that word
would be: “elders.”
THE LONG RACE
It was a beautiful day in San Diego, California, in June 2014. Thousands of
runners were lined up for a marathon. Among them was a woman who most
people would likely have pegged as 70. That alone would have made her an
outlier among the throngs of runners predominantly in their 20s, 30s, and 40s.
Except that Harriette Thompson wasn’t in her 70s. She was 91. And that day,
she broke the o cial US record for a marathon by a woman in her 90s—by
nearly two hours.
When she ran the same race again the fol owing year, she was just a tad slower
but set a new record as the oldest woman known to have completed a marathon.
She crossed the nish line to cheers of “Go, Harriette!” as red, white, and blue
confetti rained down around her.61
Thompson, who raised more than $100,000 for the Leukemia & Lymphoma
Society through her running, was an exceptional y special person, for her vigor
and her big heart. But what she did physical y doesn’t need to be special. In the
future, no one wil do a double take upon seeing a marathoner in his or her 90s
step up to the starting line among a chronological y younger crowd. The truth is
that it wil be hard to tel how old the veteran runners are.
That wil be the case in every other facet of life, too. In our classrooms, where
ninety-year-old teachers wil stand before seventy-year-old students embarking
on a new career, as my father did. In our homes, where great-great-grandparents
wil play rough-and-tumble games with their great-great-grandchildren. And in
our businesses, where older workers wil be revered and fought over by
employers. You can already see it happening in workplaces that depend on
experience.
And it’s about time.Old people were revered in traditional cultures as sources of wisdom. Of
course they were: before written text—and long before the advent of digital
information—elders were our only wel springs of knowledge. That began to
change, quickly and signi cantly, when a fteenth-century goldsmith, Johannes
Gutenberg, developed a press that led to the Printing Revolution. The
subsequent Education Revolution, in the nineteenth and twentieth centuries,
led to rates of literacy that grew to meet the availability of information. Elders
were no longer the only sources of long-held information. Rather than being
seen as an essential asset to a functioning society, the elderly came to be viewed as
a burden.
The Nobel laureate Seamus Heaney described our complicated relationship
with aging parents in his poem “The Fol ower,” ostensibly about his own father,
who had shoulders like sails, and Seamus, as a child, “tripping and fal ing” in his
father’s wake. The poem ends, “But today / It is my father who keeps stumbling
/ Behind me, and wil not go away.”
Heaney’s tragic poem echoes the sentiments expressed in a Life magazine
article from 1959 titled “Old Age: Personal Crisis, U.S. Problem. ”62
“The problem has never been so vast or the solution so inadequate,” the
author wrote. “Since 1900, with better medical care, life expectancy has
increased an average of 20 years. Today there are ve times as many aged as in
1900 . . . the problem of old age comes almost overnight—when a man retires,
after a woman’s husband dies.”
When I came upon the musty magazine in a Cape Cod bookstore on Old
King’s Highway, I rst marveled at how far gender equality has come since 1959,
but then was struck by how little has changed in the way we fret about the
calamity of the impending deluge of old people. Whatever wil we do with
them? Wil they overwhelm our hospitals? What if they want to keep working?
The impact of this shift in the way many people view elders has been
particularly hard felt in the workforce, where age discrimination is rampant.
Hiring managers hardly bother to hide their prejudices. They view older workers
as more likely to be sick, slow working, and incapable of handling new
technologies.
Absolutely none of that is true, especial y for people in management andleadership positions.
Yes, it used to be that technology was slow to catch on. But educated older
people now use technology just as frequently as those under 65. Don’t forget,
these are the generations who sent rockets to the moon, and invented the
supersonic passenger jet and personal computer.
“Every aspect of job performance gets better as we age,” Peter Cappel i, the
director of the Wharton Center for Human Resources, reported after he began
to investigate the stereotypes that often surround older workers. “I thought the
picture might be more mixed, but it isn’t. The juxtaposition between the
superior performance of older workers and the discrimination against them in
the workplace just real y makes no sense. ”63
Between 2012 and 2017, the average age of new CEOs at the largest
companies in the United States increased from 45 to 50 years. Yes, it’s true that
older people cannot work physical y the same way they did when they were 20,
but when it comes to management and leadership, it’s the opposite. Consider
some examples of leadership: Tim Cook, Apple’s CEO, is currently 58; Bil
Gates, Microsoft cofounder, is 63; Indra Noori, who recently stepped down as
CEO of PepsiCo and now sits on Amazon’s board, is 63; and Warren Bu ett,
the CEO of the investment rm Berkshire Hathaway, is 87. These people are not
what you’d cal technophobes.
It’s bad enough when companies al ow themselves to be deprived of great
workers because of untrue stereotypes. But this is done at a national and
international scale, sidelining mil ions of people in the best years of their work
lives—al because of old ideas about age that aren’t true now and that are going
to be even less true in the near future. Thanks to the Age Discrimination in
Employment Act of 1967, individuals in the United States over age 40 are legal y
protected from employment discrimination based on age. But in Europe, most
workers are forced to retire in their mid-60s, including professors, who are just
getting good at what they do. The best ones move to the United States so they
can keep on innovating.
It’s Europe’s loss, and it’s completely backward.
If you were the transportation director of a large company preparing to spendhundreds of thousands of dol ars to purchase some new trucks for your eet,
would it be better to invest in a model known to be reliable for about 150,000
miles or one known to last twice as long? Al other things being equal, of course
you’d choose the trucks that would last longer; that’s simply the right
investment.
We don’t tend to think about people this way, though. It feels cold. Humans
aren’t products that have been rol ed o assembly lines, after al . But people are
investments. Every society in our world places a bet on each one of its individual
citizens—chie y through education and training—that pays o over the course
of a taxpaying lifetime. Those investments already produce tremendous
dividends to our societies—for every dol ar a government spends on education,
that nation’s GDP grows on average by about $20.64 And this is in an era in which age-related sickness and death rob us of years of productivity. Imagine,
then, what the returns would be if we extended the best working years of
people’s lives.
Right now, about half of the people in the United States and Europe between
the ages of 50 and 74 are su ering from a mobility impairment. About a third
have hypertension. More than one in ten is ghting heart disease or diabetes.
More than one in twenty is su ering from cancer or lung disease.65 Many are
ghting several of these diseases at once. Even so, they far outperform the young
at most mental tasks, writing and vocabulary, and leadership.
When we extend healthy lives, we exponentialize this investment. The longer
people stay in the workforce, the better our return. That doesn’t mean people
should have to keep working. The way I see it, once you’ve repaid the investment
our society has made in you, and if you can support yourself, there’s little reason
why you shouldn’t be able to do whatever you want for as long as you want. But
as we continue to evolve into a species that stays healthier for a lot longer, old
ideas about who “belongs” in the workforce are going to change, and fast.
A lot of people worry that young workers wil be “crowded out” of jobs if no
one ever retires. I don’t. Countries stagnate because they don’t innovate and
don’t utilize their human capital, not because there aren’t enough jobs. This
explains why countries with an earlier retirement age have a lower GDP. In the
Netherlands, Sweden, the United Kingdom, and Norway, the retirement age is66 to 68, while in Moldova, Hungary, Latvia, Russia, and Ukraine, it is 60 to
62.66 I have nothing against young people—I teach and train them every day—
but I also know science and technology is getting more and more complex, and
young people can bene t greatly from learning the wisdom that decades of
experience can bring.
Looking through old magazines, it’s easy to see what scared previous
generations. It’s always the same; there are too many people and not enough
resources: too many people and not enough jobs.
In another edition of Life magazine, this one from 1963, an article says that
automation “displaces men. It has thrown hundreds of thousands of people out
of work and wil throw out many more. ”67
It then quotes from a then-recent study on the topic: “Within the next two
decades machines wil be available outside the laboratory that wil do a credible
job of original thinking, certainly as good as that expected of most mid-level
people who are supposed to ‘use their minds.’ ”
The foreboding article concludes, “While we are fast running out of use for
people, we are at the same time ironical y producing people faster than ever.”
Those fears never materialized as fact, not even in the face of another
tremendous disruption of the status quo. In 1950, the US labor force
participation rate of women was about 33 percent; by the turn of the century, it
had nearly doubled. Tens of mil ions of women began working during those
decades; that didn’t result in tens of mil ions of men losing their jobs.
The labor market isn’t a pizza with a limited number of pieces. Each of us can
have a slice of the pie. And in fact, greater labor participation by older people,
men and women, may be the best antidote to concerns that we’re going to
bankrupt our social insurance programs. The answer to the chal enge of keeping
Social Security solvent is not to force people to work longer but to allow them to
do so. And given the pay, respect, and advantages that wil come with extra
decades of vitality and the opportunity to continue to nd purpose through
meaningful work, many wil do so.
Even as it stands, many Americans plan to work beyond the traditional age of
retirement, at least on a part-time basis, not always because they have to but
often because they want to. 68 And as more people recognize that working wel into their so-cal ed golden years doesn’t mean feeling tired or confused at work,being treated poorly, or having to take time o to visit the doctor al the time,
the number of people who wil want to stay engaged in this part of their lives is
certain to grow. Age-related discrimination wil fal , particularly as it becomes
harder to tel who is “older” in the rst place.
And if you are a politician wondering how it wil be possible to provide
meaningful, productive work to al the people, consider the city of Boston,
where I live. Since it opened the rst American university in 1724 and the rst
American patent o ce in 1790, the city has been home to the invention of the
telephone, razor, radar, microwave oven, the internet, Facebook, DNA
sequencing, and genome editing. In 2016 alone, Boston produced 1,869 start-
ups and the state of Massachusetts registered more than 7,000 patents, about
twice as many per capita as California. 69 It is impossible to know how much wealth and how many jobs Boston has generated for the United States and
global y, but in 2016 the robotics industry alone employed more than 4,700
people in 122 start-ups and generated more than $1.6 bil ion in revenue for the
state. 70
The best way to create jobs for productive people of any age, even less skil ed
workers, is to build and attract companies that hire highly skil ed ones. If you
want a country in which your citizens ourish and that others envy, don’t reduce
the retirement age or discourage medical treatments for the elderly, hoping to
save money and make room for the young. Instead, keep your population
healthy and productive, and destroy al barriers to education and innovation.
I try my best to be aware of how lucky I am to be living in Boston and
working on things I love. So long as I am feeling physical y and mental y t, I
don’t ever want to retire. When I envision myself at 80, I see a person who
doesn’t feel a whole lot di erent than he does at 50 (and if reprogramming
works, won’t look a lot di erent, either). I imagine walking into my lab at
Harvard, much as I do these days most mornings of the week, to be bombarded
by the energy and optimism of a motley group of researchers working to make
discoveries aimed at changing bil ions of people’s lives for the better. I absolutely
love the idea of applying sixty or seventy years of experience to the task of leading and mentoring other scientists.Yes, it’s true: when people choose to keep working for eighty, ninety, or a
hundred years, it wil fundamental y change the way our economy works.
Tril ions of dol ars have been hidden away in virtual and quite a few literal
mattresses by people dreading the prospect of running out of money at a time in
their lives in which they are too frail to return to work. The option to work at
any age—if and when work is wanted and needed—wil o er a sort of freedom
that would have been unfathomable just a few years ago. The risk of spending
one’s savings on ful l ing a dream, innovating, starting a business, or going on a
new educational journey wil not be such a risk at al ; it wil simply be an
investment in a long and ful l ing life.
And it’s an investment that wil pay o in other ways, too.
UNLEASHING THE ARMY
Dana Goldman had heard from al the naysayers.
The University of Southern California economist understood—far more
than most people do—that health care costs had risen dramatical y over the past
decades, not just in his native United States but around the world. He knew
those costs were coming at a time in which human lifespans were being
extended, resulting in multitudes of patients who were sicker for longer. And he
was ful y aware of the never-ending nightmare about the future solvency of
programs such as Social Security that provide for the common welfare. The
prospect of bil ions of people growing even older seemed like a perfect storm of
economic catastrophes.
A few years back, however, Goldman began to realize that there was a
di erence between extending lives and extending healthy lives. As it stands, aging
presents a double economic whammy, because adults who get sick stop making
money and contributing to society at the same time they start costing a whole lot
to keep alive.
But what if older people could work longer? What if they were to use fewerhealth care resources? What if they were able to continue to give back to society
through volunteering, mentorship, and other forms of service? Perhaps—just
perhaps—the value of those extra healthy years would lessen the economic blow?
So Goldman began to crunch the numbers.
As any good economist would, he sought to be both rigorous and
conservative in his estimation of the bene ts of delayed aging. He and his
col eagues developed four di erent scenarios: one that simply projected
spending and savings under status quo conditions, two that estimated the
impact of modest improvements to delaying speci c diseases, and one that
evaluated the economic bene ts of delaying aging and thus reducing all the
symptoms of aging. For each scenario the researchers ran a simulation fty times
and averaged the outcomes.
When Goldman reviewed the data, something became clear: reducing the
burden of any one disease, even several, wouldn’t change much. “Making
progress against one disease means that another wil eventual y emerge in its
place,” his team reported in Perspectives in Medicine. “However, evidence
suggests that if aging is delayed, al fatal and disabling disease risks would be
lowered simultaneously.” 71
For the record, that’s precisely what I am suggesting wil happen to the total
disease burden as we slow and even reverse aging. The result wil be an upgrade
of the health care system as we know it. Treatments that once cost hundreds of
thousands of dol ars could be rendered obsolete by pil s eventual y costing
pennies to make. People wil spend the last days of their lives at home with their
families instead of racking up huge bil s in centers intended for nothing more
than “aging in place.” The idea that we once spent tril ions of dol ars trying to
eke out a few more weeks of life from people who were already teetering on the
edge of death wil be anathema.
The “peace dividend” we wil receive from ending our long war on individual
diseases wil be huge. 72 Over fty years, Goldman estimated, the potential
economic bene ts of delayed aging would add up to more than $7 tril ion in the
United States alone. And that’s a conservative estimate, based on modest
improvements in the percentages of older people living without a disease or
disability. Whatever the dol ar gure, though, the bene ts “would accruerapidly,” Goldman’s team wrote, “and would extend to al future generations,”
because once you know how to treat aging, that knowledge isn’t going away.
Even if we reinvest only a smal amount of that dividend into research, we’l
enter a new golden era of discovery. That discovery wil be hypercharged as we
unleash a vast army of bril iant people not only to continue the ght to prolong
human vitality but to combat the many other chal enges we currently face, such
as global warming, the rise of infectious diseases, moving to clean energy,
increasing access to quality education, providing food security, and preventing
extinctions. Those are chal enges we cannot e ectively ght in a world in which
we spend tens of tril ions of dol ars each year battling age-related diseases one by
one.Even now, while we spend so much of our intelectual capital on whack-a-
mole medicine, there are thousands of labs around the world with mil ions of
researchers. That sounds like a lot, but global y, researchers account for just one-
tenth of 1 percent of the population.73 How much faster would science move if
we were to unleash even a smal bit of the physical and intel ectual capital that’s
tied up in hospitals and clinics treating diseases one at a time?
This army could be augmented by bil ions of additional women if they can be
provided much longer windows of opportunity for pregnancy and parenting.
Animal studies in my lab indicate that the window of female fertility could be
extended by up to a decade. This is an exciting prospect because, in the United
States, 43 percent of women step away from their careers for a period of time,
almost always to shoulder the burden of child rearing. Many never return to
work. As a woman’s lifespan and fertility lengthens, the consequences of taking a
break wil be seen as relatively minor. By this century’s end, we wil almost
certainly look back with sadness at the world we currently inhabit, in which so
many people, particularly women, are forced to choose between parenting and
career success.
Now add to the ranks of this army the combined intel ectual power of the
men and women who are currently sidelined due to age discrimination, social y
enforced ideas about “the right time to retire,” and diseases that rob them of the
physical and intel ectual capacity to engage as they once did. Many people in
their 70s and 80s wil reenter the workforce to do something they’ve alwayswanted to do, earning more than they ever did, or serving their communities as
volunteers and helping raise their grandkids, as my father has. With the money
saved by preventing expensive medical care, a retraining fel owship could be
provided for a few years to al ow people over 70 to go back to school and start
the career they always wished they’d started but didn’t because they made the
wrong decisions or life simply got into the way.
With active people over 70 stil in the workforce, imagine the experiences that
could be shared, the institutional knowledge that could be relied upon, and the
wise leadership that would emerge. Problems that seem insurmountable today
wil look very di erent when met by the tremendous economic and intel ectual
resources o ered by prolonged human vitality.
That could be especial y true if we’re al engaging in our world with the best
version of ourselves.
THE GREATEST OF THESE
In the early 1970s, two psychologists decided to put the Parable of the Good
Samaritan to the test.
The biblical story, as you might recal , centers around the moral obligation to
help those in need, and the psychologists gured that people who had the
parable on their minds would be more likely to stop to help someone in distress.
So they hired an actor to pretend to be in pain and put the young man—who
was doubled over and coughing—into an al ey next to the doorway of the Green
Hal Annex at Princeton Theological Seminary.
The psychologists had also recruited forty seminary students to present a talk
at the annex. First, though, the students were asked to stop by another building
on campus. Once there, some of the seminarians were told they could take their
time getting to the annex, others were told they would be on time as long as they
left immediately, and a nal group was told that they needed to hurry to make it
to the annex on time.
Just 10 percent of those in the “high-hurry” group stopped to help the man.Those were seminary students, for goodness’ sake, and they ignored a brother in
need. One literal y stepped over the man in distress to get where he was supposed
to be.
In the “low-hurry” group, though, more than 60 percent stopped to help. In
that experiment, the di erence between whether a person made a compassionate
choice had nothing to do with personal morality or religious scholarship but
whether he felt rushed. 74
This isn’t a new idea, of course. Back in the days in which Christ was rst
tel ing the Good Samaritan story, his contemporary in ancient Rome Seneca the
philosopher was begging his fol owers to stop and smel the roses. “Life is very
short and anxious for those who forget the past, neglect the present, and fear the
future,” he wrote. 75
For people who don’t appreciate life, time is “reckoned very cheap . . . in fact
without any value,” he lamented. “These people do not know how precious time
is.”This might be the least considered societal advantage of prolonged vitality,
and it might just be the greatest advantage of al . Perhaps when we’re not al so
afraid of the ticking clock, we’l slow down, we’l take a breath, we’l be stoic
Samaritans.
I would like to emphasize the word “perhaps,” here. I wil be the rst to say
that this thesis is supposition more than science. But the smal -sample Princeton
experiment both fol owed and portended a lot of other research demonstrating
that humans are a lot more humane when they’ve got more time. Al of the
studies, though, take stock of how people behave when they have a few more
minutes, or perhaps a few more hours, to spare.
What would happen if we had a few more years? A few more decades? A few
more centuries?
Maybe we would do nothing di erently, even if we had two or three hundred
years. In the grand scheme of the universe, after al , three hundred years is
nothing. My rst fty years went by like a blink, and I suspect that a thousand
years, a mere twenty blinks, would also feel short.
And so it comes down to this: When those years do come, how do we wish tospend them? Wil we fol ow the perilous path that ultimately leads to a
dystopian doom? Wil we band together to create a world that exceeds our
wildest utopian dreams?
The decisions we make right now wil determine which of those futures we
create. And this is important. Preventing disease and disability is possibly the
single most impactful thing we can do to avert a global crisis precipitated by
climate change, crippling economic burdens, and future social upheavals. We
have to get this right.
Because there has been no more consequential choice in the history of our
species.
NINEA PATH FORWARD
IN 1908, ONLY FIVE YEARS after the Wright Brothers began ying, H. G. Wel s
published a book titled A War in the Air, in which Germany starts an air war
against Great Britain, France, and the United States.
To say Wel s had a penchant for prescience would be a vast understatement.
In 1914, the Institute of International Law tried to ban the dropping of
bombs from ying machines,1 but it was too little, too late. Giant German
“Gotha” planes began bombing Great Britain in 1917. That year, 180 miles west
of London, a baby was born, named Arthur, who would come to be regarded as
the preeminent science ction writer of the twentieth century. As he became
more famous, Arthur C. Clarke increasingly regarded predicting the future a
“discouraging, hazardous occupation.” That may be true, but Clarke was
awful y good at it, anticipating satel ites, home computers, email, the internet,
Google, live-streaming TV, Skype, and smart watches.
Clarke had some strong opinions about scientists: a physicist in his 30s was
already too old to be useful. In other scienti c disciplines, a 40-year-old has likely
experienced “senile decay.” And scientists of over 50 are “good for nothing but
board meetings, and should at al costs be kept out of the laboratory!”
Toward the end of his life, Clarke gave a series of interviews. Most of them
were recorded and edited because he had halting speech caused by postpolio
syndrome. In one interview, he revealed that he had a use for washed-up
scientists: “When a distinguished but elderly scientist states that something ispossible, he is almost certainly right. When he states that something is
impossible, he is very probably wrong. ”2
I’m a scientist who is now 50. Some people might cal me distinguished. And
my students de nitely don’t want me in the laboratory. So although I can’t say
I’m certain about my predictions, I’m apparently wel quali ed to make them.
I’ve been asked on occasion, by members of the US Congress and the like, for
predictions of technological breakthroughs and how they might be used for
good or evil purposes. A few years ago, I gave an opinion on the top ve future
advances in the biological sciences that would be of relevance to national
security. Though I can’t reveal what I said, I expect that most people would have
thought they were science ction. My best estimate was that they would happen
sometime before 2030. Within six months, two of them became science fact.
I don’t know precisely when the rst individual to cross the threshold of 125
years wil be, but he or she wil certainly be an outlier, as pioneers always are. In
just a few years, he or she wil be joined by another. Then dozens more. Then
hundreds. Then the fact won’t be worth noting. Even longer lifespans wil
become more and more common. The world may see the rst sesquicentenarian
sometime in the twenty-second century. (If you think that’s far o , consider that
some researchers believe half of al American children born today wil celebrate
New Year’s Eve 2120. Not outliers—half.)3
Those who think al this is impossible are ignorant of the science. Or they are
denying it. Either way, they are almost certainly wrong. And because things are
moving so fast, many of them might even live to realize they are wrong.
No biological law says there is a limit to how long we can live; there is no
scienti c mandate that the average age at death must be 80 years. And there is no
God-given mandate to die after fourscore years. Indeed, in Genesis 35:28, Isaac is
claimed to have lived “one hundred and fourscore. ”4
Thanks to the technologies I’ve described, a prolonged, healthier human
lifespan is inevitable. How and when we’l achieve it is a bit less certain, although
the general path is quite clear. The evidence of the e ectiveness of AMPK
activators, TOR inhibitors, and sirtuin activators is deep and wide. On top of
what we already know about metformin, NAD boosters, rapalogs, and
senolytics, every day the odds increase that even more e ective molecule or genetherapy wil be discovered, as bril iant researchers around the world join the
global ght to treat aging, the mother of al diseases.
Al of that comes on top of the other innovations that are on track to further
lengthen our lives and strengthen our health, such as senolytics and cel ular
reprogramming. Add to that the power of truly personalized care to keep our
bodies running, prevent disease, and get ahead of problems that could be
troublesome down the road. That’s not to mention the very easy steps we can al
take right now to engage our longevity genes in ways that wil provide us with
more good years.
With signi cantly prolonged vitality an inexorable part of our future world,
what do you want that world to look like?
Are you comfortable with a future in which the rich live much longer than
the poor and in doing so get richer with every passing year? Do you want to live
in a world in which an ever-increasing population greedily scraps for every last
remaining resource while the world continues be less and less habitable?
If so, there is nothing left for you to do. The status quo wil get us there—
regardless, in fact, of whether we prolong human life or not. You can sit back,
relax, and watch the world burn.
There is another potential future, though, one in which prolonged
youthfulness is the torch that lights the way to greater universal prosperity,
sustainability, and human decency. This is a future in which tremendous
resources are freed from a medical-industrial complex that is based on battling
diseases one by one, thus creating tremendous opportunities to tackle other
chal enges. It is a future in which people who have lived on this planet for a long
time are revered for their knowledge and skil s. It is a future of global Good
Samaritanism.
It’s also a future for which we must ght, for it is in no way guaranteed.
To get there, we have some work to do.
INVEST PUBLIC MONEY TO TACKLE AGING, NOW
I am a serial entrepreneur, a disciple of innovation, and a grateful bene ciary ofpeople’s investment in me and the teams I’ve brought together to solve hard
problems.
I also recognize, however, that the free market doesn’t magical y produce
good science or equitable outcomes when it comes to health care. In any research
endeavor, a balance of public and private funding is vital to producing the
conditions that encourage unbound scienti c exploration, the investment in
early discoveries, and a degree of common ownership that better ensures that the
bene ts of newfound knowledge wil become available to the greatest possible
number of people.
That balance has become ever more precarious in recent years. Starting in
2017, for the rst time since World War II, the US federal government was no
longer the majority source of funding for basic scienti c research in the United
States.
Federal funding for science in the United States began in the 1880s, when the
Marine Hospital Service, the predecessor of the National Institutes of Health,
was charged by Congress with examining passengers on arriving ships for clinical
signs of infectious diseases, such as cholera.5 In 1901, a routine supplemental appropriations act, providing $35,000 for a new building, became the founding
legislation of the NIH. Congress was not convinced that the money would go to
good use, so it ensured that every year, funding would be at the discretion of
Congress, and so it remains. Hopeful y Congress remains convinced that NIH
funding, which provides hundreds of competitive grants to scientists around the
country, is money wel spent, because without NIH-funded research, the
majority of the medicines and medical technologies we rely on would never have
been discovered, not to mention the thousands of new medicines stil waiting to
be discovered.
At least for now, the federal government stil makes up a large share of total
funding for medical research at hospitals and universities, ensuring that R&D
isn’t driven by pro t alone. This is important, so scientists like me can run with
their imaginations and instinct, sometimes for a decade, before any commercial
applications are apparent and long before any investor would consider
supporting the work to help it survive the innovation “val ey of death.”
Clearly, government is essential in this ecosystem, but in a world in whichthere is more competition for overal research funding than ever before, good
scientists investigating aging are having to seek more and more private nancial
support for their work; world-changing research certainly isn’t cheap, and when
it’s funded by a company with short-term goals, it isn’t free, either. That’s why
it’s important that we reverse the decline in public funding for medical research,
which fel by 11 percent in real dol ars from 2003 to 2018.6
The situation is particularly hard for researchers studying aging. Funding to
understand the “biology of aging” gets less than 1 percent of the total US
medical research budget. 7 With an aging population and ever-increasing health
care costs, why aren’t governments dramatical y increasing funding for aging
research to keep people healthier for longer?
The reason is, in nearly every nation in the world that has made a civic
investment in medical research, that research is tied back, if not completely tied
up, by the de nition of disease.
If you are a scientist with an idea for a novel way to slow the progression of
cancer or a researcher with an inventive idea for ending Alzheimer’s disease, the
NIH and similar national research funding agencies around the world are there
to help. The NIH isn’t simply a bunch of buildings in Bethesda, Maryland. It
al ocates more than 80 percent of its budget to almost 50,000 competitive grants
to about 300,000 researchers at more than 2,500 universities and research
institutions. Medical research would almost grind to a halt without that money.
It’s worth dril ing down into the NIH budget to see which of the 285
diseases that are being researched get the most attention.8
• Heart disease gets $1.7 bil ion for a disease that affects 11.7 percent of the
population.
• Cancer gets $6.3 bil ion to impact 8.7 percent.
• Alzheimer’s disease gets $3 bil ion for a disease that impacts 3 percent—at
most.9
How much does obesity, which a ects 30 percent of the population and
reduces lifespan by over a decade, get? Less than a bil ion dol ars.
Don’t get me wrong. Compared to how the government spends a lot of itsmoney—the cost of a single F-22 Raptor ghter jet is upward of $335 mil ion,
for instance—this is al money wel spent. To put it into even greater perspective,
though, consider this: US consumers spend more than $300 bil ion per year on
co ee.10
To be fair, life without co ee might not be worth living. But if you are a
researcher who wants to make life even better—by slowing or reversing diseases
of aging—you have a bit of a problem. There just isn’t that much public money
being spent on that area of science.
In 2018, Congress appropriated $3.5 bil ion for research into aging, but if
you were to dig into the budgetary documents, you’d see that the money went
almost entirely to research Alzheimer’s disease, perform clinical trials of
hormone replacement therapy, and study the lives of the elderly. Less than 3
percent of the funding for “aging research” was actual y for the study of the
biology of aging.
Aging disables 93 percent of people over the age of 50, but in 2018 the NIH
spent on aging less than a tenth of what was spent on cancer research. 11
One scientist who is particularly annoyed by the budgetary focus on
individual diseases is Leonard Hay ick, the scientist who rst discovered that
human cel s in a dish have a limited capacity to divide and eventual y senesce,
after having reached the Hay ick Limit.
“The resolution of Alzheimer’s disease as a cause of death wil add about 19
days onto human life expectancy,” he noted in 2016. 12 Hay ick has suggested
that the name of the National Institute on Aging, a division of the NIH, might
as wel be changed to the National Institute on Alzheimer’s Disease.
“Not that I support ending research on Alzheimer’s disease, I do not,” he
said, “but the study of Alzheimer’s disease and even its resolution wil tel us
nothing about the fundamental biology of aging.”
The relatively tiny amount the United States spends on research into aging,
however, is generous when compared to that of most other advanced nations,
which invest next to nothing. There is no doubt that this situation is a direct
result of the establishment view that aging is an inevitable part of life rather than
what it actual y is, a disease that kil s about 90 percent of the population.
Aging is a disease. This is so clear that it seems almost insane that those fourwords need to be repeated again and again. But I’l do so anyway: aging is a
disease. And not only is it a disease, but it is the mother of al diseases, the one we
al su er from.
Paradoxical y, no public funding agency around the world classi es aging as a
disease. Why? Because, if we are fortunate to live long enough, we all su er from
it. And thus for now, the pool of public funding available for research aimed at
prolonged vitality is rather paltry; the biggest checks are stil being written to
support initiatives aimed at recognized diseases. And at the moment I am
writing these words, aging isn’t recognized as a disease. Not in any nation.
There are several ways to speed innovation to nd and develop medicines and
technologies that prolong healthy lifespan, but the easiest is also the simplest:
de ne aging as a disease. Nothing else needs to change. Researchers working on
aging wil compete on equal footing with researchers working to cure every
other disease in the world. The science-based merits of grant proposals wil
dictate which research e orts are funded. And private investment wil continue,
as it should, to drive innovation and competition.
Labs like mine, focused speci cal y on developing innovative therapies to
treat, stop, and reverse aging, wil no longer be rare. There wil be one or more at
every health science university in the world.
And there should be, because there is no shortage of scientists lining up to
enlist in this army. Right now, I and other researchers who study aging are being
besieged by eager, experienced, and absolutely bril iant youths who want
nothing more than to devote their lives to the ght to stop aging. For lab heads
like me, it’s a virtual buyer’s market. There are far more people who want to
work in aging than there are labs they can work in. What this means is that there
are a lot of people who, despite being wicked smart and raring to tackle the aging
problem, are having to work in other elds or other professions. This wil soon
change.
The rst nations to de ne aging as a disease, both in custom and on paper,
wil change the course of the future. The rst places to provide large amounts of
public funding to augment the fast-growing private investments in this eld wil
prosper in kind. It wil be their citizens who bene t rst. Doctors wil feel
comfortable prescribing medicines, such as metformin, to their patients beforethey become irreversibly frail. Jobs wil be created. Scientists and drug makers
wil ock to that country. Industries wil thrive. Their national budget wil see a
signi cant return on investment. Their leaders’ names wil be in the history
books.
And the holders of the patents, the universities and the companies, wil have
more money than they know what to do with.
I’m proud to say that Australia is leading the charge to de ne aging as a
treatable disease. I recently made a trip to Canberra to meet with Greg Hunt, the
minister for health, and Deputy Secretatry Professor John Skerritt of the
Therapeutic Drug Authority, and about 15 of Australia’s other top aging
researchers. I learned that developing a drug for aging may be far easier in my
native land than in the United States. While the United States expects evidence
that a disease is cured or al eviated, in Australia it is possible for a drug to receive
approval for “in uencing, inhibiting or modifying a physiological process in
persons.” In the aging eld, we know how to do that!
Singapore and the United States are among the nations that are also seriously
considering a regulatory shift. Whichever does so rst wil be making a
historical y important decision, one that wil bene t itself rst and foremost.
There’s a reason why the United States virtual y owns the aerospace sector—
exporting products worth more than $131 bil ion in 2017, or more than the next
three national exporters combined. “First in Flight” isn’t just a good slogan for
North Carolina license plates; it’s a statement about why being out front
matters. Americans retain the pioneering spirit of their ancestors: anything is
possible. More than a century after the Wright brothers ew the rst planes at
Kitty Hawk, and after almost losing out to the French and British, the United
States is stil ahead in the ight game. It has the world’s most powerful air force.
It got to the moon rst. And it has a big lead in the development of public and
private initiatives to put people on Mars.
But none of that wil impact human history as much as the rst nation to
declare aging a disease.
At a bare minimum, governments have a vested interest in making sure the
innovations we develop to protect human life are used wisely and for our
col ective bene t. The time to talk about ethics and how personal privacy wil beimpacted by these coming technologies is now, for once the bottle is opened, it
wil be exceedingly hard to put the genie back. DNA-based technologies that
enable the detection of speci c pathogens, for example, could also be used to
search for speci c people. Technology now exists to create humans that are
stronger and longer lived. Wil parents choose to give their children “the best
possible start” ?13 Wil the United Nations outlaw the genetic improvement of
citizens and military?
To create a future worth living in, it won’t be enough to simply fund research
that lengthens and protects people’s lives and ban its misuse. We must also
ensure that everyone bene ts together.
IT’S TIME TO INSIST ON THE RIGHT TO BE TREATED
The dentist looked bored. “Your teeth are ne,” she told me as she peered into
my mouth. “Just the normal wear and tear. I’l send the hygienist in for your
cleaning, and we’l get you on your way.”
It seemed as though she were turning away before her ngers were even out
of my mouth.
“Doctor, if you can spare a moment,” I said, “can you tel me what you mean
by ‘normal wear and tear’?”
“You’re getting older, and your teeth are showing that,” she said. “Your two
front teeth are worn down. Total y normal. If you were a teenager, we would
probably x them but—”
“Wel , then,” I said. “I’d like to have them xed.”
Eventual y the dentist relented, although not before I told her what I did for a
living and explained to her that I was hoping to use my teeth for a very long time
to come. I also assured her that I would be happy to pay for the procedure even if
my insurance wouldn’t.
Her resistance was understandable. When dentists look into the mouths of
patients who are in their 40s and 50s, they have long been looking at teeth that
are halfway done with their jobs. But that’s no longer the case. Our teeth—likeal of our other body parts—are going to have to last a lot longer now.
My experience at the dentist was a microcosm of the way middle-aged people
are treated in every facet of the health care system. When a doctor looks at a 50-
year-old person right now, his or her goal is to keep the patient “less sick,” not to
ensure that he or she wil be healthy and happy for decades to come. Who
among us over the age of 40 has not heard a doctor say the words “Wel , you’re
not twenty anymore”?
There are two things that guide medical treatments more than anything else:
age and economics. The rst often limits what doctors are even wil ing to discuss
in terms of treatment options, because they assume that people are supposed to
slow down, begin dealing with a bit of pain, and gradual y experience the
degradation of various body parts and functions. The second dictates these
discussions even more, because regardless of how much potential a procedure
may have to improve a patient’s life, it is pointless, and even heartless, to tel
someone about care he or she can’t a ord.
Indeed, our medical system is built on ageism. When we are young, we don’t
get treatments that could keep us healthy as we grow old. When we are old, we
don’t get the treatments that are routinely used on the young.
This al has to change. The quality of our medical care should not be
predicated on age or income. A 90-year-old and a 30-year-old should be treated
with the same enthusiasm and support. There wil be enough money to pay for
this because of the tril ions of dol ars that won’t have to be spent by insurance
companies or the government, and hence ourselves, on treating the chronical y
il . 14 Everyone should be entitled to treatments and therapies that improve
quality of life, no matter what the date on his or her birth certi cate is. As we
move toward a world in which the number of our birth years indicates less about
us than ever, we wil need to adjust the assumptions, rules, and laws that govern
what medical treatments people can receive.
Equitable access to medical care, no matter how long life may be, is a
terrifying idea to many people, because it sounds awful y expensive. That’s
understandable, because as it stands, social medical programs across the globe are
straining under the ever-increasing cost of treatment, especial y treatments that
are provided to those who are very sick, very old, and likely to get nothing morethan a few extra years—if that—out of the deal.
That’s not what the future of medical care needs to look like. Right now, the
overwhelming majority of the money we spend on medical care is spent ghting
diseases. But when we are able to treat aging, we wil be tackling the biggest
driver of al disease. E ective longevity drugs wil cost pennies on the dol ar
compared to the cost of treating the diseases they wil prevent.
In 2005, a study by Dana Goldman and his col eagues at RAND in Santa
Monica put some numbers on this. They estimated the value that new
discoveries would add to society and the cost to society to extend a human life by
one year.15 The cost of an innovative medicine to prevent diabetes: $147,199. Of
a cancer treatment: $498,809. Of a pacemaker: $1,403,740. Of an “antiaging
compound” that would extend healthy years by a decade: a mere $8,790.
Goldman’s numbers support an idea that should be common sense: that there is
no cheaper way to address the health care crisis than to address aging at its core.
But what if the drugs don’t keep people healthy? What if they simply prolong
life, like many cancer chemotherapy drugs, which are approved based on their
ability to provide a longer life, not a higher quality of life? Society should debate
whether longevity medicines that don’t keep us healthier should ever be
approved. If they were to be al owed, there would be even more elderly people
with disease and disability, and, according to Goldman, health care spending in
thirty years’ time would be 70 percent higher.
Fortunately, the science suggests this nightmare scenario is not going to
happen. When we have safe and e ective drugs to slow aging, they wil also
extend our healthspans. What wil be left wil be medical maintenance, which is
exceptional y cheap; emergency medicine, which is costly but rare; and
communicable diseases, which we’l be able to track, treat, and prevent with far
greater e ciency and e ectiveness. It’s similar to making the switch from
gasoline-powered cars that need oil, belts, tuning, and regular maintenance to
electric cars that tel you occasional y to top up the windscreen washer uid.
Having lived in Australia, the United Kingdom, and the United States—
three countries with an intertwined history, language, culture, and trade—I’ve
found it interesting to see how similar they are in some ways and how di erent
they are in others. 16 One big di erence is that most Australians and Britons rarely assume that their way of doing things is the best. Americans, however,often believe that their way of doing things is assuredly the best.
I’m not saying the United States doesn’t do a lot wel and shouldn’t continue
to blaze its own path in many areas of domestic and global policy, but I’ve long
been perplexed by the American resistance to studying what actual y works
elsewhere.
In science we cal this experimentation, and it’s what propels our civilization
forward. The more experiments are conducted, the better informed we are. And
some experiments work real y wel .
Seeded as a prison colony, Australia is one of the least religious countries in
the world, but when it comes to providing for its citizens, it is a city upon a
hil . 17 Like the United States, Australia has its problems: tra c snarls, a high cost
of living, and strict rules aimed at saving lives, even if those rules often take the
fun out of life.
There is a statistic, though, of which Australians are increasingly proud: a
fty-year-long experiment to protect and preserve every citizen, regardless of
social position, education, or income. Deaths from car accidents and smoking
are the lowest in the world, thanks to strict laws and hefty nes. Even before
these laws passed, there was bigger change afoot. In the mid-1970s, a universal
health care system was enacted, one of the rst ever, and life expectancy in
Australia began to shoot up. Similar to the United States in the 2010s, the next
government tried to limit the scope of that progressive reform but ultimately
failed.
A controversial, right-wing politician, Bronwyn Bishop, helped create an
independent Australian Federal Department of Health and Ageing, which lasted
from 2002 to 2013 with a budget of around AU$36 bil ion, focusing on health
promotion, disease prevention, and services and caregivers for the aged.
During this time, Australia continued on an upward trajectory, using its
wealth to create more health and productivity in its workforce, and its health
and productivity to create more wealth, a virtuous cycle of the highest moral
order.
Between 1970 and 2018, Australian men gained an extra twelve years of life.Their healthy life expectancy is 73 years, ten years higher than the global average,
thanks to a signi cant decline in the percentage of people su ering from
disabling health conditions.18
The elderly in Australia are being less elderly, less of a burden, and much
more productive than other nations. If you visit Australia, the di erence
between its t, active elderly and those in the United States who are saddled with
obesity, diabetes, and disability is noticeable.
My father thought he was headed for the grave. Instead, he’s most often
headed to concerts or the mountains. He spends several nights a week eating out
with friends. He’s adept at computers and new high-tech gadgets and was one of
the rst people in Australia to have a smart speaker with a virtual assistant at
home. He’s unbothered by international travel, so we get to see him frequently.
He’s gone back to work. Physical y and mental y, he is at least thirty years
younger than his mother was at his age.
His remarkable health may or may not be due to the molecules he takes—the
coming years of his life wil be an indicator, whereas scienti c proof wil come
only in the form of double-blind placebo-control ed trials—but he is also helped
by frequent exercise, access to excel ent medical care, and a system that believes in
disease prevention, not just late-stage treatment. He is a shining example of a
new generation of Australians in their 70s and 80s who are not just living longer
but living far better than any of their forebears. In 2018, Australia ranked
seventh on the global Human Capital Index, a measure of the knowledge, skil s,
and health that people in a nation accumulate over their lives, just behind
Singapore, Korea, Japan, Hong Kong, Finland, and Ireland. The United States
ranked twenty-fourth. China ranked twenty- fth.
The trajectory for Australia is up, and the Aussies aren’t looking back.
Having seen what works, other, mostly European countries have adopted
similar health care systems. Australia now has reciprocal agreements with the
United Kingdom, Sweden, the Netherlands, Belgium, Finland, Italy, Ireland,
New Zealand, Malta, Norway, and Slovenia, which means that citizens from
those countries can receive the same medical care in Australia as they can at
home, and vice versa. Imagine an entire world like that.
Meanwhile, some countries are being left behind. And one, in particular, ismoving backward.
Thanks to the burgeoning addiction to calories and opioids, and a health care
system that is inadequate, if not completely inaccessible to one-third of its
population, the United States recently experienced a decline in life expectancy
for the rst time since the early 1960s. That decline may soon exceed the decline
in life expectancy caused by the Spanish u epidemic in 1918. This is happening
despite the fact that the United States spends 17 percent of its GDP on health
care, nearly double that of Australia.
I don’t mean to disparage the country in which I live—it has been very
generous to my family and me. But I am frustrated. Ever since I arrived in the
country that actual y put humans onto the moon, it has been a shock to see
opportunities to help more people for less money wasted over and over again.
The United States has been a leader in both public and private investment
into lifesaving medical research. And although it can be hard to track the origin
of every drug in this increasingly interconnected world, by one estimate 57
percent of al medications are developed in the United States. Other nations,
especial y those that don’t invest as heavily in medical research, should be
grateful to the United States for discovering and developing most of the drugs
that ensure their increasingly long lives.
In a just world, the citizens of the United States would be the greatest
bene ciaries of the medical breakthroughs they subsidize and produce. They’re
not.Australians are. Britons are. As are the Swedes, the Dutch, the Irish, and the
Slovenians. They’re al bene ting in terms of lifespans and healthspans, because
they have the sort of universal access to health care that 15 percent of registered
Democrats and half of al Republicans in the United States have come to fear.19
That the average American lifespan is just four years shorter than Australia’s20
belies the fact that in the poorest regions of the United States citizens live a
decade even shorter than that.21
As the Australian example proves, when everyone is living longer and
healthier, everyone does better. So why isn’t this a topic of discussion in the
United States? Why aren’t people charging Capitol Hil with protest signs and
the proverbial pitchforks, demanding more investment, universal access tomedicines, and the healthiest lifespan on the planet? As other countries enjoy
increasingly longer, healthier lives, perhaps Americans wil wake up and smel
the disparity. But I suspect they won’t. Though the World Health Organization
ranks the United States at number 37, below Dominica, Morocco, and Costa
Rica and one up from Slovenia, 22 it’s stil common to hear US politicians say,
without any justi cation, that the United States has the best health care system
in the world, and mil ions of people believe it. 23
The alternative to a universal right to be treated—regardless of age and
regardless of the ability to pay—is a world in which rich people increasingly
bene t from even longer and even healthier lives than they already enjoy, while
poor people su er through short, disease-ridden existences. This is a terrible idea
for rich and poor alike.
My line of work has put me into contact with some of the wealthiest people
in the world, who are understandably interested in learning the secrets of longer
and healthier lives. I’ve yet to meet a single one who wishes to see such a divide
come to pass. In that direction, after al , lie the seeds of revolution—and revolt
seldom goes wel for the ruling class. As the venture capitalist and “very large
yacht” owner Nick Hanauer wrote in a memo to “My Fel ow Zil ionaires” in
2014, “there is no example in human history where wealth accumulated like this
and the pitchforks didn’t eventual y come out. You show me a highly unequal
society, and I wil show you a police state. Or an uprising. There are no
counterexamples. None. . . . We wil not be able to predict when, and it wil be
terrible—for everybody. But especial y for us.” 24
Hanauer’s warning came before longevity genes were on most people’s radar
and long before most people had so much as contemplated what signi cantly
lengthened lifespans and healthspans could do to the rich-poor divide.
Universal access to technologies that prolong vitality won’t x every problem
associated with income inequality, but it’s a crucial start.
WE SHOULD BE ABLE TO DIE WHENEVER WE WANT TO
By cosmic standards, this region of the Milky Way isn’t a horribly inhospitableplace for life to evolve in. We’re here, after al . And the outer edges of spiral
galaxies like ours seem to hold reasonably good promise for a few life-sustaining
planets to materialize, 25 far better than the dwarf galaxies that are the most abundant type of star systems in the universe.
The way the astronomer Pratika Dayal sees it, however, the most likely places
for life to form and thrive in are the rarer, metal-rich, giant el iptical galaxies—
twice as big as the Milky Way and often much bigger, holding as many as ten
times the number of stars and perhaps 10,000 times as many habitable planets.26
By the way, if you’re under the misconception that if we screw up this planet we
can just travel to a new one, consider that the closest known habitable exoplanet
is twelve light-years away, as the crow ies. That sounds close, but barring the
discovery of a space wormhole or light sailing of tiny cargoes at near light speed,
it would take at least 10,000 years to get a few humans there27 (which, I’ve argued, is another good reason to gure out how to extend human lifespan).
The closest giant el iptical galaxy is Ma ei 1, which is about 10 mil ion light-
years away. We can assume that if explorers from Ma ei 1 ever make the trip to
visit us, they’l be from an exceptional y advanced society. I expect they’l have a
few questions, for they wil want to know how far we have advanced, too.
First, I believe, they’l be curious about the easy things: Have we gured out
pi to a mil ion decimal places? The speed of light? The fact that mass and energy
are the same thing? Quantum entanglement? The age of the universe?
Evolution?
Next they’l ask us about some of the harder stu : Have we learned to use the
resources available on our planet wisely? We’l get passing marks on that one, I
suppose, as long as we don’t mention lead pipes, nuclear bombs, and Furbys.
Have we done so sustainably? “Um, pass.”
Then they’l likely want to hear about what other worlds we’ve visited. “We
sent twelve guys to Luna,” we’l say. “Where’s that?” they’l ask. We’l point to
the big white orb in our night sky. “Hmmm,” they’l say. “Just the men of your
species?” We’l nod, and they’l rol their 146 eyes.
After that, they’l want to know about our lifespan. Have we gured out how
to live far beyond the time given to us by evolution? “Er, we didn’t know that
was a thing worth studying until a few years ago.” They’l o er a bit of overlyenthusiastic encouragement, as a human adult might do for a baby who is
learning to eat solid food.
The next question wil be a rather grave one: “How do you die?” they’l ask.
And how we answer that question is going to be an important indicator of just
how advanced we truly are.
Right now, as my mother’s death exempli ed, the way most of us die is
barbaric. We go through a long period of decline, and we’ve come up with ways
to extend that period of pain, grief, confusion, and fear so that we must
experience even more pain, even more grief, even more confusion, and even
more fear. The sorrow, sacri ce, and turmoil this creates for our families and
friends are protracted and traumatic, so that when we nal y pass on, it often
comes as a relief to those who love us.
The most popular means to the end, of course, are diseases—which can strike
in the prime of life. Heart disease at 50. Cancer at 55. Stroke at 60. Younger-
onset Alzheimer’s at 65. Way too frequently, what is said at funerals is that
someone left this life “way too early.” Or the diseases don’t kil , and the ght to
beat them back again and again is a decades-long exercise in su ering.
These are terrible answers to the question of how we die. The answer we
should strive for—just as much as we strive for prolonged vitality—is “when we
are ready, then quickly and painlessly.”
Fortunately, the science of longevity shows that the longer we make rodents
live, the faster they tend to die. They stil die of the same diseases, but, perhaps
because they are very old, and the animals are on the brink anyway, they tend to
su er for days rather than months, then keel over.
This is not the only way we should meet our end, though.
“Physician-assisted suicide.” “Death with dignity.” “Elective euthanasia.”
Whatever we cal it, we need to end the patchwork of laws and customs that
force people to travel great distances, often when they are already su ering in
one way or many, to bring their lives to a peaceful end.
These are the sorts of barriers that the eminent ecologist David Goodal faced
in 2018, at the age of 104, when he was forced to leave his home in Australia,
where physician-assisted suicide is il egal, and journey to a clinic in Switzerland,
where it is lawful and safe. No one should have to choose between dying in aforeign land and committing a crime as his or her last act on Earth.
Thus no one with a sound mind who is over the age of 40—about the age at
which one has paid back the initial societal investment in his or her education—
should be denied the right to die on his or her own terms. And anyone, at any
age, with a terminal diagnosis or painful chronic il ness should have the same
right.
Yes, there should be rules. Certainly there should be counseling involved and
a waiting period. It should never be easy to take one’s life on a whim rather than
taking arms against a sea of troubles. If it were, I and many others would
probably not have made it through our teenage years. But we should not
presume to leverage guilt and shame upon sane adults who wish to control the
day of their nal breath.
Nearly every day, and often multiple times in a day, someone tel s me that
they have no interest in living to 100, let alone many decades longer.
“If I get to a hundred, just shoot me,” they say.
“I think that seventy- ve healthy years sounds about right,” they say.
“I just can’t imagine having to live with my husband for even longer than I
already have to,” one rather distinguished scientist once told me.
That’s ne.
Indeed, there seems to be little appetite for the idea of living in perpetuity. I
recently gave a talk to a general audience of about a hundred people spread
across ages 20 to 90, a good cross section of the local community. The main
donor to the institute was late, so I had to l in time. I grabbed the microphone
and did a little experiment.
“How long do each of you want to live?” I asked.
By a show of hands, a third said they’d be happy with 80 years; I told that
group that they should al apologize to al the audience members who were older
than 80. That got a laugh.
Another third indicated that they’d like to see 120. “That’s a good goal,” I
said, “and probably not an unrealistic one.”
About a quarter wanted to make it to 150. “That’s not a sil y thing to dream
about anymore,” I said.
Only a few people wanted to live “forever.”The numbers were similar at a recent dinner at Harvard for scientists who
study aging. Very few of the attendees said they were gunning for immortality.
I’ve talked to hundreds of people about this topic. Most people who want
immortality are not afraid of death. They just love life. They love their family.
They love their careers. They would love to see what the future holds.
I’m not a fan of death, either. It’s not because I’m afraid of being dead. I can
say this without reservation. On a plane, my wife, Sandra, clings to my arm at the
rst sign of turbulence, whereas my pulse doesn’t change. I travel enough to have
experienced mechanical troubles on planes more than a few times, so I know
how I react when faced with possible death. If the plane goes down, I die.
Letting go of that fear was one of the best things I ever did.
Here’s where things get real y interesting: when I do this little survey and
then tel the audience that they could retain their health no matter how many
years they live, the numbers of those who say they’d like to live forever shoots
way up. Almost everyone wants that.
It turns out that most people aren’t afraid of losing their lives; they are afraid
of losing their humanity.
And they should be. My wife’s grandfather was sick for many years before he
died in his early 70s. At that point, he’d been in a vegetative state for several years
—a truly horrible fate—but he had a pacemaker, and so, whenever his body
would try to die, he’d be zapped back to life.
Not back to health, mind you. Back to life. There’s a big di erence.
In my mind, there are few sins so egregious as extending life without health.
This is important. It does not matter if we can extend lifespans if we cannot
extend healthspans to an equal extent. And so if we’re going to do the former,
we have an absolute moral obligation to do the latter.
Like most people, I don’t want unlimited years, just ones l ed with less
sickness and more love. And for most of those I know who are engaged in this
work, the ght against aging isn’t about ending death; it’s about prolonging
healthy life and giving more people the chance to meet death on far better terms
—indeed, on their own terms. Quickly and painlessly. When they are ready.
Either by refusing the treatments and therapies that o er a prolonged healthylife or accepting those interventions and then deciding to leave whenever the
time is right, no one who has returned what they have been given should have to
stay on this planet if he or she does not wish to do so. And we need to begin the
process of developing the cultural, ethical, and legal principles that wil al ow
that to happen.
WE MUST ADDRESS CONSUMPTION WITH INNOVATION
The environmental writer and activist George Monbiot is among those who
have observed that when it comes to the future health of our planet, people are
overly preoccupied with the number of humans on Earth while ignoring the fact
that consumption “bears twice as much responsibility for pressure on resources
and ecosystems as population growth. ”28 Monbiot, who is on the far left, isn’t
right about everything, but he’s certainly right about that. The problem isn’t
population; it is consumption.
We know that humans can live healthily and quite happily while consuming
far less than most do in the developed world. But we don’t know if they wil . It is
for this reason that among scientists who subscribe to the idea that our planet
has an absolute limit of people it can sustain, those who have o ered a generous
estimate of the Earth’s carrying capacity are those that assume that our species
wil be capable of making more from less, even, perhaps, as we increase the
standard of living of bil ions of people. The more pessimistic predictors,
meanwhile, general y assume a global “tragedy of the commons” in which we
greedily consume ourselves to death at an al -you-can-eat bu et of natural
resources. General y, people wil be people, so which way we head wil largely be
determined by politics and by technology.
At least in one regard—the “stu factor,” so to speak—technology is already
driving a tremendous and positive change, a global process of
“dematerialization” that has replaced bil ions of tons of goods with digital
products and human services. Thus it is that wal -to-wal shelves dedicated to
records and compact discs have been replaced by streaming music services;
people who once needed vehicles for once-in-a-while travel now open an app ontheir phones to request a ride share; and entire wings of hospitals once used for
storing patients’ records have been supplanted by handheld cloud-connected
tablet computers.
As Steven Pinker has pointed out, a lot of the time, energy, and money we
once spent making “stu ” is now “directed toward cleaner air, safer cars and
drugs for ‘orphan diseases.’ ” 29 Meanwhile, the “experiences, not things”
movements and the like are transforming the ways in which we save and spend
money—and leaving us with less crap in our basements. After a century of
movement toward McMansions, the latter half of the 2010s saw a signi cant
drop in the square footage of new homes and increasing demand for smal er
apartments, 30 continuing a centuries-long migration from farm-based rural
living to smal er, shared urban spaces. As the global success of WeWork proves,
today’s young adults are not only comfortable with much smal er working and
living quarters, with shared community spaces such as o ces, kitchens, gyms,
laundries, and lounges, but increasingly are demanding them.31
The slow death of stu is not the end of consumption, though. We’re as
addicted as ever to wasting food, water, and energy. As it stands, the United
Nations has warned, we are pol uting water far faster than nature can recycle and
purify it. We literal y throw away half of the world’s edible food each year, more
than a bil ion tons of it, even as mil ions of people are left hungry or
malnourished. 32
At the current pace of population growth and economic mobility, the United
Nations estimates, by 2050 it wil take the equivalent of nearly three of our
planet’s resources to sustain our lifestyles for one year. Yet the United Nations
spends surprisingly little time debating consumption, let alone forging
international agreements that would help build a world in which no society
consumes more than its share of what the Earth can produce under
contemporary technological conditions.
That last part is important: just as it is helping us reduce our “stu ”
addiction, technology absolutely has to play a role in solving these other
consumption problems—for there is no free nation in the world that can
unilateral y force its citizens to consume less while others on the planet consume
more. Laws can encourage businesses to conform, but we also have to make itattractive and easy for individuals to consume less.
Therefore, we must invest in research that al ows us to grow more healthy
food and transport it more e ectively. And please make no mistake: that
includes accepting genetical y modi ed crops, those engineered to include a trait
in the plant that doesn’t occur in its wild form, such as resistance to insects,
tolerance to drought, greater vitamin A production, or more e cient use of
sunlight to convert CO2 to sugar—as an absolutely necessary part of our food
future. With more e cient plants, we could feed up to 200 mil ion additional
people, just from plants grown in the US Midwest. 33
These crops have gotten a bad rap for being “unnatural,” although many
people who hold this view don’t recognize that most of the food we think of as
“natural” has already been subject to signi cant genetic manipulation. The ears
of corn you see at the grocery store look nothing like the wild plant from which
modern corn came; over the course of nine thousand years, the spindly nger-
length grass known as teosinte was cultivated to evolve larger cobs and more
rows of plump, soft, sugary kernels, a process of modi cation that signi cantly
altered the plant’s genome.34 The apples we’ve grown accustomed to eating have
a bit more resemblance to their smal , wild ancestors, but good luck nding one
of those ancestors; they have been nearly wiped o the planet, and that’s no great
loss to our diet, since the biggest genetic contributor to modern apples, Malus
sylvestris, is so tart it’s darn near inedible.35
In 2016, the National Academy of Sciences, in a sweeping report on
genetical y engineered crops, noted that lab-modi ed plants could be vital for
feeding the planet’s growing human population if global warming threatens
traditional farm products. And since numerous other reports over the past few
decades had not been enough to assuage continuing public concern, the report’s
authors once again rea rmed the academy’s position that GMO crops are safe
for both human consumption and the environment.
There is nothing wrong with skepticism, but after thousands of studies, the
evidence is irrefutable: if you believe climate change is a threat, you can’t say that
GMOs are, because the evidence that GMOs are safe is stronger than the
evidence that climate change is occurring.
The World Health Organization, the American Association for theAdvancement of Science, and the American Medical Association have also
a rmed that, as WHO puts it, “no e ects on human health have been shown as
a result of the consumption of such foods by the general population.” Moreover,
these foods could be vital to meeting the chal enge of feeding the bil ions of
people who are already going hungry in our world and the additional bil ions
who wil be joining us on this planet in coming years.
If we are to feed the world now and in the future, we need to embrace safe
new technologies.
According to UNICEF, up to 2 mil ion deaths each year could be prevented
if poor families had access to more vitamin A in their diets in crops that are
perfectly safe. 36 Vitamin A supplements aren’t working as wel as is needed.
Between 2015 and 2016, vitamin A supplementation coverage dropped by more
than half in the ve countries with the highest child mortality rates.
An open letter signed by more than a hundred Nobel Prize winners cal ed on
governments to approve genetical y modi ed organisms: “How many poor
people in the world must die before we consider this a ‘crime against
humanity’?” they asked. We could feed a bil ion more people with more
nutritional food. With climate change, we may have no choice.
To decrease the impact of humans, there’s also a tremendous need to gure
out how to satiate the global demand for protein without the tremendous
environmental costs of farmed animal meat. Made with 99 percent less water, 93
percent less land, and 90 percent fewer greenhouse gases, innovations that are
giving us damn-near-close-to-meat products—with plant “leghemoglobin” that
“bleeds” and some good old-fashioned mad science—are booming and wil need
to continue to boom if we are to feed our appetite for tasty protein without
further degrading our planet.
There’s no question that one of the greatest technological advances in this
century has been the discovery of precise, programmable “genome editing.” As
with most other breakthroughs, there were dozens of bril iant people involved in
the lead-up to it, 37 but Emmanuel e Charpentier, then at the Laboratory for
Molecular Infection Medicine in Sweden, and Jennifer Doudna at UC Berkeley
have garnered the most fame for their remarkable discovery that the bacterial
Cas9 protein is a DNA-cutting enzyme with an RNA-based “GPS” or“guide.” 38 The next year, Feng Zhang at MIT and George Church at Harvard
proved that the system could be used to edit human cel s. They, too, garnered
fame—and some very valuable patents.39 News of the discovery spread quickly
down the hal to my lab. It seemed too good to be true—except it was.
The technology is col oquial y known as CRISPR, for “clustered regularly
interspaced short palindromic repeats,” which are the natural DNA targets of
Cas9 cutting in bacteria. Cas9, and now dozens of other DNA-editing enzymes
from other bacteria, can alter plant genes with accuracy, without using any
foreign DNA. They can create exactly the same kind of alterations that occur
natural y. Using CRISPR is far more “natural” than bombarding seeds with
radiation, a treatment that is not banned.
That’s why the decision by the Court of Justice of the European Union in
2018 was so unexpected and upsetting to the United States. The court ruled in
favor of Confédération Paysanne, a French agricultural union that defends the
interests of smal -scale farming, and eight other groups, to ban CRISPR-made
foods.40
The ruling de es science. It bans healthy foods that could relieve the
environmental burden, increase the health of the poor, and al ow Europe to
cope better with global warming. The ruling also scared developing nations away
from CRISPR-modi ed crops; there they could make a positive impact both on
people’s lives and on their land.
The text of the ruling makes it clear that it was not a decision to protect
consumers from the dangers of GMO; it was part of a global trade war to
prevent US-patented products from entering the European Union. The US
secretary of agriculture, Sonny Perdue, made this abundantly clear in his
response: “Government policies should encourage scienti c innovation without
creating unnecessary barriers or unjusti ably stigmatizing new technologies.
Unfortunately, this week’s ECJ ruling is a setback in this regard in that it
narrowly considers newer genome editing methods to be within the scope of the
European Union’s regressive and outdated regulations governing genetical y
modi ed organisms. ”41
Of course nations should be able to help local farmers whose livelihoods arethreatened, but there are other ways to do that. It’s ultimately hurtful to
everyone on the planet to use the cover of “dangerous science” to justify trade
restrictions, especial y to those who need the new technology most.
We also need to solve the shortage of fresh, drinkable water. Cities such as Las
Vegas, a very thirsty town in the middle of the driest place in the United States,
have demonstrated that by marrying conservation and innovation, e cient
water recycling is not only possible but pro table; whereas metro Vegas grew by
half a mil ion people from 2000 to 2016, its total water use fel by a third.
We often adopt new technologies way too slowly, but when we nal y do,
they can solve some of our biggest problems. It was back in 1962 that scientist
Nick Holonyak, Jr., created the rst practical visible light–emitting diode. At
General Electric they cal ed it “the magic one.” It took another half century to
develop an LED house bulb, and even then, many US consumers revolted,
preferring to slow the phase-out of incandescent bulbs even as other nations
moved forward with the LED revolution. Eventual y, a combination of tax
incentives and laws that outlawed the Edison lightbulb forced the adoption of
LED lighting. Today’s LED lights use 75 percent less energy than incandescent
lighting and last fty times as long, which in a typical home is about two
decades.
Widespread use of LEDs in the United States is set to save the equivalent of
the annual output of forty-four large electric power plants, saving about $30
bil ion a year. 42 To put this into perspective, that money could double the
budget of the National Institutes of Health and set forty thousand scientists to
work on lifesaving medicines. Human ingenuity is not a zero-sum game.
Longer, healthier lives wil do us little good if we consume ourselves into
oblivion. The imperative is clear: whether or not we increase human longevity,
our survival depends on consuming less, innovating more, and bringing balance
to our relationship with the bounty of our natural world.
That might seem like a tal order. Indeed, it is a tal order. But I believe we can
stand tal —and together—to meet it.
In many ways, we are already doing so.
At the 2018 Global Climate Action Summit, for instance, it was announcedthat twenty-seven cities had reached peak emission levels. A peak, not a plateau.
Al of those places were seeing steep emission declines. Among that group of
cities was Los Angeles, which was once de nable by its ubiquitous smog. It had
cut its emissions by 11 percent. In one year. 43
Yes, there are more people than ever in the cities of North America, South
America, Europe, and Asia, but today the impact of each human in those
regions is declining. We’re rapidly moving from petroleum to natural gas, solar
power, and electricity. When I rst visited Bangkok, I experienced respiratory
distress. Now, more days than not, there is blue sky. When I arrived in Boston in
1995, a splash of water from the harbor could land you in hospital—or in the
grave. Now it is safe for swimming.44 The same is true of Sydney Harbor, the
Rhine River, and the Great Lakes.
Going backward or even staying put is not a viable solution to the current
crisis. The only path forward is one in which we embrace human capital and
ingenuity.
One of the best examples comes from a tiny town in South Australia. After
the closure of the last coal- red power station in the state in 2016, investors built
Sundrop Farms on the barren coast, then hired 175 people who had recently
become unemployed.45 The farm uses free energy from the sun and seawater to
make 180 Olympic-sized swimming pools’ worth of freshwater per year, an
e ort that in the past would have burned a mil ion gal ons of diesel fuel. Today,
33 thousand pounds of fresh organic tomatoes are shipped each year from the
port where coal used to come in.
Sundrop is an example of a Schumpeterian “gale of creative destruction,” the
type of technological paradigm shift we wil need to usher in the age of longevity
and prosperity. For this to happen, we need more visionary scientists, engineers,
and investors. We need more smart legislation to speed, not impede, the
adoption of Earth-saving technologies. This wil free up money and human
capital that are currently wasted. The freed-up money needs to be reinvested in
people and technologies, not in meaningless “stu ,” to ensure that humanity
and the Earth endure—indeed thrive—together.
WE NEED TO RETHINK THE WAY WE WORKThe University of Pennsylvania was a wonderful school at which to study
theology and the classics. It had recently launched a medical school, too. As a
native Philadelphian, Joseph Wharton was proud of the local col ege. But the
mil ionaire industrialist also believed the university was missing something
essential.
“With industry now powered by steam and steel, we can no longer rely on
apprenticeship alone to create future generations versed in business,” he wrote
to friends and associates on December 6, 1880, just months before o cial y
opening the world’s rst business col ege, the Wharton School. “There needs to
be institutions to instil a sense of the coming strife of the business life and of the
immense swings upward or downward that await the incompetent soldier in this
modern strife. ”46
But Wharton could scarcely have predicted the extent of the “strife” that was
on the horizon: a nascent labor movement in Europe would soon go global,
bringing with it revolutionary changes in workers’ rights.
Among those changes was something that had never existed in the history of
labor: the weekend. We tend to take the ve-day workweek as a given, but it’s an
exceptional y recent innovation. It didn’t exist as a concept—or even a phrase—
until the late 1800s.47 The same can be said of legal limits on daily working hours, the abolition of child labor, medical bene ts, and health and safety
regulations. Al of this was a response to the needs and demands of labor—and,
indeed, the best interests of business owners such as Wharton.
The global Schumpeterian transformation now at hand wil reshape the
world as profoundly as the Industrial Revolution. Every business school in the
world should be preparing its students for what is coming—and labor advocates
should be doing the same. The idea of connecting retirement to a person’s
chronological age wil be an anachronism soon enough. And just like Social
Security, the structures that support labor pensions wil need to be reevaluated.
Skil baticals, which might take the shape of a government-supported paid
year o for every ten worked, might ultimately become cultural and even legal
requisites, just as many of the labor innovations of the twentieth century have.
In this way, those who are tired of “working harder” would be a orded everyopportunity to “work smarter” by returning to school or a vocational training
program paid for by employers or the government, a variation of the universal
basic income that is being discussed in the United States and some countries in
Europe.
Meanwhile, those who believe they are happy and secure in their careers can
enjoy what has come to be known as “a miniretirement”—a year o to travel,
learn a language or musical instrument, volunteer, or refresh and reconsider the
ways in which they are spending their lives.
This is not a particularly crazy scheme; sabbatical leave is common in higher
education. Yet an idea like this might seem ridiculous to those who only consider
the way the world works today. Who would pay for such a bene t? How wil
companies retain workers in the long term without the promise of a “gold watch
retirement” plan at the end of decades of service?
But whoever engages in this discussion now wil have the upper hand when
we decide how to redistribute the resources freed by the elimination of ever-
skyrocketing insurance premiums and pyramid-scheme pensions. Yet few
business professors are so much as thinking about this coming change and even
fewer courses are being taught on the subject in places such as the Wharton
School. Labor leaders, meanwhile, are locked in an understandable but
ultimately futile ght for retirement and bene ts for workers who in the past
would have labored for forty or fty years, retired for a short spel , and then
rather promptly died. Almost no one is ghting over what the world of work
wil look like when age is truly nothing more than a number.
But that era is coming. And it is coming sooner than most people and
institutions realize.
WE NEED TO GET READY TO MEET OUR GREAT-GREAT-
GRANDKIDS
“I sure am glad I won’t be around when that happens.”
I hear this a lot—mostly, it seems, from people who are in, or soonapproaching, retirement. These are folks who have already decided that their
lives are going to end in the next couple of decades. They’re certainly hoping to
stay healthy during that time and maybe eke out a few extra good years if they
can, but they don’t think they’re going to be around for much longer than that.
To them, the middle of this century might as wel be the next mil ennium. It’s
not on their radar.
And that’s the world’s biggest problem: the future is seen as someone else’s
concern.
In part, this stems from our relationship to the past. Very few of us had the
opportunity to get to know our great-grandparents. Many of us don’t even
know their names. That relationship is an abstraction. And so most of us don’t
think about our great-grandkids as much more than a fuzzy, abstract idea.
Sure, we care about the world our children wil live in because we love our
children, but the conventional wisdom about aging and death tel s us they’l be
gone a few decades after we are. And yes, we care about our grandkids, but by
the time they come around, we’re often so close to the exit that it doesn’t seem as
though there’s much we can do about their future anyway.
This is what I want to change—more than anything else in the world. I want
everyone to expect that they wil meet not only their grandchildren but their
great-grandchildren and their great-great-grandchildren. Generations upon
generations living together, working together, and making decisions together.
We wil be accountable—in this life—for the decisions we made in the past that
wil impact the future. We wil have to look our family members, friends, and
neighbors in the eye and account for the way we lived before they came along.
That, more than anything else, is how our understanding of aging and
inevitable prolonged vitality is going to change the world. It wil compel us to
confront chal enges that we currently push down the road. To invest in research
that won’t just bene t us now, but people 100 years from now. To worry about
the planet’s ecosystems and climate 200 years from now. To make the changes
we need to make to ensure that the rich don’t enjoy an increasingly lavish way of
life while the middle class begins to tumble toward poverty. To ensure that new
leaders have a fair and legitimate opportunity to displace old ones. To bring our
consumption and waste into balance with what the world can sustain today andmany centuries into the future.
This isn’t going to be easy. The chal enges are vast. We are not only going to
have to “touch the third rail” of politics—Social Security—but to douse
ourselves in water and lie down upon it, adjusting our expectations about work,
retirement, and who deserves what and when. We’re no longer going to be able
to wait for prejudiced people to die; we’re going to have to confront them and
work to soften their hearts and change their minds. We can’t just al ow the
Anthropocene extinction to continue—at a rate thousands of times higher than
the natural rate—we need to slow it dramatical y and, if we can, stop it
altogether.
To build the next century, we’re going to have to gure out where everyone is
going to live, how they are going to live, under what rules they are going to live.
We’re going to have to ensure that the vast social and economic dividends we
receive from prolonging people’s lives are spent wisely.
We’re going to have to be more empathetic, more compassionate, more
forgiving, and more just.
My friends, we’re going to have to be more human.
CONCLUSIONLET ME TAKE YOU ON a tour of my lab at Harvard Medical School in Boston,
Massachusetts.
You’l nd us in the Genetics Department in the New Research Building,
arguably the best group of biologists in the world. That’s the same place where
Connie Cepko is working to grow mammalian eyes in a dish and studying the
potential for gene therapy to restore lost vision. Down the hal in his clean room,
David Reich, the author and scientist, is sequencing DNA from 20,000-year-old
teeth and discovering that our ancestors liked to breed with other human
subspecies. And a oor down, George Church is working on, among other
wizardries, printing an entire human genome and reviving the wool y
mammoth. Across the street, Jack Szostak has moved on from his Nobel Prize–
winning work to uncover secrets about how life began four bil ion years ago; he
comes by sometimes to visit.
Yeah, the elevator conversations are awesome.
My lab is on the ninth oor. The rst person you’l see when you walk in the
o ce is Susan DeStefano, who basical y has kept our lab and my life under
control for the past fourteen years. Susan is a devout Christian who believes in
the literal version of Genesis. She gures that we are doing God’s bidding by
helping the sick and the needy; there’s no reason why our views on God and
science can’t coincide. We both want to make the world a better place.
To the left of Susan’s doorway you’l nd lab manager Luis Rajman’s o ce.
Luis, who has a PhD in cel ular and molecular biology, ran the transgenic mouse
facility at the giant biotech company Biogen Idec, but when we rst met he was
managing a high-end framing company. He’s worked on paintings worth more
than my house—and probably more than al my neighbors’ houses together, too—so he’s the right sort of guy for a job that requires exceptional meticulousness.
Sitting with her back to Luis is Karolina Chwalek, who has a PhD in
regenerative medicine and is our chief of sta , a strict but fair manager who
makes sure our team of thirty to forty scientists is funded and remains very
much worth funding.
Daniel Vera sits next to Luis and is usual y staring at at least one and often
several screens. He’s the lab’s data guru, having established Florida State
University’s Center for Genomics. I’l never forget the day he showed me the
whole-genome analysis of the epigenetic changes in the ICE mice that helped
reinforce the Information Theory of Aging.
Down the hal , past framed copies of research papers we’ve published, there’s
a sign above a door that says “Operations Room,” as a nod to Winston
Churchil ’s central command. Inside you’l nd the lab and an ever-rotating
group of some of the best minds in the world. When I took a walk through the
lab on one recent day, one of my favorite things to do, these were some of the
characters who were there.
On my left were Israel Pichardo-Casas, a Mexican cel biologist, and Bogdan
Budnik, a Ukrainian physicist, who have found more than ve thousand new
human genes within noncoding “junk DNA.” These smal genes make smal
proteins that course through our bloodstreams, any one of which could be a
treatment to cure cancer, treat diabetes, or be the factor that al ows young mice
to rejuvenate the old. Then there were the Michaels: Bonkowski, Schultz, and
Cooney. Bonkowski played a key role in our study on reversing vascular aging,
making old mice run twice as far. 1 He holds the record for the creation of the
longest-lived mouse in scienti c history, at ve years.
Schultz, his mentee, is studying the molecular events that cause age-related
in ammation, looking for ways to suppress that reaction and thus remove a key
driver of age-aggravated diseases. He and Bonkowski are using gene therapy to
“infect” old mice with longevity genes, aiming to break their own mouse
longevity record.
Cooney is working with NASA to introduce DNA repair genes—from the
supertough, eight-legged micro creatures known as tardigrades—into human
cel s in an e ort to provide astronauts with protection from cosmic radiationand, of course, to slow aging.
There was João Amorim from Portugal; he’s studying resveratrol and a
bunch of STACs in an e ort to understand how they activate SIRT1 in the
body. He’s changed just one base pair in the mouse SIRT1 gene that makes the
enzyme resistant to resveratrol and other STACs. He’s testing if that mutant
mouse stil receives the health and lifespan bene ts of resveratrol. If resveratrol
no longer works on the mutant, then it should resolve the debate about whether
resveratrol is working by directly activating the SIRT1 enzyme, or via some other
mechanism, like activating AMPK. So far, the results look promising for the
SIRT1-activation hypothesis.
There was Jae-Hyun Yang from South Korea; he has spent the past six years
tickling the chromosomes of cel s and animals to understand how and why the
ICE mice age prematurely. It was he and João who rst showed that the
epigenetic clock in the ICE mice ticks faster. Next to him was Yuancheng Lu,
one of China’s top students, who discovered the powerful epigenetic
reprogramming system that can be delivered into aged animals via a modi ed
virus.
Xiao Tian had just used that virus to protect human nerve cel s from
chemotherapy. The normal nerves had either died or shrunk into a bal . But the
reprogrammed ones were completely healthy, with long, beautiful cel ular
projections extending out across the oor of the petri dish. Some experiments
aren’t very conclusive; this was night and day. We plan to test our virus in
patients with eye disorders within a couple of years.
Patrick Gri n, my most recent graduate student, wants to know if
stimulating a response to DNA damage, without creating actual DNA damage,
is su cient to cause aging in mammals. To test this, he has designed a way to
tether DNA damage signaling proteins to the genome using a non-cutting
version of Cas9/CRISPR. If our theory is correct, he should stil cause aging.
Jaime Ross has engineered “NICE mice” to experience accelerated epigenomic
noise just in neurons. She wants to know if the brain controls aging in the rest of
the body, and if these mice operate more like eighty-year-old humans. If so, they
could be used as better models for human brain aging and possibly Alzheimer’sdisease.
Joel Sohn has worked with some of the greatest biologists of the twentieth
century, then spent thirty years as a sherman catching and exporting marine
life, and is now searching the seas for secrets of immortality. He’s studying
cnidarians, transparent ocean animals that can do amazing tricks with their
bodies, such as regrow a new body part or spawn a baby from their feet. That
day was a good day for Joel: his decapitated sea anemone was regrowing a head,
and his immortal jel y sh were budding o baby clones. Perhaps these
regenerative processes are the same as the ones that al ow us to regenerate optic
nerves. Perhaps these creatures have access to the biological equivalent of
Shannon’s observer, the one who stores youthful epigenetic information.
Abhirup Das, who spearheaded the old mouse marathon project, was
studying the impact of precursors such as hydrogen sul de and NMN on
wound healing. Lindsay Wu, who also runs our labs in Sydney, Australia, at the
University of New South Wales, was examining molecules that activate an
enzyme cal ed G6PD, which has been shown to extend the lifespans of multiple
animals and, tragical y, is mutated in 300 mil ion people, the most common of
al mutations. He has also restored fertility in old female mice by feeding them
NMN and protecting their eggs from DNA damage.
There was our resident dental student, Roxanne Bavarian, who is working to
identify the role of sirtuins on oral toxicities and cancer. And there was Kaisa
Selesniemi, from Finland, who is one of the world’s leading experts on culturing
stem cel s from ovaries and reversing female infertility.
Parvez Mohammed, from India, was creating new chemicals in the fume
hood, and Conrad Rinaldi was testing if the latest batch worked to rejuvenate
skin cel s from aged people. Giuseppe Coppotel i, from Italy, was examining
new human longevity genes we’d discovered, including one cal ed Copine2 that
is mutated in Parkinson’s and Alzheimer’s disease patients.
Alice Kane, an Australian, was examining some mice to develop a mouse
frailty clock to predict how long a mouse wil live and is helping al of us see and
appreciate sex di erences. Jun Li, our lab’s senior biochemist, was investigating
why our ability to repair DNA goes down with age and has found that NMNreverses the process. 2
And those were just the people who were in the lab on that speci c day.
There are others—a lot of them—who are doing world-changing work.
These people are bril iant. They could be working on answering any question
in the universe. But they’ve come to Harvard to work on aging. Some of them
are introverts, as scientists often are. A few are cautious, conservative researchers,
a trait I’m working to x. Yet there isn’t a single one who doesn’t believe that
prolonged human vitality is on its way.
And this is just one lab. There are three more labs in the Paul F. Glenn
Center for the Biology of Aging at Harvard that are focused on helping people
live longer and healthier lives. At Bruce Yankner’s lab, they’re exploring the
impact of aging speci cal y on the human brain. Marcia Haigis’s lab is
investigating the role that mitochondria play in aging and disease and has
uncovered the role of sirtuin mutations in cancer. Amy Wagers’s lab was one of
the rst to show the blood of young mice rejuvenates the old, and vice versa,
prompting people to infuse themselves with serum from young donors. Amy
and I are col aborating to nd the factors in the blood and develop new,
advanced pharmaceuticals to treat age-related diseases without the creepiness.
At another Glenn Center, just across the river at MIT, Lenny Guarente,
Angelika Amon, and Li-Huei Tsai are al working on fundamental questions
related to slowing, stopping, and reversing aging. In other cities in the United
States, Thomas Rando, Anne Brunet, Tony Wyss-Coray, Elizabeth Blackburn,
Nir Barzilai, Rich Mil er, and others are al running large labs or centers aimed at
changing the way we think about aging. North of San Francisco there is an
entire building, cal ed the Buck Institute for Research on Aging, completely
devoted to understanding and combating aging. The list goes on and on.
Those are just a few of the labs. Around the world, more than a dozen
independent research centers are working hard on these same questions, and
there is now at least one scientist in every major university in the world who is
working on aging. Most of these labs obtain their research grants for other
diseases but increasingly are turning their attention toward understanding aging,
reasoning that xing that problem wil x whatever disease they’re funded to
ght. This is, after al , an environment in which a huge source of researchfunding is o limits for those who are ghting something that most people
believe is inevitable and few people recognize is a disease.
Meanwhile, private enterprise is leading the way in the development of neural
net-based initiatives for drug discovery and development, gene analysis,
biotracking, and disease detection to dramatical y extend our lives. And every
day research into the simple things anyone can do to prolong their lifespans and
healthspans is mounting, too—o ering better and better maps to good health
and long lives.
A decade or two ago, when even the most optimistic scientists were only just
beginning to envision a world in which aging wasn’t inevitable, and when there
were only a handful of researchers in the world speci cal y working to slow, stop,
or reverse aging, I certainly could understand when people listened politely
about my work and then gave me a look as though I were crazy. Today I have a
hard time understanding how anyone could look at this vast and bril iant army
of researchers and not believe that a tremendous change in human aging is
coming—and soon.
I have some compassion for those who say “It can’t be done.” They are, in my
view, the same kind of people who said vaccines couldn’t work and humans
couldn’t y. But given the bene ts that longevity research can bring to the
world, I have far less patience—indeed, real y none at al —for those who say “It
shouldn’t be done.”
BEYOND BELIEF
There are those who would have you believe that the people in my lab—and
those like them in labs around the world—are engaged in an unnatural and even
immoral campaign to change what it means to be human. That view is rooted in
ideas about human nature that might charitably be described as subjective but
that are probably more accurately cal ed zealotry.
That, it seems to me, was the guiding force behind a 2003 report submitted
to the White House by the President’s Council on Bioethics entitled Beyond
Therapy: Biotechnology and the Pursuit of Happiness, which ominously warnedagainst aging research because it goes against “the human grain” and violates the
purportedly orderly cycle of birth, marriage, and death.
Would people “be more or less inclined to swear lifelong delity ‘until death
do us part,’ if their life expectancy at the time of marriage were eighty or a
hundred more years, rather than, as today, fty?” 3 the council wondered. What
kind of unhappy marriages, I wondered in return, would drive people to even
ask such a question? I would love to have an extra fty years with my wife,
Sandra.
Aging, the council pro ered, “is a process that mediates our passage through
life, and that gives shape to our sense of the passage of time”; without it, the
council’s members cautioned, we could become “unhinged from the life cycle.” 4
Our so-cal ed natural life cycle, of course, is one in which the vast majority of
our ancestors never got old enough to get gray hair or wrinkles and in which
consumption by carnivore was a perfectly ordinary way to go. If you’d like to be
hinged to that, be my guest.
“Might we be cheating ourselves,” the council asked, “by departing from the
contour and constraint of natural life (our frailty and nitude), which serve as a
lens for a larger vision that might give al of life coherence and sustaining
signi cance? ”5
Oh, for goodness’ sake, if we truly believed that frailty was a requisite for
meaningful life, we’d never mend a broken bone, vaccinate against polio, or
encourage women to stave o osteoporosis by maintaining adequate calcium
levels and exercising.
I know I shouldn’t get worked up about these sorts of things. It’s a tale as old
as science, after al —just ask Galileo what happens when you “disrupt the
natural order of things.”
But this was more than a tri ing report from moralizing bureaucrats. The
chair of the committee that wrote it, Leon Kass, is one of the most in uential
bioethicists of our time and came to be known, during the tenure of George W.
Bush, as “the President’s Philosopher.” For years after the report was issued,
aging research was framed not as a ght against a disease but as a ght against
our humanity. That’s hogwash, and, in my mind, it’s rather deadly hogwash.
Yet once the framing has been set, the e ort to shift ideas, understandings,and biases becomes a herculean one. The ght to help people see aging for what
it is, rather than “just the way it goes,” wil be a long one.
More funding for the kind of research happening in my lab and others like it
could bring about these advancements even sooner. But because of a lack of
funding, people over sixty today may not live long enough to be helped. If you
and your family members end up the last of humanity to live a life that ends al
too early with decay and decrepitude, or our children never see the bene ts of
this research, you can thank those bioethicists.
After al of these arguments, if you stil think that extending the healthy part
of your life would not be for you—perhaps it would reduce your life’s urgency
or be against the natural course of things—consider your friends and family.
Would you subject your loved ones to a decade or two of unnecessary hardship
having to look after you physical y, emotional y, and nancial y in your nal
years if you didn’t have to?
Spend a day in a nursing home like my wife does every few days. Go feed
people who can’t chew. Wipe their bottoms. Bathe them with a sponge. Watch
as they struggle to remember where they are and who they are. When you are
done, I think you wil agree that it would be negligent and cruel for you not to
do what you can to combat your own age-related deterioration.
There are stil a lot of people like Kass out there. But if they live long enough,
they wil also have to come to terms with reality. The momentum makes the
future I have described, or one close to it, unstoppable. Prolonged healthspans
are inevitable.
More and more people recognize this every day, and they want in.
Because no matter what people say or believe, whether they are optimists or
scaremongers, scientists or bioethicists, there is change in the air.
On June 18, 2018, WHO released the eleventh edition of the International
Classification of Diseases, known as ICD-11. It is a fairly unremarkable
document, except that someone slipped in a new disease code. At rst no one
saw it. Here is the line, which you can nd on the WHO website6 if you type in
code MG2A. It reads:
MG2A Old age• old age without mention of psychosis
• senescence without mention of psychosis
• senile debility
Every country in the entire world is encouraged to start reporting using ICD-
11 on January 1, 2022. What this means is that it is now possible to be diagnosed
with a condition cal ed “old age.” Countries wil have to report back to the
WHO with their statistics on who dies from aging as a condition.
Wil this lead to changes at the regulatory level, directing bil ions of dol ars in
investment to develop the medicines we deserve? Wil federal regulators and
doctors nal y accept that it is ethical y okay to prescribe medicines to slow aging
and al the diseases that aging causes? Wil they recognize it is indeed within a
patient’s rights to receive them? Wil insurance companies reimburse patients for
the cost of antiaging treatments that wil save money down the line?
We wil see. I certainly hope the winds build. Until that time comes, though,
there is plenty we can do.
WHAT I DO
Save for “Eat fewer calories,” “Don’t sweat the smal stu ,” and “Exercise,” I
don’t give medical advice. I’m a researcher, not a medical doctor; it’s not my
place to tel anyone what to do, and I don’t endorse supplements or other
products.
I don’t mind sharing what I do, though, albeit with some caveats:
• This isn’t necessarily, or even likely, what you should do.
• I have no idea if this is even the right thing for me to be doing.
• While human trials are under way, there are no treatments or therapies for
aging that have been through the sort of rigorous long-term clinical testing
that would be needed to have a more complete understanding of the wide
range of potential outcomes.
People often wonder, when I tel them things like this, why on earth I would
subject myself to the potential for unexpected and adverse side e ects or even the
possibility—low though it seems to be—that I could expedite my own demise.The answer is simple: I know exactly what is going to happen to me if I don’t
do anything at al —and it’s not pretty. So what do I have to lose?
And so, with al that on the table, what do I do?
• I take 1 gram (1,000 mg) of NMN every morning, along with 1 gram of
resveratrol (shaken into my homemade yogurt) and 1 gram of metformin.7
• I take a daily dose of vitamin D, vitamin K2, and 83 mg of aspirin.
• I strive to keep my sugar, bread, and pasta intake as low as possible. I gave
up desserts at age 40, though I do steal tastes.
• I try to skip one meal a day or at least make it real y smal . My busy
schedule almost always means that I miss lunch most days of the week.
• Every few months, a phlebotomist comes to my home to draw my blood,
which I have analyzed for dozens of biomarkers. When my levels of various
markers are not optimal, I moderate them with food or exercise.
• I try to take a lot of steps each day and walk upstairs, and I go to the gym
most weekends with my son, Ben; we lift weights, jog a bit, and hang out in
the sauna before dunking in an ice-cold pool.
• I eat a lot of plants and try to avoid eating other mammals, even though
they do taste good. If I work out, I wil eat meat.
• I don’t smoke. I try to avoid microwaved plastic, excessive UV exposure, X-
rays, and CT scans.
• I try to stay on the cool side during the day and when I sleep at night.
• I aim to keep my body weight or BMI in the optimal range for healthspan,
which for me is 23 to 25.
About fty times a day I’m asked about supplements. Before I answer, let me
say that I never recommend supplements, I don’t test or study products, nor do
I endorse them; if you see a product implying that I do, it’s certainly a scam.
Supplements are far, far less regulated than medicines, so if I do take a
supplement, I look for a large manufacturer with a good reputation, seek highly
pure molecules (more than 98 percent is a good guide), and look for “GMP” on
the label, which means the product was made under “good manufacturing
practices.” Nicotinamide riboside, or NR, is converted to NMN, so some people
take NR instead of NMN because it is cheaper. Cheaper stil are niacin and
nicotinamide, but they don’t seem to raise NAD levels as NMN and NR do.
Some people have suggested NAD boosters could be taken with a compoundthat provides cel s with methyl groups, such as trimethylglycine, also known as
betaine or methylfolate. Conceptual y, this makes sense—the “N” in NR and
NMN stands for nicotinamide, a version of vitamin B3 that the body methylates
and excretes in urine when it is in excess, potential y depleting cel s of methyls—
but this remains a theory.
My father fol ows almost the same regimen as I do, and I can’t remember the
last time he was sick. He claims he’s speeding up. This summer, he left his busy
social calendar behind in Australia, and, having helped us with home repairs in
Boston for six weeks while working remotely in his second career at the
University of Sydney, he then drove around the US East Coast for a few weeks
with his lifelong friend on their annual pilgrimage to the Summer Theater
Festival in Wooster, Ohio.
Dad ew home at the end of summer, only to come back a few weeks later to
see me get “knighted,” as he cal ed it, in Washington, DC. Now that he’s home
in Sydney again, he’s planning on driving six hundred miles north for a few days
to “see a couple of friends.” He is loving life, seemingly more than he ever has.
As I get older, I spend more and more time thinking how lucky I’ve been in
life. As an Aussie, I was taught that “big boys don’t cry.” But, nowadays, when I
have the time and the sense to pause for a few moments to ponder my life, it’s
easy to get a little teary.
I grew up in a free country, then moved to an even freer one. I have three
amazing children and friends who treat my family like their own. I am very
proud of Sandra, my wife, who was one of the top students in Germany. She
aced her botany degree, then came to Boston to be with me, got into the PhD
program at MIT, and worked in a lab that was cloning mice for the rst time. To
earn her PhD, she gured out how to cure mice of a lethal genetic disease cal ed
Rett syndrome that disrupts the epigenome and prevents brain development in
infant girls. By strange coincidence, the gene she worked on, MECP2, binds to
methylated DNA and might be a cel ular observer that stores youthful age
correction data.
Sandra has been teaching me a lot these past twenty- ve years about how to
be a better husband and parent, not to mention the names of al the plants,
insects, and animals we see on our walks. When we were rst married, we argueda lot. She had “ethical issues” with my research, which pained me. Now, having
examined and discussed the wealth of biological and economic data over the
years, we no longer argue as much, and in fact she’s started taking NMN.
It’s impossible to say if my regimen is working for us, but it doesn’t seem to
be hurting. I am now 50, and I feel the same as I did when I was 30. My heart
looks 30, too, according to a video of my heart in 3D that one of my col eagues
kindly made by inserting me into an experimental magnetic resonance imager. I
don’t have a gray hair, and I’m not superwrinkly—wel , at least not yet.
A year ago, my younger brother, Nick, was going gray and losing his hair
when he demanded to be put on the same regimen after accusing me, only half
jokingly, of using him as a negative control. I insisted that I would never do that
to my own brother, but I can’t say the thought hadn’t crossed my mind. He’s
now on my dad’s regimen, too.
Living longer makes no sense if you don’t have your friends and family
around you. Even our three dogs—a smal 10-year-old poodle cross named
Charlie and two 3-year-old black Labradors, Caity and Melaleuca—have been
on NMN for a couple of years. Charlie is a therapy dog whose job it is to calm
people, but he becomes too hyperactive if Sandra gives him NMN the day he
heads to work, so on those days he’s o it. Caity su ers from a congenital kidney
defect, and we hope NMN wil al ow her to make it past her predicted ve-year
expiry date. The results of tests in mice with kidney damage say it’s possible.8
A lot of people think a regimen intended to promote prolonged vitality must
be hard to stick to, but if it were, my family couldn’t do it. We are just an average
bunch trying to get through the day. I do live life as mindful y as possible, focus
on feeling good, and check my blood markers occasional y. Over time, I’ve
identi ed the diet, exercise, and supplement routines that work best for me. And
I’m con dent that my family and I wil continue to ne-tune these practices in
response to the evolving research as our lives go on.
And on.
And on.
Because, yes, I do hope to be here for a long time to come. There are plenty of
X factors that could interfere with that goal. I could get hit by a bus tomorrow,

after al . But it’s getting easier and easier to imagine being around—happy,
healthy, and connected to friends, family members, and col eagues—past my
100th year.
How long past my 100th year?
Wel , I think it would be nice to see the twenty-second century. That would
mean making it to my 132nd year. To me, that is a remote chance but not
beyond the laws of biology or way o our current trajectory. And if I do make it
that far, perhaps I’l want to stick around even longer.
There’s so much I want to do—and so many people I’d like to help. I’d love
to keep nudging humanity down what I believe is a path to greater health,
happiness, and prosperity, and to live long enough to know what path we take.
BUSHWALKING. If you head north from my childhood home, you’l move through hundreds of
miles of consecutively larger national parks, a seemingly endless undulation of saltwater estuaries
and craggy mountain ridges decorated by ancient rock carvings left by the original inhabitants,
the Garigal clan. Dad is now 80, the age his mother, Vera, was when she lost the wil to live—
aging has that e ect on people. Instead, my father hikes mountains, travels the world, and has
started a new career, representing hope for al of us.
BUSHWALKINGI recently returned to the neighborhood where I grew up, in the northern
suburbs of Sydney, on the edge of Garigal National Park. Dad and Sandra were
both there, and so was my twelve-year-old son, Benjamin.
We’d come for a hike along the trail, the exact same one my grandmother Vera
used to take my brother and me on when we were that age. She’d tel us stories
about her di cult childhood, about how lucky we were to have grown up in a
free country, and about the wisdom of A. A. Milne:
“What day is it?” asked Pooh.
“It’s today,” squeaked Piglet.
“My favorite day,” said Pooh.
Dad was raring to go. Ben was, too. Tigers, those boys are. But as I stood at
the beginning of the trail, on the edge of a high sandstone cli overlooking a
gul y l ed with ambrosial eucalyptus and with deafening cicadas overhead, I
found myself frozen in awe by the way in which the city quickly gave way to the
bush, how the present and the deep past came together, and how it felt to be on
the edge of something so vast and so beautiful.
If you wind your way south, down the rocky trail leading o Melaleuca
Drive, the street I lived on as a boy, you’l reach Middle Harbor, an estuary lined
by a canopy of bloodwoods, angophoras, and scribbly gum trees that ends in
Sydney Harbor. If instead you head north, you’l move through hundreds of
miles of consecutively larger national parks: Garigal to Ku-ring-gai to
Marramarra, Dharug to Yengo to Wol emi, a seemingly endless undulation of
saltwater estuaries and craggy mountain ridges decorated by ancient rock
carvings. You could walk for days, even for weeks, and not hear anyone, save for
the distant echoes of the original inhabitants of this land.
On that day in Garigal Park we were planning on walking only a few hours,
but I’d been looking forward to it for weeks.
There is, at least to me, a subtle but important di erence between hiking and
bushwalking. When people hike, they are most often looking for exercise,
serenity, beauty, or time together with loved ones. When Australians go for a
bushwalk, they are seeking al of those things but with the intention of ndingwisdom, too.
I’m not sure how long I was standing on the cli . A minute or two, perhaps.
Five or ten, maybe. However long it was, my family didn’t seem to mind. When
the spel of nostalgia and wonderment released me, I found them a short way
down the trail.
Ben was peeling the paperlike bark o a melaleuca tree, while Dad was trying
to explain something to him about the cli s being made of sand that had been
deposited when mammals rst appeared. Sandra was examining a banksia—the
strange, prickly ower Sir Joseph Banks col ected to show the Royal Society—
which, she delighted in reminding us, for the umpteenth time, is a member of
the Proteacea family.
As I write this, Ben is in seventh grade. He’s a good boy. A smart boy. He
wants to work in my lab someday and take over from me to “ nish the job.” I tel
him he’l have a lot of competition and won’t get any special treatment from me,
and he says, “Wel , if that’s true, I can always work for Lenny Guarente.”
Yes, he’s a funny boy, too.
Our two older children are making their own paths: Natalie, I reckon, as a
veterinarian; Alex, perhaps, as a diplomat or a politician.
Dad is now 80, the age his mother, Vera, was when the re in her eyes had
completely gone. She had lost the wil to live and never ventured outside again. I
cannot predict the future, but when I look at the ful life Dad leads now, his
world travel, his optimism, and the state of his health, I think he’l be around for
a long time to come. I sure hope he wil .
Not just because he represents hope for al of us, but because I’d like to come
back to this place again and again with Dad and Sandra and al those I love.
Searching for serenity. Hearing stories. Finding beauty. Making memories.
Sharing wisdom.
With Ben and Natalie and Alex, yes. But also with their children. And with
their children’s children.
Why not? Nothing is inevitable.
AcknowledgmentsDAVID
I cannot begin to express my love and gratitude to my wife, Sandra Luikenhuis,
who has stood by me for two decades and tolerated my writing and rewriting
this book for one of them. To my children, Alex, Natalie, and Benjamin: I could
not have asked for better o spring.
Writing a book requires considerable kinship among al those involved in the
creative process. I’m indebted to Matthew LaPlante for his friendship, sense of
humor, and polymath’s ability to turn hundreds of discussions and dozens of
whiteboard diagrams into a coherent narrative. Matt and I are both so lucky to
have worked with Caity Delphia, this book’s masterful and veracious il ustrator,
who bravely took on the chal enge to turn our words and ideas into stunning
works of art—and pul ed it o . Every day I’m thankful for the camaraderie of
Susan DeStefano, my assistant for the past fourteen years who keeps my life and
our labs running smoothly; she deserves an entire page of thanks for her ability
to handle al that’s thrown at her.
I’m indebted to Luis Rajman and Karolina Chwalek, who help run the
research lab in Boston, and Lindsay Wu, who runs our sister lab in Sydney. I’m
fortunate to work with such a dedicated, intel igent, and practical team. Bruce
Yankner, my co-director at Harvard’s Glenn Center for Aging Research, has
been a wonderful col aborator and col eague.
My deep and heartfelt thanks to Celeste Fine, John Maas, and Laurie
Bernstein, my agents; to Sarah Pelz, our editor, for her careful and skil ful
editing; to Melanie Iglesias Pérez and Lisa Sciamba; to Lynn Anderson for
copyediting; and al the sta at Simon & Schuster who believed in this book.
Thanks to Laura Tucker for beginning this journey a decade ago; to the publicrelations team Carrie Cook, Sandi Mendelson, Rob Mohr, and Nicholas Platt.
Matt and I are so thankful to everyone who read and made suggestions to
improve the manuscript, especial y Stephen Dark, who coedited the glossary and
endnotes, Mark Jones, Sandra Luikenhuis, Mehmood Kahn, John Kempler, Lise
Kempler, Tristan Edwards, Emil and Dariel Liathovetski (the RockCel os), Dave
Deamer, Terri Sinclair, Andrew Sinclair, and Nick Sinclair. My appreciation to
Brigitte Lacombe, master photographer, for the unkempt headshot (Instagram
brigittelacombe).
Thanks to al the teams that work tirelessly to make this world a better place,
including and in order of incorporation, CohBar, Vium, InsideTracker,
MetroBiotech, Arc Bio, Liberty Biosecurity, Dovetail Genomics, Life
Biosciences, Continuum Biosciences, Jumpstart Fertility, Senolytic
Therapeutics, Animal Biosciences, Spotlight Therapeutics, Selphagy
Therapeutics, and Iduna Therapeutics.
When I chose to be a scientist, I thought that the greatest reward would be
discovering things, but it’s actual y the lifelong friends you make, the ones who
stand up for you when times are tough. So I am grateful to have the friendship
and receive the wise counsel of Nir Barzilai, Rafael de Cabo, Stephen Helfand,
Edward Schulak, Jason Anderson, Todd Dickinson, Raj Apte, Anthony Sauve,
David Livingston, Peter El iott, Darren Higgins, Mark Boguski, Carlos
Bustamante, Tristan Edwards, Lindsay Wu, Bruce Ksander, Meredith Gregory
Ksander, Zhigang He, Michel e Berman, Pinchas “Hassy” Cohen, Mark Tatar,
Alice Park, Sri Devi Narasimhan, Kyle Landry, James Watson, David Ewing
Duncan, Joseph Maroon, John Henry, Duncan Purvis, Li-Huei Tsai, Christoph
Westphal, Rich Aldrich, Michel e Dipp, Bracken Darrel , Charles de Portes,
Stuart Gibson, Adam Neumann, Adi Neumann, Ari Emanuel, Vonda Shannon,
Joel and Cathy Sohn, Alejandro Quiroz Zarate, Mathilde Thomas, Bertrand
Thomas, Joseph Vercauteren, Nicholas Wade, Karen Weintraub, Jay Mitchel ,
Marcia Haigis, Amy Wagers, Yang Shi, Raul Mostoslavsky, Tom Rando, Jennifer
Cermak, Phil Lambert, Bruce Szczepankiewicz, Ekat Peheva, Matt Easterday,
Rob Mohr, Kyle Meetze, Joanna Schulak, Ricardo Godinez, Pablo Costa,
Andreas Pfenning, Fernando Fontove, Abraham Solis, Jaques Estaban, Carlos
Sermeño and the entire C3 team, Peter Buchthal, Mark Tatar, Dean Ornish,Margaret Morris, Peter Smith, David Le Couteur, Thomas Watson, Kyle
Landry, Meredith Carpenter, Margaret Morris, Steven Simpson, Mark Sumich,
Adam Hanft, David Chin, Jim Cole, Ed Green, Phil Lambert, Shal y Bhasin,
Lawrence Gozlan, Daniel Kraft, Mark Hyman, Marc Hodosh, Felipe Sierra,
Michael Sistenic, Bob Kain, David Coomber, Ken Rideout, Bob Bass, Tim Bass,
John Monsky, Jose Morey, Michael Bonkowski, David Gold, Matt Westfal , Julia
Dimon, Richard Hersey, Joe Hockey, Bjarke Ingels, Margo McInnes, Joe Rogan,
Mhairi Anderson, Lon Augustenborg, Mike Harris, Sean Riley, Greg Keeley, Ari
Patrinos, Andy, Henny, Ian, Josh, and al the other special people who have
served and risked their lives to make the world a better place. To everyone I’ve
worked with over the years: thank you for the encouragement and inspiration to
keep working on this book. I am deeply indebted to those who took the time to
be my mentors: my grandmother Vera; my father, Andrew; my mother, Diana;
my uncle and aunt Barry and Anne Webb; my PhD mentors, Ian Dawes,
Richard Dickinson, and Je Kornfeld; my postdoc mentor, Lenny Guarente;
and my Harvard mentors, Peter Howley, George Church, and Cli Tabin, and
everyone who’s stood up for and supported our research.
My lab and its research would not have been possible without the support of
grants from the Helen Hay Whitney Foundation, an Australian graduate
fel owship, the US National Institutes of Health, the National Health and
Medical Research Council of Australia, Mark Col ins, Leonard Judson and
Kevin Lee at the Glenn Foundation for Medical Research, the American
Federation for Aging Research, Caudalie, Hood Foundation, Leukemia and
Lymphoma Society, Lawrence El ison Medical Foundation, Hank and Elenor
Rasnow, Vincent Giampapa, and Edward Schulak. I’m so very grateful to the
hundreds of donors, both large and smal , who have contributed to our lab’s
research.
And nal y, I cannot begin to express my gratitude for the vision, wisdom,
and kindness of Paul Glenn, whose funding of aging research wil change the
world.
MATTHEWWhile I truly value my professional partnership with David, I absolutely treasure
our friendship, and I am so grateful to have him in my life. I am also deeply
appreciative of Sandra, Alex, Natalie, and Ben Sinclair, who always treated me
like family when I was in Boston, and of Susan DeStefano, who always greeted
me with a hug at Harvard. I am in debt to the researchers in David’s lab and to
the leaders and employees of the companies in which David is involved for their
kindness and patience during my visits. I would not have known any of these
amazing people if not for my truly amazing agent, Trena Keating. Most of al , I
am grateful to my wife, Heidi, and our daughter, Mia, who lifted me up through
the simultaneous writing of two books.
Sinclair DisclosureDr. Sinclair is committed to turning key discoveries into medicines and technologies that help
the world. He is involved in a variety of activities beyond being an academic including being a
founder, equity owner, adviser, member of the board of directors, consultant, investor,
col aborator with, and inventor on patents licensed to companies working to improve the human
condition. These include Vium; CohBar; Galileo Bioscience; Wel omics; EdenRoc Sciences and
its a liates Arc Bio, Dovetail Genomics, Claret Medical, Revere Biosciences, UpRNA,
MetroBiotech, and Liberty Biosecurity; and Life Biosciences and its a liates Selphagy
Therapeutics, Senolytic Therapeutics, Spotlight Therapeutics, Lua, Animal Biosciences, Iduna,
Continuum Innovation, Prana (now Alterity); and Jumpstart Fertility. He is an inventor on over
forty patents, most of which are licensed to industry or have been led by companies, including a
patent application led by Mayo Clinic and Harvard Medical School and licensed to Elysium
Health, of which any proceeds to him are donated to research. He gives lectures at conferences,
museums, not-for-pro t events, and occasional y at companies, and he sits on the boards of not-
for-pro t organizations, including the American Federation for Aging Research. He also serves
as an adviser to the Lorraine Cross Award. For an updated list of activities, see
https://genetics.med.harvard.edu/sinclair/.
The Scale of Things1 grain of sand = 10 skin cel s
0.5 mil imeter
1 skin cel = 5 blood cel s
50 micrometers
1 blood cel = 2 X chromosomes or ~2 yeast cel s
10 micrometers
1 X chromosome = 1 yeast cel = 10 E. coli
5 micrometers
1 E. coli or mitochondrion = 2 M. superstes
0.5 micrometer
1 M. superstes = 4 ribosomes
0.25 micrometer
1 ribosome = 6 catalase enzymes
30 nanometers
1 catalase enzyme = 5 glucose molecules
5 nanometers
1 glucose molecule or amino acid = approximately 4–6 water molecules
1 nanometer
1 water molecule = 275,000 atomic nuclei
0.275 nanometer
1 atomic nucleus
1 picometer
1 inch = 25.4 mil imeters
1 foot (12 inches) = 0.3048 meter
1 yard (3 feet) = 0.9144 meter
1 mile = 1.6093 kilometers
1 mil ion = 106 (1 with 6 zeros)
1 bil ion = 109 (1 with 9 zeros)
1 tril ion = 1012 (1 with 12 zeros)
mil i = 10-3 (1 thousandth)
micro = 10-6 (1 mil ionth)nano = 10-9 (1 bil ionth)
pico = 10-12 (1 1,000 bil ionth, or a tril ionth)
32°F = 0°C
212°F = 100°C




Cast of Characters
JOSEPH BANKS (February 24, 1743–June 19, 1820): English naturalist, botanist, and former
president of the Royal Society who accompanied Captain James Cook on his voyage round the
world. With Lord Sydney a staunch advocate of starting a colony in Australia at Botany Bay on
Cape Banks. Namesake of the ower cal ed the Banksia.
NIR BARZILAI (December 23, 1955–): Israeli-born American endocrinologist and professor at
the Albert Einstein Col ege of Medicine in New York best known for his work to elucidate genes
that enable members of Ashkenazi families to live over 100, hormones that control lifespan, and
the e ects of metformin on lifespan.
ELIZABETH BLACKBURN (November 26, 1948–): an Australian American Nobel laureate who,
with Carol W. Greider and Jack W. Szostak, discovered telomerase, the enzyme that extends
telomeres. In 2004, she was controversial y dismissed from the Bush administration’s President’s
Council on Bioethics, al egedly for her advocacy of stem cel research and politics-free scienti c
enquiry.




ARTHUR C. CLARKE (December 16, 1917–March 19, 2008): British science ction writer and
futurist known as the “Prophet of the Space Age.” Spent most of his adult life in Sri Lanka
foreseeing the advent of space travel and satel ites. Advocate for protection of goril as. Polio in
1962 led to postpolio syndrome.
ALVISE (LUIGI) CORNARO (1464 or 1467–May 8, 1566): Venetian nobleman and patron of arts
who wrote four books of Discorsi about the path to health and longevity that included fasting
and sobriety.
EILEEN M. CRIMMINS: American demographer at the University of Southern California who
was the rst to combine indicators of disability, disease, and mortality to predict healthy life expectancy. She showed that the prevalence of dementia in women stems largely from their
longer life.
RAFAEL DE CABO (January 20, 1968–): Spanish-born scientist at the National Institutes of
Health, an expert in the study of the e ects of diet on health and lifespan in rodents and
primates.
BENJAMIN GOMPERTZ (March 5, 1779–July 14, 1865): British self-educated mathematician
who is best known for the Gompertz-Makeham Law of Human Mortality, a demographic model
(1825). He became a Fel ow of the Royal Society and then an actuary at Al iance Assurance
company, founded by his brother-in-law Sir Moses Monte ore with his relative Nathan Mayer
Rothschild.





LEONARD P. GUARENTE (June 6, 1952–): American molecular biologist and professor at MIT,
best known for codiscovering the role of the sirtuins in aging and the necessity of NAD+ for
sirtuin activity, linking energy metabolism to longevity.
ALEXANDRE GUÉNIOT (1832–1935): Centenarian and French physician who wrote the book
Pour vivre cent ans. L’Art de prolonger ses jours (To Live a Century). He attributed great signi cance to the “hereditary vital force” that he suggested determines the natural duration of
human life at no less than 100 years.
JOHN B. GURDON (October 2, 1933–): British biologist who in 1958 cloned a frog using a
nucleus from an adult tadpole’s cel , demonstrating that aging can be reset, for which he shared
the Nobel Prize with Shinya Yamanaka in 2012.
DENHAM HARMAN (February 14, 1916–November 25, 2014): American chemist who
formulated the “Free Radical Theory of Aging” and the “Mitochondrial Theory of Aging.”
Harman was a founder of the American Aging Association, ran two miles a day until he was 82,
and eventual y died at the age of 98.
LEONARD HAYFLICK (May 20, 1928–): American biologist who invented the inverted
microscope; best known for his 1962 discovery that normal mammalian cel s have a limited
capacity for replication. The Hay ick limit on cel division overturned a long-held belief





promulgated by the French surgeon and biologist Alexis Carrel in the early twentieth century
that normal cel s in culture would proliferate continuously.
STEVE HORVATH (October 25, 1967–): Austrian-born American professor at the University of
California at Los Angeles known for his pioneering work on epigenetics and aging and for
codeveloping algorithms that predict the age of organisms based on DNA methylation patterns,
known as the Horvath aging clock.
SHIN-ICHIRO IMAI (December 9, 1964–): Japanese-born American biologist known for his
Heterochromatin Hypothesis of Aging, his work on mammalian sirtuins, and the discovery with
Lenny Guarente that sirtuins need NAD+ for their activity.
CYNTHIA J. KENYON (February 21, 1954–): American geneticist who showed that Daf-2
mutations double nematode worm lifespan, after studying under Nobel Prize winner Sydney
Brenner using nematodes as a model organism. Kenyon is a professor at the University of
California, San Francisco, and vice president of aging research at Calico.
JAMES L. KIRKLAND: American physician and biologist working at the Mayo Clinic in
Rochester, New York; a pioneer in the study of senescent “zombie” cel s and the development of
drugs cal ed senolytics that kil them.




THOMAS B. L. KIRKWOOD (July 6, 1951–): South African–born biologist and associate dean
for ageing at Newcastle University, UK. Proposed the Disposable Soma hypothesis, the idea that
species aim to balance energy and resources between reproduction and building a robust, long-
lasting body.
PIERRE LECOMTE DU NOÜY (December 20, 1883–September 22, 1947): French biophysicist
and philosopher who noticed that the wounds of older soldiers healed more slowly than those of
younger ones. His “tele nalist” hypothesis that God directs evolution was criticized as
unscienti c.
CLIVE M. McCAY (March 21, 1898–June 8, 1967): American nutritionist and biochemist who
spent decades at Cornel University researching the soybean and our. Best known for his early
work con rming that calorie restriction extends the lifespan of rats. In 1955, he and his wife published “You Can Make Cornel Bread.”
PETER B. MEDAWAR (February 28, 1915–October 2, 1987): British biologist born in Brazil
whose work on graft rejection and the discovery of acquired immune tolerance was fundamental
to the practice of tissue and organ transplants. Realized the force of natural selection declines
with age due to reduced “reproductive value.”
ARTHUR PHILLIP (October 11, 1738–August 31, 1814): British admiral of the Royal Navy and
rst governor of New South Wales who sailed to Australia to establish the British penal colony in
Botany Bay that later, after moving one harbor north, became the city of Sydney, Australia.





CLAUDE E. SHANNON (April 30, 1916–February 24, 2001): American mathematician and
engineer who worked at MIT and is known as the “father of information theory.” His paper “A
Mathematical Theory of Communication” (1948) solved problems of information loss and its
restoration, concepts that laid the foundation for the TCP/IP protocols that run the internet.
His hero was Thomas Edison, who he later learned was his relative.
JOHN SNOW (March 15, 1813–June 16, 1858): English anesthesiologist and leader in the
adoption of anesthesia and medical hygiene; best known for his work tracing the source of a
cholera outbreak arising from the Broad Street pump in Soho, London, in 1854.
LEO SZILARD (February 11, 1898–May 30, 1964): Hungarian-born American physicist and
humanist who proposed the DNA Damage Hypothesis of Aging. Wrote the letter that resulted
in the Manhattan Project. Conceived of the nuclear chain reaction, nuclear power, chemostat,
electron microscopes, enzyme feedback inhibition, and cloning of a human cel .
CONRAD H. WADDINGTON (November 8, 1905–September 26, 1975): British geneticist and
philosopher who laid the foundations of systems biology and epigenetics. His Waddington
Landscape was proposed to help understand how a cel can divide to become the hundreds of
di erent cel types in the body.
ROY L. WALFORD (June 29, 1924–April 27, 2004): American biologist who rejuvenated the
eld of caloric restriction. One of eight crew members inside Arizona’s Biosphere 2 from 1991 to



1993. In medical school, reportedly used statistical analysis to predict the results of a roulette
wheel in Reno, Nevada, to pay for medical school and a yacht, and sailed the Caribbean for over
a year.
H. G. WELLS (September 21, 1866–August 13, 1946): British science ction writer who
foresaw air raids in World War II, tanks, nuclear weapons, satel ite television, and the internet.
Best known for The War of the Worlds, The Shape of Things to Come, and The Time Machine.
His epitaph is from A War in the Air: “I told you so. You damned fools.”
GEORGE C. WILLIAMS (May 12, 1926–September 8, 2010): American evolutionary biologist at
the State University of New York, Stony Brook, known for developing a gene-centric view of
evolution and “Antagonistic Pleiotropy,” a leading theory about why we age; essential y that a
gene that helps young individuals survive can come back to bite them when they are older.
SHINYA YAMANAKA (September 4, 1962–): Japanese biologist who discovered reprogramming
genes that turn regular cel s into stem cel s, for which he shared the Nobel Prize in Physiology or
Medicine with John Gurdon in 2012.



Glossary
ALLELE: One of several possible versions of a gene. Each one contains a distinct variation in its
DNA sequence. For example, a “deleterious al ele” is a form of a gene that leads to disease.
AMINO ACID: The chemical building block of proteins. During translation, di erent amino
acids are strung together to form a chain that folds into a protein.
ANTAGONISTIC PLEIOTROPY: A theory proposed by George C. Wil iams as an evolutionary
explanation for aging: a gene that reduces lifespan in late life can be selected for if its early bene ts outweigh its late costs. An example of this is the survival circuit.
BASE: The four “letters” of the genetic code, A, C, T, and G, are chemical groups cal ed bases or
nucleobases. A= adenine, C = cytosine, T = thymine, and G = guanine. Instead of thymine,
RNA contains a base cal ed uracil (U).
BASE PAIR: “Teeth” on the twisted “zipper” of DNA. Chemicals known as bases make up a
DNA strand, each strand runs in the opposite direction, and bases attract their opposite partner



to make a base pair: C pairs with G, A pairs with T (except for in RNA, where it’s a U).
BIOTRACKING/BIOHACKING: The use of devices and lab tests to monitor the body to make
decisions about food, exercise, and other lifestyle choices to optimize the body. Not to be
confused with biohacking, which is do-it-yourself body enhancement.
CANCER: A disease caused by uncontrol ed growth of cel s. Cancerous cel s may form clumps or
masses known as tumors and can spread to other parts of the body through a process known as
metastasis.
CELL: The basic unit of life. The number of cel s in a living organism ranges from one (e.g., in
yeast) to quadril ions (e.g., in a blue whale). A cel is composed of four key macromolecules that
al ow it to function: protein, lipids, carbohydrates, and nucleic acids. Among other things, cel s
can build and break down molecules, move, grow, divide, and die.
CELLULAR REPROGRAMMING: The changing of cel s from one type of tissue to a prior stage of
development.
CELLULAR SENESCENCE: The process that occurs when normal cel s stop dividing and start to
release in ammatory molecules, sometimes caused by telomere shortening, damage to DNA, or
epigenomic noise. Despite their seeming “zombie” state, senescent cel s remain alive, damaging
nearby cel s with their in amatory secretions.




CHROMATIN: Strands of DNA wound around protein sca olds known as histones.
Euchromatin is open chromatin that al ows genes to be switched on. Heterochromatin is closed
chromatin that prevents the cel from reading a gene, also known as gene silencing.
CHROMOSOME: The compact structure into which a cel ’s DNA is organized, held together by
proteins. The genomes of di erent organisms are arranged into varying numbers of
chromosomes. Human cel s have 23 pairs.
COMPLEMENTARY: Describes any two DNA or RNA sequences that can form a series of base
pairs with each other. Each base forms a bond with a complementary partner: T (in DNA) and
U (in RNA) bond with A, and C bonds with G.
CRISPR: Pronounced “crisper.” An immune system found in bacteria and archaea, co-opted as a
genome-engineering tool to cut DNA at precise places in a genome. CRISPR, which stands for
“clustered regularly interspaced short palindromic repeats,” is a section of the host genome
containing alternating repetitive sequences and snippets of foreign DNA. CRISPR proteins
such as Cas9, a DNA-cutting enzyme, use these as molecular “mug shots” as they seek out and
destroy viral DNA.
DAF-16/FOXO: An al y of sirtuins, DAF-16/FOXO is a gene control protein cal ed a
transcription factor that activates cel defense genes, upregulation of which extends lifespan in
worms, ies, mice, and perhaps humans; required for Daf-2 to extend lifespan in worms.
DEACETYLATION: The enzymatic removal of acetyl tags from proteins. Removal of acetyls from
histones by histone deacetylases (HDACs) causes them to be more tightly packed, switching o a
gene. Sirtuins are NAD-dependent deacetylases. Deacylation is a catchal term that includes
deacetylation and the removal of other, more exotic tags such as butyryls and succinyls.

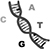
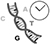
DEMETHYLATION: Demethylation is the removal of methyls and is carried out by enzymes cal ed
histone demethylases (KDMs) and DNA demethylases (TETs). Attachment of methyls is
achieved by a histone or DNA methyltransferases (DMTs).
DISPOSABLE SOMA: A hypothesis proposed by Tom Kirkwood to explain aging. Species evolve
to grow and multiply quickly or build a long-lasting body, but not both; limited resources in the
wild don’t al ow for both.
DNA: Abbreviation of deoxyribonucleic acid, the molecule that encodes the information needed
for a cel to function or a virus to replicate. Forms a double-helix shape that resembles a twisted
ladder, similar to a zipper. Bases, abbreviated as A, C, T, and G, are found on each side of the
ladder, or strand, which run in opposite directions. The bases have an attraction for each other,
making A stick to T and C stick to G. The sequence of these letters is cal ed the genetic code.
DNA DOUBLE-STRAND BREAK (DSB): What happens when both strands of DNA are broken
and two free ends are created. May be done intentional y with an enzyme such as Cas9 or I- Ppo I.
Cel s repair their DNA to prevent cel death, sometimes changing the DNA sequence at the site
of the break. Initiating or control ing this process with the intent to alter a DNA sequence is
known as genome engineering.
DNA METHYLATION CLOCK: Changes in the number and sites of DNA methylation tags on
DNA can be used to predict lifespan, marking time from birth. During epigenomic
reprogramming or cloning of an organism, methyl marks are removed, reversing the age of the
cel .
ENZYME: A protein made up of strings of amino acids that folds into a bal that can carry out
chemical reactions that would normal y take much longer or otherwise never happen. Sirtuins,
for example, are enzymes that use NAD to remove acetyl chemical groups from histones.



EPIGENETIC: Refers to changes to a cel ’s gene expression that do not involve altering its DNA
code. Instead the DNA and the histones that the DNA is wrapped around are “tagged” with
removable chemical signals (see Demethylation and deacetylation). Epigenetic marks tel other
proteins where and when to read the DNA, comparable to sticking a note that says “Skip” onto a
page of a book. A reader wil ignore the page, but the book itself has not been changed.
EPIGENETIC DRIFT AND EPIGENETIC NOISE: Alterations to the epigenome that take place with
age due to changes in methylation, often related to an individual’s exposure to environmental
factors. Epigenomic drift and noise may be a key driver of aging in al species. Damage to DNA,
especial y DNA breaks, is a driver of this process.
EXDIFFERENTIATION: The loss of cel identity due to epigenetic noise. Exdi erentiation may be
a major cause of aging (see Epigenetic Noise).
EXTRACHROMOSOMAL RIBOSOMAL DNA CIRCLE (ERC): The generation of
extrachromosomal ribosomal DNA circles leads to the breaking apart of the nucleolus in old
cel s, and in yeast they distract the sirtuins and cause aging.
GENE: A segment of DNA that encodes the information used to make a protein. Each gene is a
set of instructions for making a particular molecular machine that helps a cel , organism, or virus
function.
GENE EXPRESSION: A product based on a gene; can refer to either RNA or protein. When a
gene is turned on, cel ular machines express this by transcribing the DNA into RNA and/or




translating the RNA into a chain of amino acids. For example, a highly expressed gene wil have
many RNA copies produced, and its protein product is likely to be abundant in the cel .
GENE THERAPY: The delivery of corrective DNA to human cel s as a medical treatment. Certain
diseases can be treated or even cured by adding a healthy DNA sequence into the genomes of
particular cel s. Scientists and doctors typical y use a harmless virus to shuttle genes into targeted
cel s or tissues, where the DNA is incorporated somewhere within the cel s’ existing DNA.
CRISPR genome editing is sometimes referred to as a gene therapy technique.
GENETICALLY MODIFIED ORGANISM (GMO): An organism that has had its DNA altered
intentional y using scienti c tools. Any organism can be engineered in this manner, including
microbes, plants, and animals.
GENOME: The entire DNA sequence of an organism or virus. The genome is essential y a huge
set of instructions for making individual parts of a cel and directing how everything should run.
GENOMICS: The study of the genome, al the DNA of a given organism. Involves a genome’s
DNA sequence, the organization and control of genes, the molecules that interact with the
DNA, and the way in which these di erent components a ect the growth and function of cel s.
GERM CELLS: The cel s involved in sexual reproduction: eggs, sperm, and precursor cel s that
develop into eggs or sperm. The DNA in germ cel s, including any mutations or intentional
genetic edits, may be passed down to the next generation. Genome editing in an early embryo is
considered to be germline editing since any DNA changes wil likely end up in al cel s of the
organism that is eventual y born.




HISTONES: The proteins that form the core of DNA packaging in the chromosome and the
reason three feet of DNA can t inside a cel . DNA wraps around each histone almost two times,
like beads on a string. The packaging of histones is control ed by enzymes such as the sirtuins
that add and subtract chemical groups. Tight packaging forms “silent” heterochromatin, while
loose packaging forms open euchromatin, where genes are turned on.
HORMESIS: The idea that whatever doesn’t kil you makes you stronger. A level of biological
damage or adversity that stimulates repair processes that provide cel survival and health bene ts.
Original y discovered when plants were sprayed with diluted herbicide and afterward grew faster.
INFORMATION THEORY OF AGING: The idea that aging is due to the loss of information over
time, primarily epigenetic information, much of which can be recovered.
METFORMIN: A molecule derived from the French hel ebore used to treat type 2 (age-associated)
diabetes that may be a longevity medicine.
MITOCHONDRIA: Often cal ed the cel ’s powerhouse, mitochondria break down nutrients to
create energy in a process cal ed cel ular respiration. They contain their own circular genome.
MUTATION: A change from one genetic letter (nucleotide) to another. Variation in the DNA
sequence gives rise to the incredible diversity of species among di erent organisms of the same
genus. Though some mutations have no consequences at al , others can directly cause disease.
Mutations may be caused by DNA-damaging agents such as ultraviolet light, cosmic radiation,
or DNA copying by enzymes. They can also be created deliberately via genome-engineering
methods.




NAD: Nicotinamide adenine nucleotide, a chemical used for more than ve hundred chemical
reactions and for sirtuins to remove acetyl groups of other proteins such as histones to turn genes
o or give them cel protective functions. A healthy diet and exercise raise NAD levels. The “+”
sign you sometimes see, as in NAD+, indicates that it is not carrying a hydrogen atom.
NUCLEASE: An enzyme that breaks apart the backbone of RNA or DNA. Breaking one strand
generates a nick, and breaking both strands generates a double-strand break. An endonuclease
cuts in the middle of RNA or DNA, while an exonuclease cuts from the end of the strand.
Genome engineering tools such as Cas9 and I- Ppo I are endonucleases.
NUCLEIC ACIDS OR NUCLEOTIDES: The basic chemical units that are strung together to make
DNA or RNA. They consist of a base, a sugar, and a phosphate group. The phosphates link with
sugars to form the DNA/RNA backbone, while the bases bind to their complementary partners
to form base pairs.
NUCLEOLUS: Located inside the nucleus of eukaryotic cel s, the nucleolus is a region where the
ribosomal DNA (rDNA) genes are situated and where the cel ular machines for stitching
together amino acids to form proteins are assembled.
PATHOGEN: A microbe that causes il ness. Most microorganisms are not pathogenic to humans,
but some strains or species are.
PROTEIN: A string of amino acids folded into a three-dimensional structure. Each protein is specialized to perform a speci c role to help cel s grow, divide, and function. Proteins are one of
the four macromolecules that make up al living things (proteins, lipids, carbohydrates, and
nucleic acids).


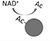
RAPAMYCIN: Also known as sirolimus, rapamycin is a compound with immunosuppressant
functions in humans. It inhibits activation of T cel s and B cel s by reducing their sensitivity to
the signaling molecule interleukin-2. Extends lifespan by inhibiting mTOR.
REDIFFERENTIATION: The reversal of epigenetic changes that occur during aging.
RIBOSOMAL DNA (rDNA): A key component of the manufacture of new proteins within cel s;
the source of the genetic code for ribosomal RNA, which is the building block of the ribosome.
These molecules knit together amino acids that become new proteins.
RNA: Abbreviation of ribonucleic acid. Transcribed from a DNA template and typical y used to
direct the synthesis of proteins. CRISPR-associated proteins use RNAs as guides to nd
matching target sequences in DNA.
SENOLYTICS: Pharmaceuticals currently under development that are hoped to kil senescent
cel s in order to slow down or even reverse aging-related issues.
SIRTUINS: Enzymes that control longevity; they are found in organisms from yeast to humans
and need NAD+ to function. They remove acetyl and acyl groups from proteins to instruct
them to protect cel s from adversity, disease, and death. During fasting or exercise, sirtuin and
NAD+ levels increase, which may explain why those activities are healthy. Named after the yeast
SIR2 longevity gene, SIRT1–7 (Sir2 homologs 1 to 7) genes in mammals play key roles in protecting against disease and deterioration.
SOMATIC CELLS: Al the cel s in a multicel ular organism except for germ cel s (eggs or sperm).
Mutations or changes to the DNA in the soma wil not be inherited by subsequent generations


unless cloning takes place.
STEM CELLS: Cel s with the potential to turn into a specialized type of cel or divide to make
more stem cel s. Most cel s in your body are di erentiated; that is, their fate has already been
decided and they cannot morph into a di erent kind of cel . For example, a cel in your brain
cannot suddenly transform into a skin cel . Adult stem cel s replenish the body as it becomes
damaged over time.
STRAND: A string of connected nucleotides; can be DNA or RNA. Two strands of DNA can
zip together when complementary; bases match up to form base pairs. DNA typical y exists in
this double-stranded form, which takes the shape of a twisted ladder or double helix. RNA is
typical y composed of just a single strand, though it can fold up into complex shapes.
SURVIVAL CIRCUIT: An ancient control system in cel s that may have evolved to shift energy
away from growth and reproduction toward cel ular repair during times of adversity. After
response to adversity, the system may not ful y reset, which, over time, leads to a disruption of
the epigenome and loss of cel identity leading to aging (see Antagonistic Pleiotropy).
TELOMERES/TELOMERE LOSS: A telomere is a cap that protects the end of the chromosome
from attrition, analogous to the aglet at the end of a shoelace or a burned end of a rope to stop it
fraying. As we age, telomeres erode to the point where the cel reaches the Hay ick limit. This is
when the cel regards the telomere as a DNA break, stops dividing, and becomes senescent.
TRANSCRIPTION: The process by which genetic information is copied into a strand of RNA;
performed by an enzyme cal ed RNA polymerase.



TRANSLATION: The process by which proteins are made based on instructions encoded in an
RNA molecule. Performed by a molecular machine cal ed the ribosome, which links together a
series of amino acid building blocks. The resulting polypeptide chain folds up into a particular
3D object, known as a protein.
VIRUS: An infectious entity that can persist only by hijacking a host organism to replicate itself
in. Has its own genome but is technical y not considered a living organism. Viruses infect al
organisms, from humans to plants to microbes. Multicel ular organisms have sophisticated
immune systems that combat viruses, while CRISPR systems evolved to stop viral infection in
bacteria and archaea.
WADDINGTON’S LANDSCAPE: A biological metaphor for how cel s are endowed with an
identity during embryonic development in the form of a 3D relief map. Marbles representing
stem cel s rol down into bifurcating val eys, each of which marks a di erent developmental
pathway for the cel s.
XENOHORMESIS HYPOTHESIS: The idea that our bodies evolved to sense the stress cues of
other species, such as plants, in order to protect themselves during times of impending adversity.
Explains why so many medicines come from plants.

ABOUT THE AUTHORS
DAVID A. SINCLAIR, PHD, AO, is one of the world’s most famous scientists and
entrepreneurs, best known for understanding why we age and how to reverse it.
He is a tenured professor of genetics, Blavatnik Institute, Harvard Medical
School; co-director of the Paul F. Glenn Center for the Biology of Aging
Research at Harvard; co-joint professor and head of the Aging Labs at the
University of New South Wales in Sydney, Australia; and an honorary professor
at the University of Sydney. His work is regularly featured in print, podcasts,
TV, and books, including 60 Minutes, a Barbara Walters special, NOVA, and
Morgan Freeman’s Through the Wormhole. He is best known for his work on
genes and smal molecules that delay aging, including the sirtuin genes,
resveratrol, and NAD precursors. He has published more than 170 scienti c
papers, is a coinventor on more than 50 patents, and has cofounded 14
biotechnology companies in the areas of aging, vaccines, diabetes, fertility,



cancer, and biodefense. He serves as co–chief editor of the scienti c journal
Aging, works with national defense agencies and with NASA, and has received
35 honors, including being one of Australia’s leading scientists under 45, the
Australian Medical Research Medal, the NIH Director’s Pioneer Award, Time
magazine’s list of the “100 Most In uential People in the World” (2014) and the
“Top 50 People in Healthcare” (2018). In 2018, he became an O cer of the
Order of Australia for his work in medicine and national security.
MATTHEW D. LAPLANTE is an associate professor of journalistic writing at Utah
State University. A selection of his work as a journalist, radio host, author, and
cowriter can be found at www.mdlaplante.com.
Authors.SimonandSchuster.com/David-A-Sinclair
Authors.SimonandSchuster.com/Matthew-D-Laplante



Connect with Dr. David A. Sinclair on
We hope you enjoyedreading this Simon &
Schuster ebook.
Get a FREE ebook when you join our mailing list. Plus, get updates on new
releases, deals, recommended reads, and more from Simon & Schuster.
Click below to sign up and see terms and conditions.
Already a subscriber? Provide your email again so we can register this ebook
and send you more of what you like to read. You wil continue to receive
exclusive offers in your inbox.
NotesINTRODUCTION: A GRANDMOTHER’S PRAYER
1. In a wide-ranging interview to promote his memoirs, Lanzmann said of his masterpiece lm about the Holocaust, “I wanted to get as close as possible to death. No personal accounts are
told in Shoah, no anecdotes. It’s only about death. The lm is not about the survivors.”
“ ‘Shoah’ Director Claude Lanzmann: ‘Death Has Always Been a Scandal,’ ” Spiegel,
September 10, 2010, http://www.spiegel.de/international/zeitgeist/shoah-director-claude-
lanzmann-death-has-always-been-a-scandal-a-716722.html.
2. The study looked at three concepts about death that children come to understand before they are seven years old: irreversibility, nonfunctionality, and universality. M. W. Speece and
S. B. Brent, “Children’s Understanding of Death: A Review of Three Components of a
Death Concept,” Child Development 55, no. 5 (October 1984): 1671–86,
https://www.ncbi.nlm.nih.gov/pubmed/6510050.
3. The author attended the birth of her daughter’s rst child along with her son-in-law. R. M.
Henig, “The Ecstasy and the Agony of Being a Grandmother,” New York Times, December
https://www.nytimes.com/2018/12/27/style/self-care/becoming-a-
4. The lm’s exhortations to make the most of every day took on a darker hue after the suicide of its star, Robin Wil iams. P. Weir, director, Dead Poets Society, United States: Touchstone
Pictures, 1999.
5. The author argues that rather than focusing on cancer and cardiovascular issues, medical research should be focusing on “reducing ageing and age-related morbidity, thereby
increasing both our health and our wealth.” G. C. Brown, “Living Too Long,” EMBO
Reports
16,
no.
2
(February
2015):
137–41,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4328740/.
6. In a survey coconducted by the Economist, the majority of respondents from four countries expressed the wish to die at home, although only a smal number thought that they would
do so. With the exception of Brazilians, most felt that dying without pain was more
important than extending life. “A Better Way to Care for the Dying,” Economist, April 29,
2017, https://www.economist.com/international/2017/04/29/a-better-way-to-care-for-the-7. See my con ict disclosures at the end of this book and at
https://genetics.med.harvard.edu/sinclair-test/people/sinclair-other.php.
8. My editor made me write self-centered things about myself to give me credibility. I hope she doesn’t see this endnote and make me delete it.
9. In 2018, my family and I made a pilgrimage to London to see the original account of Captain James Cook’s “voyage round the world” and the original Australian botanical specimens
col ected by Sir Joseph Banks. There were stop-o s to see Watson and Crick’s original model
of DNA, fossils of early life, a Moai statue from Rapa Nui, a cross-sectional cut through a
1,500-year-old sequoia tree trunk, a statue of Charles Darwin, the Broad Street pump,
Winston Churchil ’s War Rooms, and the Royal Society, of course. Tracing the path of Cook
along the lower east coast of Australia, or “New Hol and,” as it was cal ed then, it is obvious
that Banks already had a colony in mind, one that would never forget him. Not only was the
original site named Botany Bay, the coast was named “Cape Banks.” After exploring Botany
Bay, the explorers’ tal ship, the HMS Endeavor, sailed north, past the heads of a harbor they
cal ed Port Jackson, which, thanks to its much deeper waters and the presence of a stream to
supply fresh water, ended up being a far superior site for Governor Phil ip to start a penal
colony eight years later.
10. “Phil ip’s Exploration of Middle Harbour Creek,” Fel owship of the First Fleeters, Arthur
Phil ip Chapter, http://arthurphil ipchapter.weebly.com/exploration-of-middle-harbour-
11. The Spanish explorer and conquistador’s search for the mystical spring known as the Fountain of Youth is apocryphal, but it makes for a good story. J. Greenspan, “The Myth of
Ponce de León and the Fountain of Youth,” “History Stories,” April 2, 2013, A&E
Television Networks, https://www.history.com/news/the-myth-of-ponce-de-leon-and-the-
12. According to the Creation Wiki: the Encyclopedia of Creation Science (a website of the Northwest Creation Network, http://creationwiki.org/Human_longevity), in Genesis, most of us once got to 900 years, then we didn’t. Then most of us got to 400, then we didn’t.
Then most of us got to 120, then we didn’t. In more recent times, as Oeppen and Vaupel
have written, “Mortality experts have repeatedly asserted that life expectancy is close to an
ultimate ceiling; these experts have repeatedly been proven wrong. The apparent leveling o
of life expectancy in various countries is an artifact of laggards catching up and leaders fal ing
behind.” J. Oeppen and J. W. Vaupel, “Broken Limits to Life Expectancy,” Science 296, no.
5570 (May 10, 2002): 1029–31.
13. There is some debate as to what constitutes veri able age. There are humans who have claimed, and provided considerable evidence, of being of great age, but who don’t have
formal Western-style records of their year of birth. In any case, these people are one in a
bil ion, if that. In November 2018, the Russian gerontologist Valery Novoselov and the mathematician Nikolay Zak claimed that after much research, they believe that JeanneCalment’s daughter, Yvonne, usurped Jeanne’s identity in 1934, claiming that the daughter
had died instead of the mother to avoid paying estate taxes. The debate continues. “French
Scientists Dismiss Russian Claims over Age of World’s Oldest Person,” Reuters, January 3,
https://www.reuters.com/article/us-france-oldest-woman-controversy/french-
scientists-dismiss-russian-claims-over-age-of-worlds-oldest-person-idUSKCN1OX145.
14. Italian researchers found after studying 4,000 elderly people that if you make it to age 105, the risk of death e ectively plateaus from one birthday to the next, the odds of dying in the
next year becoming approximately fty- fty. E. Barbi, F. Lagona, M. Marsili, et al., “The
Plateau of Human Mortality: Demography of Longevity Pioneers,” Science 360, no. 396
(June 29, 2018): 1459–61, http://science.sciencemag.org/content/360/6396/1459.
15. “If people live on average to 80 or 90, like they do now, then the very long lived make it to 110 or 120,” says Siegfried Hekimi, professor of genetics at McGil University in Canada.
“So if the average lifespan keeps expanding, that would mean the long-lived would live even
longer, beyond 115 years”; A. Park, “There’s No Known Limit to How Long Humans Can
Live, Scientists Say,” Time, June 28, 2017, http://time.com/4835763/how-long-can-
16. “Any su ciently advanced technology is indistinguishable from magic.” “Arthur C. Clarke,”
Wikiquote, https://en.wikiquote.org/wiki/Arthur_C._Clarke.
ONE. VIVA PRIMORDIUM
1. D. Damer and D. Deamer, “Coupled Phases and Combinatorial Selection in Fluctuating Hydrothermal Pools: A Scenario to Guide Experimental Approaches to the Origin of
Cel ular Life,” Life 5, no. 1 (2015): 872–87, https://www.mdpi.com/2075-1729/5/1/872.
2. According to precise radiological and geological readings and recent discoveries about the early chemistry of life, this is an accurate picture of how the inanimate was animated and life
took hold. M. J. Van Kranendonk, D. W. Deamer, and T. Djokic, “Life on Earth Came from
a Hot Volcanic Pool, Not the Sea, New Evidence Suggests,” Scientific American, August
2017, https://www.scienti camerican.com/article/life-on-earth-came-from-a-hot-volcanic-
pool-not-the-sea-new-evidence-suggests/.
3. J. B. Iorgulescu, M. Harary, C. K. Zogg, et al., “Improved Risk-Adjusted Survival for Melanoma Brain Metastases in the Era of Checkpoint Blockade Immunotherapies: Results
from a National Cohort,” Cancer Immunology Research, 6, no. 9 (September 2018): 1039–
45, http://cancerimmunolres.aacrjournals.org/content/6/9/1039.long; R. L. Siegel, K. D.
Mil er, and A. Jemal, “Cancer Statistics, 2019,” CA: A Cancer Journal for Clinicians 69, no. 1
(January–February
2019):
7–34,
https://onlinelibrary.wiley.com/doi/ful /10.3322/caac.21551.
4. As far back as Aristotle, scientists and philosophers have struggled to resolve the enigma of aging, the authors wrote. D. Fabian and T. Flatt, “The Evolution of Aging,” NatureEducation
Knowledge
3,
no.
10
(2011):
9,
https://www.nature.com/scitable/knowledge/library/the-evolution-of-aging-23651151.
5. A bat from Siberia set a world record when it reached 41 years of age. R. Locke, “The Oldest Bat: Longest-Lived Mammals O er Clues to Better Aging in Humans,” BATS Magazine 24,
no. 2 (Summer 2006): 13–14, http://www.batcon.org/resources/media-education/bats-
6. Smal colonies of lizards on a series of Caribbean islands were likely to explore islands where there weren’t predators, while less adventurous animals survived better when predators were
present. O. Lapiedra, T. W. Schoener, M. Leal, et al., “Predator-Driven Natural Selection on
Risk-Taking Behavior in Anole Lizards,” Science 360, no. 3692 (June 1, 2018): 1017–20,
http://science.sciencemag.org/content/360/6392/1017.
7. Richard Dawkins eloquently made this point in River Out of Eden, arguing that primitive societies don’t have a place in science, using as an example their belief the moon is an old
calabash tossed into the sky. R. Dawkins, River Out of Eden (New York: Basic Books, 1995).
8. See “The Scale of Things” at the end of this book.
9. Szilard spent his last years as a fel ow of the Salk Institute for Biological Studies in La Jol a, California, as a resident fel ow. He lived in a bungalow on the property of the Hotel del
Charro and died on May 30, 1964.
10. R. Anderson, “Ionizing Radiation and Aging: Rejuvenating an Old Idea,” Aging 1, no. 11
(November
17,
2009):
887–902,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2815743/.
11. L. E. Orgel, “The Maintenance of the Accuracy of Protein Synthesis and Its Relevance to Ageing,” Proceedings of the National Academy of Sciences of the United States of America 49,
no. 4 (April 1963): 517–21, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC299893/.
12. Harman concluded that the diseases related to aging, as wel as aging itself, stem fundamental y from “the deleterious side attacks of free-radicals on cel constituents and on
the connective tissues.” The source of the free radicals, he continued, was “molecular oxygen
catalyzed in the cel by the oxidative enzymes” and metal traces. D. Harman, “Aging: A
Theory Based on Free Radical and Radiation Chemistry,” Journal of Gerontology 11, no. 3
https://academic.oup.com/geronj/article-
abstract/11/3/298/616585?redirectedFrom=ful text.
13. Nutraceuticals World predicts that a rising appetite for synthetic antioxidants at the same time as a fal in costs, combined with increasing demand for them by food and beverage
companies, wil power market growth for the next few years. “Global Antioxidants Market
Expected to Reach $4.5 Bil ion by 2022,” Nutraceuticals World, January 26, 2017,
https://www.nutraceuticalsworld.com/contents/view_breaking-news/2017-01-26/global-
antioxidants-market-expected-to-reach-45-bil ion-by-2022
14. The sharp growth in demand for drinks with a health bene t, a beverage industry website nds, goes hand in hand with consumers wanting ingredients they value. A. Del Buono,“Consumers’ Understanding of Antioxidants Grows,” Beverage Industry, January 16, 2018,
https://www.bevindustry.com/articles/90832-consumers-understanding-of-antioxidants-
15. I. Martincorena, J. C. Fowler, A. Wabik, et al., “Somatic Mutant Clones Colonize the Human Esophagus with Age,” Science 362, no. 6417 (November 23, 2018): 911–17,
https://www.ncbi.nlm.nih.gov/pubmed/30337457.
16. The authors concluded that their data “cal s into serious question the hypothesis that alterations in oxidative damage/stress play a role in the longevity of mice.” V. I. Pérez, A.
Bokov, H. Van Remmen, et al., “Is the Oxidative Stress Theory of Aging Dead?,” Biochimica
et
Biophysica
Acta
1790,
no.
10
(October
2009):
1005–14,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2789432/.
17. A. P. Gomes, N. L. Price, A. J. Ling, et al., “Declining NAD(+) Induces a Pseudohypoxic State Disrupting Nuclear-Mitochondrial Communication During Aging,” Cell 155, no. 7
(December 19, 2013): 1624–38, https://www.ncbi.nlm.nih.gov/pubmed/24360282.
18. W. Lanouette and B. Silard, Genius in the Shadows: A Biography of Leo Szilard: The Man Behind the Bomb (New York: Skyhorse Publishing, 1992).
19. According to the NIH fact sheet, “clones created from a cel taken from an adult might have chromosomes that are already shorter than normal, which may condemn the clones’ cel s to
a shorter life span.” “Cloning,” National Human Genome Research Institute, March 21,
2017, https://www.genome.gov/25020028/cloning-fact-sheet/.
20. In the debates over Dol y the cloned sheep, the question that has proved to be chal enging to answer is how old an animal is at birth when cloned from an adult’s cel . The answer an
author on the site The Conversation found was that other clones born from the same cel as
Dol y lived normal lifespans. “The new Dol ies are now tel ing us that if we take a cel from
an animal of any age, and we introduce its nucleus into a nonfertilized mature egg, we can
have an individual born with its lifespan ful y restored.” J. Cibel , “More Lessons from Dol y
the Sheep: Is a Clone Real y Born at Age Zero?,” The Conversation, February 17, 2017,
https://theconversation.com/more-lessons-from-dol y-the-sheep-is-a-clone-real y-born-at-
21. Though some cloned animals match their species’ rates of normal aging, it’s a eld that stil needs further analysis to get beyond the largely anecdotal evidence so far col ected. J. P.
Burgstal er and G. Brem, “Aging of Cloned Animals: A Mini-Review,” Gerontology 63, no. 5
(August 2017): 417–25, https://www.karger.com/Article/Ful Text/452444.
22. University of Bath researchers found in cloned mice that the telomeres protecting the ends of chromosomes were, surprisingly, slightly longer in successive generations and demonstrated
no evidence of premature aging. T. Wakayama, Y. Shinkai, K. L. K. Tamashiro, et al.,
“Ageing: Cloning of Mice to Six Generations,” Nature 407 (September 21, 2000): 318–19.
“Despite the length of telomeres reported in di erent studies, most clones appear to be aging normal y. In fact, the rst cattle clones ever produced are alive, healthy, and are 10 years old asof January 2008”; “Myths About Cloning,” U.S. Food & Drug Administration, August 29,
2018, https://www.fda.gov/animalveterinary/safetyhealth/animalcloning/ucm055512.htm.
23. The authors discovered mitochondrial DNA in a Neanderthal bone in Croatia that revealed older dates of survival than previously thought. T. Devièse, I. Karavanié, D. Comeskey, et al.,
“Direct Dating of Neanderthal Remains from the Site of Vindija Cave and Implications for
the Middle to Upper Paleolithic Transition,” Proceedings of the National Academy of Sciences
of the United States of America 114, no. 40 (October 3, 2017): 10606–11,
https://www.ncbi.nlm.nih.gov/pubmed/28874524.
24. A. S. Adikesevan, “A Newborn Baby Has About 26,000,000,000 Cel s. An Adult Has About
1.9 × 103 Times as Many Cel s as a Baby. About How Many Cel s Does an Adult Have?,”
Socratic, January 26, 2017, https://socratic.org/questions/a-newborn-baby-has-about-26-
000-000-000-cel s-an-adult-has-about-1-9-10-3-times-.
25. C. B. Brachmann, J. M. Sherman, S. E. Devine, et al., “The SIR2 Gene Family, Conserved from Bacteria to Humans, Functions in Silencing, Cel Cycle Progression, and Chromosome
Stability,” Genes & Development 9, no. 23 (December 1, 1995): 2888–902,
http://genesdev.cshlp.org/content/9/23/2888.long; X. Bi, Q. Yu, J. J. Sandmeier, and S.
Elizondo, “Regulation of Transcriptional Silencing in Yeast by Growth Temperature,”
Journal of Molecular Biology 34, no. 4 (December 3, 2004): 893–905,
https://www.ncbi.nlm.nih.gov/pubmed/15544800.
26. It is one of the most interesting and important papers I’ve ever read. C. E. Shannon, “A Mathematical Theory of Communication,” Bell System Technical Journal 27, no. 3 (July
1948):
379–423,
and
27,
no.
4
(October
1948):
623–66,
http://math.harvard.edu/~ctm/home/text/others/shannon/entropy/entropy.pdf.
27. Research by the authors showed that mTORC1 signaling in cancer cel s increases survival by
“suppressing endogenous DNA damage, and may control cel fate through the regulation of
CHK1.” X. Zhou, W. Liu, X. Hu, et al., “Regulation of CHK1 by mTOR Contributes to
the Evasion of DNA Damage Barrier of Cancer Cel s,” Nature Scientific Reports, May 8,
2017, https://www.nature.com/articles/s41598-017-01729-w; D. M. Sabatini, “Twenty- ve Years of mTOR: Uncovering the Link from Nutrients to Growth,” Proceedings of the
National Academy of Sciences of the United States of America 114, no. 45 (November 7,
2017): 11818–25, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5692607/.
28. E. J. Calabrese, “Hormesis: A Fundamental Concept in Biology,” Microbial Cell 1, no. 5
(May 5, 2014): 145–49, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5354598/.
TWO. THE DEMENTED PIANIST
1. Up to 69 percent of the human genome may be repetitive or derived from endogenous viral DNA repeats, compared to previous estimates of around half. A. P. de Konig, W. Gu, T. A.Castoe, et al., “Repetitive Elements May Comprise over Two-thirds of the Human
Genome,”
PLOS
Genetics
7,
no.
12
(December
7,
2011),
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3228813/.
2. Just what do we mean by the word finished when it comes to the sequencing of the human genome? Turns out, more than we thought back in the early 2000s. Regions of the genome
previously thought of as nonfunctional are now emerging as playing potential roles in
cancer, autism, and aging. S. Begley, “Psst, the Human Genome Was Never Completely
Sequenced. Some Scientists Say It Should Be,” STAT, June 20, 2017,
https://www.statnews.com/2017/06/20/human-genome-not-ful y-sequenced/.
3. Dating back to the 1960s, every three or four years the center has published a catalog of its strains of Saccharomyces cerevisiae. R. K. Mortimer, “Yeast Genetic Stock Center,”
Grantome, 1998, http://grantome.com/grant/NIH/P40-RR004231-10S1.
4. Yeast researchers have interesting names. John Johnston and my adviser Dick Dickinson are just two of them.
5. In 2016, Dr. Yoshinori Ohsumi won the Nobel Prize in Physiology or Medicine for his work on autophagy in yeast. That’s when cel s stave o extinction during hard times by digesting
nonkey parts of themselves. B. Starr, “A Nobel Prize for Work in Yeast. Again!,” Stanford
University, October 3, 2016, https://www.yeastgenome.org/blog/a-nobel-prize-for-work-in-
6. Dawes’s delightful tour of his experiences in the world of academe and cel biology research is a refreshingly direct and personal account of a remarkable journey into yeast research over
four decades. I. Dawes, “Ian Dawes—the Third Pope—Lucky to Be a Researcher,” Fems
Yeast
Research
6,
no.
4
(June
2016),
https://academic.oup.com/femsyr/article/16/4/fow040/2680350.
7. I also learned, the hard way, that I should not drink copious quantities of yeasty beer.
8. For four years after that, I sent Professor Melton a bottle of red wine for New Year’s, just to say thanks for changing my life. He never acknowledged any of them or ever smiled at me,
either because he didn’t think that’s what an awardee should do or because he’s a very private
person. At least he knew I was grateful. The selection of red wine turned out to be ironic, as
that foodstu helped propel my career a second time nine years later.
9. C. E. Yu, J. Oshima, Y. H. Fu, et al., “Positional Cloning of the Werner’s Syndrome Gene,”
Science
27,
no.
5259
(April
12,
1996):
258–62,
https://www.ncbi.nlm.nih.gov/pubmed/8602509.
10. SIR2 stands for “silent information regulator 2.” When SIR2 is written in capitals and italics, it refers to the gene; when it’s written Sir2, it refers to the protein.
11. In a paper published in late 1997, I showed how ERCs—rDNA circles—cause aging and
shorten the life of yeast cel s. D. A. Sinclair and L. Guarente, “Extrachromosomal rDNA
Circles—A Cause of Aging in Yeast,” Cell 91, no. 7 (December 26, 1997): 1033–42,https://www.ncbi.nlm.nih.gov/pubmed/9428525.
12. One way to think of the epigenome is as a cel ’s software. In the same way digital les are stored in a phone’s memory and the software uses the ones and zeros to turn a phone into a
clock, calendar, or music player, a cel ’s information is stored as As, Ts, Gs, and Ts, and the
epigenome uses those letters to direct a yeast cel to become male or a female and turn a
mammalian cel into a nerve, a skin cel , or an egg.
13. I am not the rst person to use this analogy. One of the earliest uses of the piano metaphor I can nd came from a study guide intended to accompany a Nova ScienceNOW program on
epigenetics
in
2007.
“Nova
ScienceNOW:
Epigenetics,”
PBS,
http://www.pbs.org/wgbh/nova/education/viewing/3411_02_nsn.html.
14. C. A. Makarewich and E. N. Olson, “Mining for Micropeptides,” Trends in Cell Biology 27, no. 9 (September 27, 2017): 685–96, https://www.ncbi.nlm.nih.gov/pubmed/28528987.
15. D. C. Dolinoy, “The Agouti Mouse Model: An Epigenetic Biosensor for Nutritional and
Environmental Alterations on the Fetal Epigenome,” Nutrition Reviews 66, suppl. 1 (August
2008): S7–11, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2822875/.
16. The more extroverted you are, the longer your lifespan, while, perhaps unsurprisingly, pessimists and psychotics see signi cant increases in the risk of death at an earlier age. That’s
according to a study of 3,752 twins 50 years or older that looked at the relationship between
personality and lifespan through the prism of genetic in uences. M. A. Mosing, S. E.
Medland, A. McRae, et al., “Genetic In uences on Life Span and Its Relationship to
Personality: A 16-Year Fol ow-up Study of a Sample of Aging Twins,” Psychosomatic
Medicine
74,
no.
1
(January
2012):
16–22,
https://www.ncbi.nlm.nih.gov/pubmed/22155943. The authors considered de nitions of extreme longevity, using multiple European twin registries. A. Skytthe, N. L. Pedersen, J.
Kaprio, et al., “Longevity Studies in GenomEUtwin,” Twin Research 6, no. 5 (October 2003): 448–54, https://www.ncbi.nlm.nih.gov/pubmed/14624729.
17. It was a eureka moment—discovering why yeast cel s age. Supercoiled circles of ribosomal DNA pinch o the yeast chromosome and accumulate as the yeast divide, distracting the
Sir2 enzyme from its main role of control ing genes for sex and reproduction. David A.
Sinclair and Leonard Guarente, “Extrachromosomal rDNA Circles—A Cause of Aging in
Yeast,” Cell 91 (December 26, 1997): 1033–42.
18. D. A. Sinclair, K. Mil s, and L. Guarente, “Accelerated Aging and Nucleolar Fragmentation in Yeast SGS1 Mutants,” Science 277, no. 5330 (August 29, 1997): 1313–16,
https://www.ncbi.nlm.nih.gov/pubmed/9271578.
19. Sinclair and Guarente, “Extrachromosomal rDNA Circles—A Cause of Aging in Yeast.”
20. K. D. Mil s, D. A. Sinclair, and L. Guarente, “MEC1-Dependent Redistribution of the Sir3
Silencing Protein from Telomeres to DNA Double-Strand Breaks,” Cell 97, no. 5 (May 28,
1999): 609–20, https://www.ncbi.nlm.nih.gov/pubmed/10367890.
21. Sinclair, Mil s, and Guarente, “Accelerated Aging and Nucleolar Fragmentation in Yeast SGS1 Mutants.”22. P. Oberdoer er, S. Michan, M. McVay, et al., “SIRT1 Redistribution on Chromatin Promotes Genomic Stability but Alters Gene Expression During Aging,” Cell 135, no. 5
(November 28, 2008): 907–18, https://www.cel .com/cel /ful text/S0092-8674(08)01317-2;
Z. Mao, C. Hine, X. Tian, et al., “SIRT6 Promotes DNA Repair Under Stress by Activating
PARP1,”
Science
332,
no.
6036
(June
2011):
1443–46,
https://www.ncbi.nlm.nih.gov/pubmed/21680843.
23. A. Ianni, S. Hoelper, M. Krueger, et al., “Sirt7 Stabilizes rDNA Heterochromatin Through Recruitment of DNMT1 and Sirt1,” Biochemical and Biophysical Research Communications
492,
no.
3
(October
21,
2017):
434–40,
https://www.ncbi.nlm.nih.gov/m/pubmed/28842251/.
24. The authors show how SIRT7, in protecting against the instability of rDNA, also guards against the death of human cel s. S. Paredes, M. Angulo-Ibanez, L. Tassel i, et al., “The
Epigenetic Regulator SIRT7 Guards Against Mammalian Cel ular Senescence Induced by
Ribosomal DNA Instability,” Journal of Biological Chemistry 293 (July 13, 2018): 11242–
50, http://www.jbc.org/content/293/28/11242.
25. Oberdoer er et al., “SIRT1 Redistribution on Chromatin Promotes Genomic Stability but Alters Gene Expression During Aging.”
26. M. W. McBurney, X. Yang, K. Jardine, et al., “The Mammalian SIR2alpha Protein Has a
Role in Embryogenesis and Gametogenesis,” Molecular and Cellular Biology 23, no. 1
(January 23, 2003): 38–54, https://mcb.asm.org/content/23/1/38.long.
27. R.-H. Wang, K. Sengupta, L. Cuiling, et al., “Impaired DNA Damage Response, Genome
Instability, and Tumorigenesis in SIRT1 Mutant Mice,” Cancer Cell 14, no. 4 (October 7,
2008): 312–23, https://www.cel .com/cancer-cel /ful text/S1535-6108(08)00294-8.
28. R. Mostoslavsky, K. F. Chua, D. B. Lombard, et al., “Genomic Instability and Aging-like Phenotype in the Absence of Mammalian SIRT6,” Cell 124 (January 27, 2006): 315–29,
https://doi.org/10.1016/j.cel .2005.11.044.
29. The treatments work better in male mice, for reasons that are not yet known, but my former postdoc Haim Cohen at Bar-Ilan University in Israel wins the award for the best-ever name
given to a transgenic mouse strain: MOSES. A. Satoh, C. S. Brace, N. Rensing, et al., “Sirt1
Extends Life Span and Delays Aging in Mice Through the Regulation of Nk2 Homeobox 1
in the DMH and LH,” Cell Metabolism 18, no. 3 (September 3, 2013): 416–30,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3794712.
30. When we write SIR2 in capitals and italics, it refers to the gene; when we write Sir2, it refers to the protein the gene encodes.
31. It’s possible that by not al owing mating-type genes to turn on, yeast with additional copies of SIR2 have less e cient DNA repair by homologous recombination, which is what the
expression of mating-type genes also does when switched on besides preventing mating. Thisneeds to be tested. But at least under safe lab conditions, the cel s grow perfectly ne.
32. M. G. L. Bail ie, A Slice Through Time: Dendrochronology and Precision Dating (London: Routledge, 1995).
33. Along with bristlecones, Matthew LaPlante, my coauthor on Lifespan, looks at a wide variety of biology’s outliers that de ne the very edges of our understanding of plants and animals,
from ghost sharks and elephants to beetles and microbacteria. M. D. LaPlante, Superlative:
The Biology of Extremes (Dal as: BenBel a Books, 2019).
34. When researchers compared trees of a variety of ages to look for a steady incremental decline in annual shoot growth, they found “no statistical y signi cant age-related di erences.” R.
M. Lanner, and K. F. Connor, “Does Bristlecone Pine Senesce?,” Experimental Gerontology
36,
nos.
4–6
(April
2001):
675–85,
https://www.sciencedirect.com/science/article/pi /S0531556500002345?via%3Dihub.
35. Investigating mutations in the gene Daf-2, researchers made a remarkable nd: the largest reported lifespan extension of any living thing, namely twice as long. This relied on the
involvement of two genes, Daf-2 and Daf-16, opening the door to new horizons of ways to
understand how to prolong life. C. Kenyon, J. Chang, E. Gensch, et al., “A C. elegans
Mutant That Lives Twice as Long as Wild Type,” Nature 366 (December 2, 1993): 461–64,
https://www.nature.com/articles/366461a0; F. Wang, C.-H. Chan, K. Chen, et al.,
“Deacetylation of FOXO3 by SIRT1 or SIRT2 Leads to Skp2-Mediated FOXO3
Ubiquitination and Degradation,” Oncogene 31, no. 12 (March 22, 2012): 1546–57,
https://www.nature.com/articles/onc2011347.
36. Why do genes often have a variety of names? The language of genetics is just like any other language; its words contain the echoes of history. Knowing the entire genome of a yeast cel ,
a nematode worm, or a human was the stu of dreams less than a quarter century ago. Now,
of course, I can sequence my own genome in a day on a USB drive–sized sequencer. When I
was a student, genes would be given a name based on the characteristics of mutants we
would generate with mutagenic chemicals. Typical y, al we knew about a gene when we
named it was its rough location on a particular chromosome. Only later were its distant
cousins identi ed.
37. A. Brunet, L. B. Sweeney, J. F. Sturgil , et al., “Stress-Dependent Regulation of FOXO
Transcription Factors by the SIRT1 Deacetylase,” Science 303, no. 5666 (March 24, 2004):
2011–15, https://www.ncbi.nlm.nih.gov/pubmed/14976264.
38. O. Medvedik, D. W. Lamming, K. D. Kim, and D. A. Sinclair, “MSN2 and MSN4 Link Calorie Restriction and TOR to Sirtuin-Mediated Lifespan Extension in Saccharomyces
cerevisiae,” PLOS Biology, October 2, 2007, http://journals.plos.org/plosbiology/article?
id=10.1371/journal.pbio.0050261.
39. The authors found convincing evidence linking FOXO3 and longevity in humans. L. Sun, C.
Hu, C. Zheng, et al., “FOXO3 Variants Are Bene cial for Longevity in Southern Chinese
Living in the Red River Basin: A Case-Control Study and Meta-analysis,” Nature Scientific Reports, April 27, 2015, https://www.nature.com/articles/srep09852.40. H. Bae, A. Gurinovich, A. Malovini, et al., “E ects of FOXO3 Polymorphisms on Survival to Extreme Longevity in Four Centenarian Studies,” Journals of Gerontology, Series A: Biological
Sciences and Medical Sciences 73, no. 11 (October 8, 2018): 1437–47,
https://academic.oup.com/biomedgerontology/article/73/11/1439/3872296.
41. If you’re a dedicated exerciser in middle age or an athlete in her fties, chances are your heart is going to resemble that of someone much younger, several studies have revealed. Not so for
the o ce worker who doesn’t exercise or someone who hits the gym or runs in the street on a
sporadic basis. What isn’t clear, though, is whether commencing an aggressive exercise
program in your middle years can turn around the e ects of a sedentary lifestyle on the
heart’s functioning and structure. G. Reynolds, “Exercise Makes the Aging Heart More
Youthful,”
New
York
Times,
July
25,
2018,
https://www.nytimes.com/2018/07/25/wel /exercise-makes-the-aging-heart-more-
42. “These ndings have implications for improving blood ow to organs and tissues, increasing human performance, and reestablishing a virtuous cycle of mobility in the elderly.”A. Das, G.
X. Huang, M. S. Bonkowski, et al., “Impairment of an Endothelial NAD+-H2S Signaling
Network Is a Reversible Cause of Vascular Aging,” Cell 173, no. 1 (March 22, 2018): 74–89,
https://www.cel .com/cel /pdf/S0092-8674(18)30152-1.pdf.
THREE. THE BLIND EPIDEMIC
1. F. Bacon, Of the Proficience and Advancement of Learning, Divine and Human (Oxford, UK: Leon Lich eld, 1605). An original of this book sits on our mantelpiece at home, a gift
from Sandra, my wife.
2. C. Kenyon, J. Chang, E. Gensch, et al., “A C. elegans Mutant That Lives Twice as Long as Wild Type,” Nature 366, no. 6454 (December 2, 1993): 461–64,
https://www.nature.com/articles/366461a0.
3. L. Partridge and P. H. Harvey, “Methuselah Among Nematodes,” Nature 366, no. 6454
(December 2, 1993): 404–5, https://www.ncbi.nlm.nih.gov/pubmed/8247143.
4. “Decelerated aging,” Gems wrote, “has an element of tragic inevitability: its bene ts to health compel us to pursue it, despite the transformation of human society, and even human
nature, that this could entail.” D. Gems, “Tragedy and Delight: The Ethics of Decelerated
Ageing,” Philosophical Transactions of the Royal Society B: Biological Sciences 366 (January 12,
2011): 108–12, https://royalsocietypublishing.org/doi/pdf/10.1098/rstb.2010.0288.
5. “You know the cartoon where Bugs Bunny is driving an old car that suddenly fal s apart, every bolt sprung, with the last hubcap rattling in a circle until it comes to rest?” Washington
Post reporter David Brown wrote in 2010. “Some people die like that, too. The trouble is
there’s not a good name for it.” D. Brown, “Is It Time to Bring Back ‘Old Age’ as a Cause ofDeath?” Washington Post, September 17, 2010, http://www.washingtonpost.com/wp-
dyn/content/article/2010/09/17/AR2010091703823.html?sid=ST2010091705724.
6. “Real y, people don’t die of old age,” Chris Wel er wrote on Medical Daily. “Something else has to be going on.” C. Wel er, “Can People Real y Die of Old Age?,” “The Unexamined
Life,” Medical Daily, January 21, 2015, http://www.medicaldaily.com/can-people-real y-die-
7. B. Gompertz, “On the Nature of the Function Expressive of the Law of Human Mortality,
and on a New Mode of Determining the Value of Life Contingencies,” Philosophical
Transactions of the Royal Society 115 (January 1, 1825): 513–85,
https://royalsocietypublishing.org/doi/10.1098/rstl.1825.0026.
8. D. A. Sinclair and L. Guarente, “Extrachromosomal rDNA Circles—A Cause of Aging in
Yeast,”
Cell
91,
no.
7
(December
26,
1997):
1033–42,
https://www.ncbi.nlm.nih.gov/pubmed/9428525.
9. Based on global population estimates and census reports, among other sources, the World Bank plotted out a fty-six-year period ending in 2016 that showed life expectancy increasing
from 52 to 72. “Life Expectancy at Birth, Total (Years),” The World Bank,
https://data.worldbank.org/indicator/SP.DYN.LE00.IN.
10. I inherited the SERPINA1 mutation from my mother. Even though I have never smoked, I
nd it hard to breathe in some situations, such as when I am visiting a place with substantial
pol ution. Armed with this information, I avoid breathing in dust and other contaminants
when possible. I feel empowered knowing the genetic instructions within each of my cel s, an
experience that previous generations never had.
11. A. M. Binder, C. Corvalan, V. Mericq, et al., “Faster Ticking Rate of the Epigenetic Clock Is Associated with Faster Pubertal Development in Girls,” Epigenetics 13, no. 1 (February 15,
2018): 85–94, https://www.tandfonline.com/doi/ful /10.1080/15592294.2017.1414127.
12. Women over 65 are more prone to hip fractures, with sepsis being the main cause of death.
Researchers have linked the sepsis to poor medical care, a lack of family support, and
dementia. “Time wise, mortality was found to be higher within the rst six months, with 10
deaths (50%), and within the rst year, with six deaths (30%).” J. Negrete-Corona, J. C.
Alvarano-Soriano, and L. A. Reyes-Santiago, “Hip Fracture as Risk Factor for Mortality in
Patients over 65 Years of Age. Case-Control Study” (abstract translation from Spanish), Acta
Ortopédica Mexicana 28, no. 6 (November–December 2014): 352–62,
https://www.ncbi.nlm.nih.gov/pubmed/26016287,
(Spanish)
http://www.medigraphic.com/pdfs/ortope/or-2014/or146c.pdf.
13. Up to 74 percent of patients who have a foot amputated due to diabetes die within ve years of surgery. The authors argue for more aggressive focus on the issue by doctors and patients
alike. “New-onset diabetic foot ulcers should be considered a marker for signi cantly
increased mortality and should be aggressively managed local y, systemical y, and
psychological y.”J. M. Robbins, G. Strauss, D. Aron, et al., “Mortality Rates and DiabeticFoot Ulcers: Is It Time to Communicate Mortality Risk to Patients with Diabetic Foot
Ulceration?,” Journal of the American Podiatric Medical Association 98, no. 6 (November–
December 2008): 489–93, https://www.ncbi.nlm.nih.gov/pubmed/19017860.
14. Have we made a deal with the medical devil that’s back red? Olshansky certainly thinks so, contrasting the quest for human longevity and health to the dark narrative of Faust’s
ultimately pyrrhic deal with Mephistopheles. “It’s possible that humanity has squeezed
about as much healthy life out of public health interventions as possible and that the human
body is now running up against inherent limits that the genetical y xed attributes of our
biology impose.” S. J. Olshansky, “The Future of Health,” Journal of the American Geriatrics
Society
66,
no.
1
(December
5,
2017):
195–97,
https://onlinelibrary.wiley.com/doi/ful /10.1111/jgs.15167.
15. The numbers are indeed staggering: close to 800,000 Americans die annual y of cardiovascular-related diseases; medical costs related to cardiovascular issues are expected to
be over $818 bil ion by 2030 and lost productivity costs above $275 bil ion. “Heart Disease
and Stroke Cost America Nearly $1 Bil ion a Day in Medical Costs, Lost Productivity,”
CDC Foundation, April 29, 2015, https://www.cdcfoundation.org/pr/2015/heart-disease-
and-stroke-cost-america-nearly-1-bil ion-day-medical-costs-lost-productivity.
16. As treatments for patients with disease have prolonged their lives, so the amount of disease in society has augmented. This situation means that the only way to increase the human
healthspan wil be by “ ‘delaying aging,’ or delaying the physiological change that results in
disease and disability,” the author argues. Along with scienti c breakthroughs, changes in
socioeconomic inequalities, lifestyle, and behavior can contribute to improving both
healthspan and lifespan. E. M. Crimmins, “Lifespan and Healthspan: Past, Present, and
Promise,”
Gerontologist
55,
no.
6
(December
2015):
901–11,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4861644/.
17. According to the World Health Organization, one DALY can be thought of as one lost year of “healthy” life. The sum of these DALYs across the population, or the burden of disease,
can be thought of as a measurement of the gap between current health status and an ideal
health situation in which the entire population lives to an advanced age, free of disease and
disability. “Metrics: Disability-Adjusted Life Year (DALY),” World Health Organization,
https://www.who.int/healthinfo/global_burden_disease/metrics_daly/en/.
18. And almost everyone at that age spends a considerable part of his or her life visiting the doctor. According to the study, published in 2009 by the British Medical Journal, 94 percent
of 85-year-olds had had contact with a doctor in the past year, and one in ten was in
institutional care. J. Col erton, K. Davies, C. Jagger, et al., “Health and Disease in 85 Year
Olds: Baseline Findings from the Newcastle 85+ Cohort Study,” British Medical Journal,
December 23, 2009, https://www.bmj.com/content/339/bmj.b4904.
19. The possibility that both genetic and epigenetic aging are needed for a tumor to develop we’ve termed “geroncogenesis,” and it explains why tumors don’t occur in young peopleeven after extreme sun exposure, why it often takes decades for DNA damage to lead to a
tumor even if you avoid the sun later in life, and why cancers often have an unusual
metabolism (named after the physicist Otto Warburg), one that directly consumes glucose,
has decreased mitochondrial activity, and uses less oxygen to make energy, similar to the
metabolism of old cel s.
20. According to the World Health Organization, “The State of Global Tobacco Control,” 2008,
http://www.who.int/tobacco/mpower/mpower_report_global_control_2008.pdf.
21. R. A. Mil er, “Extending Life: Scienti c Prospects and Political Obstacles,” Milbank Quarterly
80,
no.
1
(March
2002):
155–74,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2690099/; graph redrawn from D. L.
Hoyert, K. D. Kochanek, and S. L. Murphy, “Deaths: Final Data for 1997,” National Vital
Statistics
Report
47,
no.
19
(June
30,
1999):1–104,
https://www.ncbi.nlm.nih.gov/pubmed/10410536.
22. Using a survey of 593 people that was then repeated four years later, the authors explored the role of “subjective age” (meaning how old an individual feels in contrast to his or her
biological age) in shaping the process of aging. A. E. Kornadt, T. M. Hess, P. Voss, and K.
Rothermund, “Subjective Age Across the Life Span: A Di erentiated, Longitudinal
Approach,” Journals of Gerontology: Psychological Sciences 73, no. 5 (June 1, 2018): 767–77,
http://europepmc.org/abstract/med/27334638.
23. “David A. Sinclair’s Past and Present Advisory Roles, Board Positions, Funding Sources, Licensed Inventions, Investments, Funding, and Invited Talks,” Sinclair Lab, Harvard
Medical School, November 15, 2018, https://genetics.med.harvard.edu/sinclair-
test/people/sinclair-other.php.
FOUR. LONGEVITY NOW
1. It seems likely that he had sex at least once again, as he had one daughter, Clara, with his wife, Veronica. L. Cornaro, Sure and Certain Methods of Attaining a Long and Healthful Life:
With Means of Correcting a Bad Constitution, &c. , https://babel.hathitrust.org/cgi/pt?
id=dul1.ark:/13960/t0dv2fm86;view=1up;seq=1.
2. There are other translations. This comes from the edition published in Milwaukee by Wil iam F. Butler in 1903.
3. A 3-year-old rat measured in terms of human lifespan would be akin to a 90-year-old human, according to a researcher quoted by the authors. One of their rats, raised on an experimental
diet from 6 weeks of age, lived to 40 months, while of those rats raised on a normal diet, the
oldest reached 34 months, with “less than a third of the rats in our colony . . . expected to live
to be more than two years old.” T. B. Osborne, L. B. Mendel, and E. L. Ferry, “The E ect of
Retardation of Growth upon the Breeding Period and Duration of Life of Rats,” Science 45, no. 1160 (March 23, 1917): 294–95, http://science.sciencemag.org/content/45/1160/294.4. I. Bjedov, J. M. Toivonen, F. Kerr, et al., “Mechanisms of Life Span Extension by Rapamycin in the Fruit Fly Drosophila melanogaster,” Cell Metabolism 11, no. 1 (January 6, 2010): 35–
46, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2824086/.
5. Among Kagawa’s ndings on the impact of Western diets on the Japanese were signi cant increases in colon and lung cancer and decreases in stomach and uterine cancers, although
the subjects’ food consumption was stil much smal er than that of Americans or Europeans.
When he looked at the residents of Okinawa, they had “the lowest total energy, sugar and
salt, and the smal est physique, but had healthy longevity and the highest centenarian rate.”
Y. Kagawa, “Impact of Westernization on the Nutrition of Japanese: Changes in Physique,
Cancer, Longevity and Centenarians,” Preventive Medicine 7, no. 2 (June 1978): 205–17,
https://www.sciencedirect.com/science/article/pi /0091743578902463.
6. Two of the authors of the report were themselves part of the crew who elected to be locked up inside the Biosphere for two years and live on a low-calorie diet, with just 12 percent
protein and 11 percent fat in terms of calorie consumption. Despite this calorie restriction
and a 17±5 percent weight loss, al eight crew members were healthy and highly active during
the two-year period. R. L. Walford, D. Mock, R. Verdery, and T. MacCal um, “Calorie
Restriction in Biosphere 2: Alterations in Physiologic, Hematologic, Hormonal, and
Biochemical Parameters in Humans Restricted for a 2-Year Period,” Journals of Gerontology,
Series A: Biological Sciences and Medical Sciences 57, no. 6 (June 2002): 211–24,
https://www.ncbi.nlm.nih.gov/pubmed/12023257.
7. L. K. Heilbronn, and E. Ravussin, “Calorie Restriction and Aging: Review of the Literature and Implications for Studies in Humans,” American Journal of Clinical Nutrition 3, no. 178
(September 2003): 361–69, https://academic.oup.com/ajcn/article/78/3/361/4689958.
8. The authors used the results of a publicly accessible, 24-month trial run by the National Institute on Aging of calorie restriction in nonobese youth. D. W. Belsky, K. M. Hu man,
C. F. Pieper, et al., “Change in the Rate of Biological Aging in Response to Caloric
Restriction: CALERIE Biobank Analysis,” Journals of Gerontology, Series A: Biological
Sciences
and
Medical
Sciences
73,
no.
1
(January
2018):
4–10,
https://academic.oup.com/biomedgerontology/article/73/1/4/3834057.
9. McGlothin wrote in an article, “I am delighted that a 70-year-old can have biomarkers that are like those of a healthy school-age child.” P. McGlothin, “Growing Older and Healthier
the
CR
Way®,”
Life
Extension
Magazine,
September
2018,
https://www.lifeextension.com/Magazine/2018/9/Calorie-Restriction-Update/Page-01.
10. The authors are in no doubt of the potential bene ts calorie restriction o ers humans in terms of addressing diseases and aging. “A clear understanding of the biology of ageing, as
opposed to the biology of individual age-related diseases, could be the critical turning point
for novel approaches in preventative strategies to facilitate healthy human ageing,” they
wrote. “Caloric restriction (CR) o ers a powerful paradigm to uncover the cel ular and molecular basis for the age-related increase in overal disease vulnerability that is shared by almammalian species.” J. A. Mattison, R. J. Colman, T. M. Beasley, et al., “Caloric Restriction
Improves Health and Survival of Rhesus Monkeys,” Nature Communications, January 17,
2017, https://www.nature.com/articles/ncomms14063.
11. Y. Zhang, A. Bokov, J. Gelfond, et al., “Rapamycin Extends Life and Health in C57BL/6
Mice,” Journals of Gerontology, Series A: Biological Sciences and Medical Sciences 69, no. 2
(February 2014): 119–30, https://www.ncbi.nlm.nih.gov/pubmed/23682161.
12. “We real y study this as a paradigm to understand aging,” she told Scientific American in 2017. “We’re not recommending people do it.” R. Conni , “The Hunger Gains: Extreme
Calorie-Restriction Diet Shows Anti-aging Results,” Scientific American, February 16, 2017,
https://www.scienti camerican.com/article/the-hunger-gains-extreme-calorie-restriction-
diet-shows-anti-aging-results/.
13. “The optimum amount of fasting appeared to be fasting 1 day in 3 and this increased the life span of littermate males about 20% and littermate females about 15%.” A. J. Carlson and F.
Hoelzel, “Apparent Prolongation of the Life Span of Rats by Intermittent Fasting: One
Figure,” Journal of Nutrition 31, no. 3 (March 1, 1946): 363–75,
https://academic.oup.com/jn/article-abstract/31/3/363/4725632?redirectedFrom=ful text.
14. H. M. Shelton, “The Science and Fine Art of Fasting,” in The Hygienic System, vol. III, Fasting and Sunbathing (San Antonio, Texas: Dr. Shelton’s Health School, 1934).
15. C. Tazearslan, J. Huang, N. Barzilai, and Y. Suh, “Impaired IGF1R Signaling in Cel s Expressing Longevity-Associated Human IGF1R Al eles,” Aging Cell 10, no. 3 (June 2011):
551–54, https://onlinelibrary.wiley.com/doi/ful /10.1111/j.1474-9726.2011.00697.
16. One in three Ikarians reaches the age of 90, and most do so free of dementia and many other chronic diseases of aging. “Ikaria, Greece. The Island where People Forget to Die,” Blue
Zones, https://www.bluezones.com/exploration/ikaria-greece/.
17. The fasting extends to 180 days in the year and requires abstinence from primarily dairy products and red-blooded animals and sh, which means that octopus and squid can stil be
eaten. In the run-up to Holy Communion, fasting encompasses al food. N. Gaifyl ia,
“Greek Orthodox 2018 Calendar of Holidays and Fasts,” The Spruce Eats, October 6, 2018,
https://www.thespruceeats.com/greek-orthodox-calendar-1706215.
18. Bapan has been widely ignored by Western researchers, mostly because people in that part of southern China—a region long reputed to have large populations of very healthy
centenarians—don’t have formal birth records. The cardiologist John Day and his col eagues,
however, have argued that there is good reason to believe their claims. J. D. Day, J. A. Day,
and M. LaPlante, The Longevity Plan: Seven Life-Transforming Lessons from Ancient China
(New York: HarperCol ins, 2017).
19. Avoiding animal protein is not easy. One of the main reasons is that protein consumption produces satiety. No one has done more to understand why eating carbohydrates doesn’t
stave o hunger than Stephen Simpson, the director of the Charles Perkins Centre in Sydney, Australia. Simpson started his career trying to understand why locusts swarm. If hecould gure that out, he felt, perhaps he could prevent the global loss of mil ions of tons of
crops each year. What he discovered was that locusts seek protein. They crave it. They march
along consuming anything edible in their path, but if there’s not enough protein in their
diet, they transform into ravenous, hungry creatures that seek protein from any possible
source. And the closest source of protein is the locust in front of it. Under these conditions,
the best way to stay alive is to keep moving forward, occasional y pausing to snack on a
slower relative. Simpson’s latest work is fascinating: it shows that this same trigger exists in
the mammalian brain. When we lack protein, we also turn ravenous, and although we don’t
normal y try to eat our neighbors, in the throes of extreme hunger, who hasn’t considered it?
What this al tel s us is that it’s best not to eat a lot of animal protein, but it’s hard to avoid it
altogether. F. P. Zanotto, D. Raubenheimer, and S. J. Simpson, “Selective Egestion of Lysine
by Locusts Fed Nutritional y Unbalanced Foods,” Journal of Insect Physiology 40, no. 3
(March
1994):
259–65,
https://www.sciencedirect.com/science/article/pi /0022191094900493.
20. Though it seems the occasional hot dog or hamburger is acceptable, a review of 800 studies by twenty-two experts found that a daily diet that included 50 grams of processed meat
appeared to increase subjects’ chances of developing colorectal cancer by 18 percent. S.
Simon, “World Health Organization Says Processed Meat Causes Cancer,” American Cancer
Society, October 26, 2015, https://www.cancer.org/latest-news/world-health-organization-
says-processed-meat-causes-cancer.html.
21. With processed, calorie-rich food largely absent from their diet and a lifestyle dominated by physical activity, there’s little in the way of obesity or cardiovascular disease to be found in
hunter-gatherer communities. H. Pontzer, B. M. Wood, and D. A. Raichlen, “Hunter-
Gatherers as Models in Public Health,” Obesity Reviews 19, suppl. 1 (December 2018): 24–
35, https://onlinelibrary.wiley.com/doi/ful /10.1111/obr.12785.
22. M. Song, T. T. Fung, F. B. Hu, et al., “Association of Animal and Plant Protein Intake with Al -Cause and Cause-Speci c Mortality,” JAMA Internal Medicine 176, no. 10 (October 1,
2016):
1453–63,
https://jamanetwork.com/journals/jamainternalmedicine/ful article/2540540.
23. A 2011 study identi ed a new signaling pathway used by amino acids to activate mTOR. I.
Tato, R. Bartrons, F. Ventura, and J. L. Rosa, “Amino Acids Activate Mammalian Target of
Rapamycin Complex 2 (mTORC2) via PI3K/Akt Signaling,” Journal of Biological
Chemistry
286,
no.
8
(February
25,
2011):
6128–42,
http://www.jbc.org/content/286/8/6128.ful .
24. C. Hine, C. Mitchel , and J. R. Mitchel , “Calorie Restriction and Methionine Restriction in Control of Endogenous Hydrogen Sul de Production by the Transsulfuration Pathway,”
ExperimentalGerontology
68
(August
2015):
26–32,
https://www.ncbi.nlm.nih.gov/pubmed/25523462.
25. Rather than caloric restriction, the researchers at the Lamming Lab devised a short-term methionine deprivation regimen that reduced fat mass, restored normal body weight, and
reinstituted glycemic control to male and female mice alike. D. Yu, S. E. Yang, B. R. Mil er, et
al., “Short-Term Methionine Deprivation Improves Metabolic Health via Sexual y
Dimorphic, mTORC1-Independent Mechanisms,” FASEB Journal 32, no. 6 (June 2018):
3471–82, https://www.ncbi.nlm.nih.gov/pubmed/29401631.
26. The eternal quest for a wel -balanced diet, the answer, the authors suggest, may have to do with how “longevity can be extended in ad libitum–fed animals by manipulating the ratio of
macronutrients to inhibit mTOR activation.” S. M. Solon-Biet, A. C. McMahon, J. W.
Bal ard, et al., “The Ratio of Macronutrients, Not Caloric Intake, Dictates Cardiometabolic
Health, Aging, and Longevity in Ad Libitum–Fed Mice,” Cell Metabolism 3, no. 19 (March
4, 2014): 418–30, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5087279/.
27. In other words, the speci c amino acid composition of a person’s diet may be more important than limiting al aminos. The easiest way to do this, though, is stil to reduce meat
intake. L. Fontana, N. E. Cummings, S. I. Arriola Apelo, et al., “Decreased Consumption of
Branched Chain Amino Acids Improves Metabolic Health,” Cell Reports 16, no. 2 (July 12,
2016): 520–30, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4947548/.
28. Some have suggested that a better understanding of this connection could help researchers develop mTOR-targeted therapies to prevent muscle wasting. M.-S. Yoon, “mTOR as a Key
Regulator in Maintaining Skeletal Muscle Mass,” Frontiers in Physiology 8 (2017): (October
17, 2017): 788, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5650960/.
29. Just cutting one’s consumption of branched-chain amino acids for one day rapidly improves insulin sensitivity. F. Xiao, J. Yu, Y. Guo, et al., “E ects of Individual Branched-Chain
Amino Acids Ceprivation on Insulin Sensitivity and Glucose Metabolism in Mice,”
Metabolism
63,
no.
6
(June
2014):
841–50,
https://www.ncbi.nlm.nih.gov/pubmed/24684822/.
30. There are certainly other lifestyle factors at play. But a meta-analysis of seven studies including nearly 125,000 participants, published in 2012 in Annals of Nutrition and
Metabolism, is compel ing evidence. Among vegetarians, the researchers who conducted the
study observed a 16 percent lower mortality from circulatory diseases and a 12 percent lower
mortality from cerebrovascular disease. T. Huang, B. Yang, J. Zheng, et al., “Cardiovascular
Disease Mortality and Cancer Incidence in Vegetarians: A Meta-analysis and Systematic
Review,” Annals of Nutrition & Metabolism 4, no. 60 (June 1, 2012): 233–40,
https://www.karger.com/Article/Ful Text/337301.
31. The study looked at nearly 6,000 men and women enrol ed in the National Health and Nutrition Examination Survey. If you want a reminder of how little a sedentary life does for
prolonging existence, the fol owing jumps out of the report: “Adults with High activity were
estimated to have a biologic aging advantage of 9 years (140 base pairs ÷ 15.6) over Sedentary adults. The di erence in cel aging between those with High and Low activity was alsosigni cant, 8.8 years, as was the di erence between those with High and Moderate PA (7.1
years).” L. A. Tucker, “Physical Activity and Telomere Length in U.S. Men and Women: An
NHANES Investigation,” Preventive Medicine 100 (July 2017): 145–51,
https://www.sciencedirect.com/science/article/pi /S0091743517301470.
32. Intrigued by the potential insights into aging a orded by the health and physicality of middle-aged, regular bike riders, British scientists, among whose ranks were recreational
athletes, looked at how exercise might in uence longevity. They recruited older male and
female cyclists between 55 and 79 for their study and contrasted them to older and younger
sedentary people. “The cyclists proved to have re exes, memories, balance and metabolic
pro les that more closely resembled those of 30-year-olds than of the sedentary older group.”
G. Reynolds, “How Exercise Can Keep Aging Muscles and Immune Systems ‘Young,’ ” New
York Times, March 14, 2018, https://www.nytimes.com/2018/03/14/wel /move/how-
exercise-can-keep-aging-muscles-and-immune-systems-young.html.
33. D. Lee, R. R. Pate, C. J. Lavie, et al., “Leisure-Time Running Reduces Al -Cause and Cardiovascular Mortality Risk,” Journal of the American College of Cardiology 54, no. 5
(August 2014): 472–81, http://www.onlinejacc.org/content/64/5/472.
34. The authors show how cardiorespiratory tness algorithms can identify those at risk of cardiovascular disease and also potential y help to develop appropriate exercise regimes
depending on an individual’s initial tness level. E. G. Artero, A. S. Jackson, X. Sui, et al.,
“Longitudinal Algorithms to Estimate Cardiorespiratory Fitness: Associations with
Nonfatal Cardiovascular Disease and Disease-Speci c Mortality,” Journal of the American
College
of
Cardiology
63,
no.
21
(June
3,
2014):
2289–96,
https://www.sciencedirect.com/science/article/pi /S0735109714016301?via%3Dihub.
35. T. S. Church, C. P. Earnest, J. S. Skinner, and S. N. Blair, “E ects of Di erent Doses of Physical Activity on Cardiorespiratory Fitness Among Sedentary, Overweight or Obese
Postmenopausal Women with Elevated Blood Pressure: A Randomized Control ed Trial,”
Journal of the American Medical Association 297, no. 19 (May 16, 2007): 2081–91,
https://jamanetwork.com/journals/jama/ful article/1108370.
36. M. M. Robinson, S. Dasari, A. R. Konopka, et al., “Enhanced Protein Translation Underlies Improved Metabolic and Physical Adaptations to Di erent Exercise Training Modes in
Young and Old Humans,” Cell Metabolism 25, no. 3 (March 7, 2017): 581–92,
https://www.cel .com/cel -metabolism/comments/S1550-4131(17)30099-2.
37. Sage recommendations from the Mayo Clinic include dedicating 150 minutes a week to activities such as swimming or mowing the lawn or doing 75 minutes of more demanding
exercise, such as spinning or running. “Be realistic and don’t push yourself too hard, too
fast,” the clinic sta wrote. “Fitness is a lifetime commitment, not a sprint to a nish line,”
“Exercise Intensity: How to Measure It,” Mayo Clinic, June 12, 2018,
https://www.mayoclinic.org/healthy-lifestyle/ tness/in-depth/exercise-intensity/art-38. Investigating how the hypothalamus potential y controls aspects of aging, the authors found that “immune inhibition or GnRH restoration in the hypothalamus/brain” o ers two
possible directions for extending lifespan and ghting health issues that come with aging. G.
Zhang, J. Li, S. Purkayasatha, et al., “Hypothalamic Programming of Systemic Ageing
Involving IKK-β, NF-ĸB and GnRH,” Nature 497, no. 7448 (May 9, 2013): 211–16,
https://www.nature.com/articles/nature12143.
39. The team couldn’t say why this happened, only that it happened. Back then they theorized that lowering the mice’s body temperature might slow down metabolism and thus reduce
the notorious free radicals. We’ve learned a lot since then. B. Conti, M. Sanchez-Alvarez, R.
Winskey-Sommerer, et al., “Transgenic Mice with a Reduced Core Body Temperature Have
an Increased Life Span,” Science 314, no. 5800 (November 3, 2006): 825–28,
https://www.ncbi.nlm.nih.gov/pubmed/17082459.
40. The mice su ered from increased rates of obesity, beta cel dysfunction, and type 2 diabetes.
C.- Y. Zhang, G. Ba y, P. Perret, et al., “Uncoupling Protein-2 Negatively Regulates Insulin
Secretion and Is a Major Link Between Obesity, β Cel Dysfunction, and Type 2 Diabetes,”
Cell
105,
no.
6
(June
15,
2001):
745–55,
https://www.sciencedirect.com/science/article/pi /S0092867401003786.
41. The researchers also believed this occurred because of a reduction in oxidative damage. Y.-W.
C. Fridel , A. Sánchez-Blanco, B. A. Silvia, et al., “Targeted Expression of the Human
Uncoupling Protein 2 (hUCP2) to Adult Neurons Extends Life Span in the Fly,” Cell
Metabolism
1,
no.
2
(February
2005):
145–52,
https://www.sciencedirect.com/science/article/pi /S155041310500032X.
42. The researchers concluded that UCP2 regulates brown adipose tissue thermogenesis through nonesteri ed fatty acids. A. Caron, S. M. Labbé, S. Carter, et al., “Loss of UCP2 Impairs
Cold-Induced Non-shivering Thermogenesis by Promoting a Shift Toward Glucose
Utilization in Brown Adipose Tissue,” Biochimie 134 (March 2007): 118–26,
https://www.sciencedirect.com/science/article/pi /S030090841630270X?via%3Dihub.
43. The researchers, led by Justin Darcy at the University of Alabama, demonstrated enhanced brown adipose tissue function in animals that lived 40 to 60 percent longer than their
littermates. J. Darcy, M. McFadden, Y. Fang, et al., “Brown Adipose Tissue Function Is
Enhanced in Long-Lived, Male Ames Dwarf Mice,” Endocrinology 157, no. 12 (December 1,
2016): 4744–53, https://academic.oup.com/endo/article/157/12/4744/2758430.
44. “How brown fat is regulated in humans and how it relates to metabolism, though, remain unclear,” the authors of a study wrote in 2014. Since then the mechanism has become
clearer. Endocrine Society, “Cold Exposure Stimulates Bene cial Brown Fat Growth,”
Science
Daily,
June
23,
2014,
https://www.sciencedaily.com/releases/2014/06/140623091949.htm.
45. T. Shi, F. Wang, E. Stieren, and Q. Tong, “SIRT3, a Mitochondrial Sirtuin Deacetylase, Regulates Mitochondrial Function and Thermogenesis in Brown Adipocytes,” Journal ofBiological
Chemistry
280,
no.
14
(April
8,
2005):
13560-67,
http://www.jbc.org/content/280/14/13560.long.
46. A. S. Warthin, “A Fatal Case of Toxic Jaundice Caused by Dinitrophenol,” Bulletin of the International Association of Medical Museums 7 (1918): 123–26.
47. W. C. Cutting, H. G. Mertrens, and M. L. Tainter, “Actions and Uses of Dinitrophenol: Promising Metabolic Applications,” Journal of the American Medical Association 101, no. 3
(July 15, 1933): 193–95, https://jamanetwork.com/journals/jama/article-abstract/244026.
48. The authors calculated that with 1.2 mil ion capsules supplied by the Stanford Clinics in 1934, that corresponded to 4,500 patients taking the drug over a three-month period.
Overal , they estimated that in the United States, at least 100,000 people had been treated
with the drug. M. L. Tainter, W. C. Cutting, and A. B. Stockton, “Use of Dinitrophenol in
Nutritional Disorders: A Critical Survey of Clinical Results,” American Journal of Public
Health
24,
no.
10
(1935):
1045–53,
https://ajph.aphapublications.org/doi/pdf/10.2105/AJPH.24.10.1045.
49. Dinitrophenol has a variety of names on the internet. The authors list, along with DNP,
“ ‘Dinosan,’ ‘Dnoc,’ ‘Solfo Black,’ ‘Nitrophen,’ ‘Alidfen,’ and ‘Chemox.’ ” In the 2000s, there
was a spike in DNP-related deaths as it was marketed online to bodybuilders and the weight
conscious. J. Grundlingh, P. I. Dargan, M. El-Zanfaly, and D. M. Wood, “2,4-Dinitrophenol
(DNP): A Weight Loss Agent with Signi cant Acute Toxicity and Risk of Death,” Journal of
Medical
Toxicology
7,
no.
3
(September
2011):
205–12,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3550200/.
50. T. L. Kurt, R. Anderson, C. Petty, et al., “Dinitrophenol in Weight Loss: The Poison Center and Public Health Safety,” Veterinary and Human Toxicology 28, no. 6 (December 1986):
574–75, https://www.ncbi.nlm.nih.gov/pubmed/3788046.
51. A horrifying death from an overdose of DNP is described in a story on Vice; see G. Haynes,
“The Kil er Weight Loss Drug DNP Is Stil Claiming Young Lives,” Vice, August 6, 2018,
https://www.vice.com/en_uk/article/bjbyw5/the-kil er-weight-loss-drug-dnp-is-stil -
claiming-young-lives; see also Grundlingh et al., “2,4-Dinitrophenol (DNP).”
52. This happens di erently from species to species, but the general trend is clear: cold and exercise together build brown fat. F. J. May, L. A. Baer, A. C. Lehnig, et al., “Lipidomic
Adaptations in White and Brown Adipose Tissue in Response to Exercise Demonstrates
Molecular Species-Speci c Remodeling,” Cell Reports 18, no. 6 (February 7, 2017): 1558–
72, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5558157/.
53. “Until further research is available,” an international team of researchers concluded in 2014,
“athletes should remain cognizant that less expensive modes of cryotherapy, such as local ice-
pack application or cold-water immersion, o er comparable physiological and clinical
e ects.” C. M. Bleakley, F. Bieuzen, G. W. Davison, and J. T. Costel o, “Whole-Body
Cryotherapy: Empirical Evidence and Theoretical Perspectives,” Open Access Journal of Sports Medicine5
(March
10,
2014):
25–36,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3956737/.
54. The average time spent in the sauna was 15 minutes at 80°C. T. E. Strandberg, A.
Strandberg, K. Pitkälä, and A. Benetos, “Sauna Bathing, Health, and Quality of Life Among
Octogenarian Men: The Helsinki Businessmen Study,” Aging Clinical and Experimental
Research
30,
no.
9
(September
2018):
1053–57,
https://www.ncbi.nlm.nih.gov/pubmed/29188579.
55. T. Laukkanen, H. Khan, F. Zaccardi, and J. A. Laukkanen, “Association Between Sauna Bathing and Fatal Cardiovascular and Al -Cause Mortality Events,” JAMA Internal
Medicine
175,
no.
4
(April
2015):
542–48,
https://www.ncbi.nlm.nih.gov/pubmed/25705824.
56. H. Yang, T. Yang, J. A. Baur, et al., “Nutrient-Sensitive Mitochondrial NAD+ Levels Dictate Cel Survival,” Cell 130, no. 6 (September 21, 2007): 1095–107,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3366687/.
57. R. Madabhushi, F. Gao, A. R. Pfenning, et al., “Activity-Induced DNA Breaks Govern the Expression of Neuronal Early-Response Genes,” Cell 161, no. 7 (June 18, 2015): 1592–605,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4886855/.
58. H. Katoka, “Quantitation of Amino Acids and Amines by Chromatography,” Journal of Chromatography
Library
70
(2005):
364–404,
https://www.sciencedirect.com/topics/chemistry/aromatic-amine.
59. Another highly prevalent chemical used in plastic bottles and food and drink cans is bisphenol A, or BPA. It’s so ubiquitous that it can be found in the urine of nearly every
American; in high quantities it has been linked to “cardiovascular disease and diabetes and
may be associated with an increased risk for miscarriages with abnormal embryonic
karyotype.” P. Al ard and M. P. Colaiácovo, “Bisphenol A Impairs the Double-Strand Break
Repair Machinery in the Germline and Causes Chromosome Abnormalities,” Proceedings of
the National Academy of Sciences of the United States of America 107, no. 47 (November 23,
2010): 20405–10, http://www.pnas.org/content/107/47/20405.
60. “Our ndings suggest that this colorant could cause harmful e ects to humans if it is metabolized or absorbed through the skin.” F. M. Chequer, V. de Paula Venâncio, et al.,
“The Cosmetic Dye Quinoline Yel ow Causes DNA Damage in Vitro,” Mutation
Research/Genetic Toxicology and Environmental Mutagenesis 777 (January 1, 2015): 54–61,
https://www.ncbi.nlm.nih.gov/pubmed/25726175.
61. Beer drinkers take note: “Beer is one source of NDMA, in which as much as 70 micrograms l(-1) has been reported in some types of German beer, although usual levels are much lower
(10 or 5 micrograms l(-1)); this could mean a considerable intake for a heavy beer drinker of
several liters per day.” The good news, the writer adds, is that in recent decades there’s been
not only a reduction in the level of nitrates in food but also “greater control of exposure of
malt to nitrogen oxides in beer making.” W. Lijinsky, “N-Nitroso Compounds in the Diet,”Mutation
Research
443,
nos.
1–2
(July
15,
1999):
129–38,
https://www.ncbi.nlm.nih.gov/pubmed/10415436.
62. L. Robbiano, E. Mereto, C. Corbu, and G. Brambil a, “DNA Damage Induced by Seven N-
nitroso Compounds in Primary Cultures of Human and Rat Kidney Cel s,” Mutation
Research 368, no. 1 (May 1996): 41–47, https://www.ncbi.nlm.nih.gov/pubmed/8637509.
63. The state of Massachusetts did a study in 1988 to get to grips with the prevalence of radon by county. It found that one in four houses apparently had levels over the EPA-identi ed level
of 4pCi/L, which requires additional investigation. “Public Health Fact Sheet on Radon,”
Health and Human Services, Commonwealth of Massachusetts, 2011,
http://web.archive.org/web/20111121032816/http://www.mass.gov/eohhs/consumer/com
munity-health/environmental-health/exposure-topics/radiation/radon/public-health-fact-
64. “Most of the mercury that contaminates sh comes from household and industrial waste that is incinerated or released during the burning of coal and other fossil fuels. Products
containing mercury that are improperly thrown in the garbage or washed down drains end
up in land l s, incinerators, or sewage treatment facilities.” “Contaminants in Fish,”
Washington
State
Department
of
Health,
https://www.doh.wa.gov/CommunityandEnvironment/Food/Fish/ContaminantsinFish.
65. S. Horvath, “DNA Methylation Age of Human Tissues and Cel Types,” Genome Biology 14, no. 10 (2013): R115, https://www.ncbi.nlm.nih.gov/pubmed/24138928.
FIVE. A BETTER PILL TO SWALLOW
1. If Schrödinger couldn’t exactly answer the question of what life is, his book arguably did everything else but that. It’s credited with being a key in uencer of the development of
scienti c thought in the twentieth century and helped lay the groundwork for the emergence
of molecular biology and the discovery of DNA. E. Schrödinger, What Is Life? The Physical
Aspect of the Living Cell (Cambridge, UK: Cambridge University Press, 1944).
2. V. L. Schramm and S. D. Schwartz, “Promoting Vibrations and the Function of Enzymes.
Emerging Theoretical and Experimental Convergence,” Biochemistry 57, no. 24 (June 19,
2018): 3299–308, https://www.ncbi.nlm.nih.gov/pubmed/29608286.
3. “Cel Size and Scale,” Genetic Science Learning Center, University of Utah,
http://learn.genetics.utah.edu/content/cel s/scale/.
4. The macromolecular biological catalysts whose names end in -ase are enzymes.
5. Out of so many eminent quotes, this is one that scientists hold as wisdom for the ages: “The rst principle is that you must not fool yourself—and you are the easiest person to fool.” R.
P. Feynman, The Quotable Feynman, ed. Michel e Feynman (Princeton, NJ: Princeton
University Press, 2015), 127.
6. After Sehgal’s employer was bought by the international health care company Wyeth, he resumed his work on rapamycin. “In 1999, the U.S. Food and Drug Administrationapproved rapamycin as a drug for transplant patients. Sehgal died a few years after the FDA
approval, too soon to see his brainchild save the lives of thousands of transplant patients and
go on to make Wyeth hundreds of mil ions of dol ars.” B. Gi ord, “Does a Real Anti-aging
Pil
Already
Exist?,”
Bloomberg,
February
12,
2015,
https://www.bloomberg.com/news/features/2015-02-12/does-a-real-anti-aging-pil -already-
7. The authors concluded that “up-regulation of a highly conserved response to starvation-induced stress is important for life span extension by decreased TOR signaling in yeast and
higher eukaryotes.” R. W. Powers III, M. Kaeberlein, S. D. Caldwel , et al., “Extension of
Chronological Life Span in Yeast by Decreased TOR Pathway Signaling,” Genes &
Development
20,
no.
2
(January
15,
2006):
174–84,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1356109/.
8. I. Bjedov, J. M. Toivonen, F. Kerr, et al., “Mechanisms of Life Span Extension by Rapamycin in the Fruit Fly Drosophilia melanogaster,” Cell Metabolism 11, no. 1 (January 6, 2010): 35–
46, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2824086/.
9. The authors noted that these were the rst results to show that mTOR could play a role in extending life: “Rapamycin may extend lifespan by postponing death from cancer, by
retarding mechanisms of ageing, or both.” D. E. Harrison, R. Strong, Z. D. Sharp, et al.,
“Rapamycin Fed Late in Life Extends Lifespan in Genetical y Heterogeneous Mice,” Nature
460 (July 8, 2009): 392–95, https://www.nature.com/articles/nature08221.
10. K. Xie, D. P. Ryan, B. L. Pearson, et al., “Epigenetic Alterations in Longevity Regulators, Reduced Life Span, and Exacerbated Aging-Related Pathology in Old Father O spring
Mice,” Proceedings of the National Academy of Sciences of the United States of America 115,
no. 10 (March 6, 2018): E2348–57, https://www.pnas.org/content/115/10/E2348.
11. How do they pick so many winners? According to a press release, a Thomson Reuters’
executive explained it thus: “Highly-cited papers turn out to be one of the most reliable
indicators of world-class research, and provide a glimpse at what research stands the best
chance at being recognized with a Nobel Prize.” Thomson Reuters, “Web of Science Predicts
2016
Nobel
Prize
Winners,”
PR
Newswire,
September
21,
2016,
https://www.prnewswire.com/news-releases/web-of-science-predicts-2016-nobel-prize-
12. In this case, the authors showed that 3 months of rapamycin increased middle-aged mice’s life expectancy by 60 percent as wel as improving their healthspan. A. Bitto, K. I. Takashi, V. V.
Pineda, et al., “Transient Rapamycin Treatment Can Increase Lifespan and Healthspan in
Middle-Aged
Mice,”
eLife
5
(August
23,
2016):
5,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4996648/.
13. Low-level doses of a drug cal ed everolimus were given to people over 65. Their response to u vaccines improved by around 20 percent. A. Regalado, “Is This the Anti-aging Pil We’veAl Been Waiting For?,” MIT Technology Review, March 28, 2017,
https://www.technologyreview.com/s/603997/is-this-the-anti-aging-pil -weve-al -been-
14. Metformin, given to patients with diabetes, was particularly promising, two researchers noted. “While there are caveats with any study of this nature, the ndings suggest that
metformin may be a ecting basic aging processes that underlie multiple chronic diseases and
not just type II diabetes.” B. K. Kennedy, and J. K. Pennypacker, “Aging Interventions Get
Human,”
Oncotarget
6,
no.
2
(January
2015):
590–91,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4359240/.
15. C. J. Bailey, “Metformin: Historical Overview,” Diabetologia 60 (2017): 1566–76,
https://link.springer.com/content/pdf/10.1007%2Fs00125-017-4318-z.pdf.
16. Patients who took metformin displayed lower rates of mortality not only compared to diabetics but also compared to nondiabetics, the researchers found. Other results included
less cancer and less cardiovascular disease in those being treated with metformin. J. M.
Campbel , S. M. Bel man, M. D. Stephenson, and K. Lisy, “Metformin Reduces Al -Cause
Mortality and Diseases of Ageing Independent of Its E ect on Diabetes Control: A
Systematic Review and Meta-analysis,” Ageing Research Reviews 40 (November 2017): 31–
44, https://www.sciencedirect.com/science/article/pi /S1568163717301472.
17. R. A. DeFronzo, N. Barzilai, and D. C. Simonson, “Mechanism of Metformin Action in
Obese and Lean Noninsulin-Dependent Diabetic Subjects,” Journal of Clinical
Endocrinology & Metabolism 73, no. 6 (December 1991): 1294–301,
https://www.ncbi.nlm.nih.gov/pubmed/1955512.
18. A. Martin-Montalvo, E. M. Mercken, S. J. Mitchel , et al., “Metformin Improves Healthspan and
Lifespan
in
Mice,”
Nature
Communications
4
(2013):
2192,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3736576/.
19. V. N. Anisimov, “Metformin for Aging and Cancer Prevention,” Aging 2, no. 11 (November 2010): 760–74.
20. S. Andrzejewski, S.-P. Gravel, M. Pol ak, and J. St-Pierre, “Metformin Directly Acts on Mitochondria to Alter Cel ular Bioenergetics,” Cancer & Metabolism 2 (August 28, 2014):
12, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4147388/.
21. N. Barzilai, J. P. Crandal , S. P. Kritchevsky, and M. A. Espeland, “Metformin as a Tool to
Target Aging,” Cell Metabolism 23 (June 14, 2016): 1060–65, https://www.cel .com/cel -
metabolism/pdf/S1550-4131(16)30229-7.pdf.
22. C.-P. Wang, C. Lorenzo, S. L. Habib, et al. “Di erential E ects of Metformin on Age Related Comorbidities in Older Men with Type 2 Diabetes,” Journal of Diabetes and Its
Complications
31,
no.
4
(2017):
679–86,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5654524/.
23. J. M. Campbel , S. M. Bel man, M. D. Stephenson, and K. Lisy, “Metformin Reduces Al -Cause Mortality and Diseases of Ageing Independent of Its E ect on Diabetes Control: A
Systematic Review and Meta-analysis,” Ageing Research Reviews 40 (November 2017): 31–
44, https://www.ncbi.nlm.nih.gov/pubmed/28802803.
24. N. Howlader, A. M. Noone, M. Krapcho, et al., “SEER Cancer Statistics Review, 1975–
2009,”
National
Cancer
Institute,
August
20,
2012,
https://seer.cancer.gov/archive/csr/1975_2009_pops09/.
25. By the time you hit 90, the authors found, there’s a threefold decrease in the probability of developing cancer. If you make it to 100, from there the probability is minimal, 0 to 4
percent. N. Pavlidis, G. Stanta, and R. A. Audisio, “Cancer Prevalence and Mortality in
Centenarians: A Systematic Review,” Critical Reviews in Oncology/Hematology 83, no. 1
(July 2012): 145–52, https://www.ncbi.nlm.nih.gov/pubmed/22024388.
26. I. Elbere, I. Silamikelis, M. Ustinova, et al., “Signi cantly Altered Peripheral Blood Cel DNA Methylation Pro le as a Result of Immediate E ect of Metformin Use in Healthy
Individuals,” Clinical Epigenetics 10, no. 1 (2018), https://doi.org/10.1186/s13148-018-
27. B. K. Kennedy, M. Gotta, D. A. Sinclair, et al., “Redistribution of Silencing Proteins from Telomeres to the Nucleolus Is Associated with Extension of Lifespan in S. cerevisiae,” Cell
89, no. 3 (May 2, 1997): 381–91, https://www.ncbi.nlm.nih.gov/pubmed/?term=SIR4-
42+sinclair+gotta; D. A. Sinclair and L. Guarente, “Extrachromosomal rDNA Circles—A
Cause of Aging in Yeast,” Cell 91, no. 7 (December 26, 1997): 1033–42,
https://www.ncbi.nlm.nih.gov/pubmed/9428525; D. Sinclair, K. Mil s, and L. Guarente,
“Accelerated Aging and Nucleolar Fragmentation in Yeast SGS1 Mutants,” Science 277, no.
5330 (August 29, 1997): 1313–16, https://www.ncbi.nlm.nih.gov/pubmed/9271578.
28. The research into resveratrol suggests that it is promising for both cancer and cardiovascular disease prevention. Resveratrol’s ability to act on tumor growth points to other possibilities.
“Since tumor promoting agents alter the expression of genes whose products are associated
with in ammation, chemoprevention of cardiovascular diseases and cancer may share the
same common mechanisms.” E. Ignatowicz and W. Baer-Dubowska, “Resveratrol, a Natural
Chemopreventive Agent Against Degenerative Diseases, “Polish Journal of Pharmacology 53,
no. 6 (November 2001): 557–69, https://www.ncbi.nlm.nih.gov/pubmed/11985329.
29. The title of our paper is a combination of two Greek words: “xenos, the Greek word for stranger, and hormesis, the term for health bene ts provided by mild biological stress, such as
cel ular damage or a lack of nutrition.” K. T. Howitz and D. A. Sinclair, “Xenohormesis:
Sensing the Chemical Cues of Other Species,” Cell 133, no. 3 (May 2, 2008): 387–91,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2504011/.
30. An average glass of red wine contains about 1 to 3 mg of resveratrol. There is no resveratrol in white wine because resveratrol is produced largely by the skins of the grape, which are not
used in white wine production. For more information on and dietary sources of resveratrol,
see J. A. Baur and D. A. Sinclair, “Therapeutic Potential of Resveratrol: The in Vivo Evidence,” Nature Reviews Drug Discovery 5, no. 6 (June 2006): 493–506,https://www.ncbi.nlm.nih.gov/pubmed/16732220.
31. Continuing on from our work, the researchers proposed “a novel pathway by which products of the plant stress response confer stress tolerance and extend longevity in animals.”
They also highlighted how xenohormesis may boost the health-giving and medicinal
properties of plants, while also tackling issues around adapting in a world that’s forever
changing. P. L. Hooper, P. L. Hooper, M. Tytel , and L. Vigh, “Xenohormesis: Health
Bene ts from an Eon of Plant Stress Response Evolution,” Cell Stress & Chaperones 15, no. 6
(November 2010): 761–70, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3024065/.
32. The implications for overweight humans were clear, we found. “This study shows that an oral y available smal molecule at doses achievable in humans can safely reduce many of the
negative consequences of excess caloric intake, with an overal improvement in health and
survival.” J. A. Baur, K. J. Pearson, N. L. Price, et al., “Resveratrol Improves Health and
Survival of Mice on a High-Calorie Diet,” Nature 444, no. 7117 (November 1, 2006):
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4990206/.
33. J. A. Baur and D. A. Sinclair, “Therapeutic Potential of Resveratrol: The In Vivo Evidence,”
Nature
Reviews
Drug
Discovery
5,
(2006):
493–506,
https://www.nature.com/articles/nrd2060.
34. K. J. Pearson, J. A. Baur, K. N. Lewis, et al., “Resveratrol Delays Age-Related Deterioration and Mimics Transcriptional Aspects of Dietary Restriction Without Extending Life Span,”
Cell Metabolism 8, no. 2 (August 6, 2008): 157–68, https://www.cel .com/cel -
metabolism/abstract/S1550-4131%2808%2900182-4.
35. Our ndings inevitably stirred up excitement in the media that drinking red wine may increase longevity, as wel as admittedly a more sedate example, such as the article “Life-Extending Chemical Is Found in Some Red Wines” in the New York Times. K. T. Howitz, K.
J. Bitterman, H. Y. Cohen, et al., “Smal Molecule Activators of Sirtuins Extend
Saccharomyces cerevisiae Lifespan,” Nature 425, no. 6954 (September 11, 2003): 191–96,
https://www.ncbi.nlm.nih.gov/pubmed/12939617.
36. To combat aging in mice, we fed them the equivalent of about 100 glasses of red wine a day, not “1,000,” neither of which I recommend.
37. Martin-Montalvo et al., “Metformin Improves Healthspan and Lifespan in Mice.”
38. Forty patients with varying degrees of psoriasis took part in the study, of whom just over a third had “good to excel ent” improvement, according to skin biopsies. J. G. Kreuger, M.
Suárez-Fariñas, I. Cueto, et al., “A Randomized, Placebo-Control ed Study of SRT2104, a
SIRT1 Activator, in Patients with Moderate to Severe Psoriasis,” PLOS One, November 10,
2015, https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0142081.
39. Hydrogen is used for hundreds of so-cal ed redox reactions in the cel . NAD is a “hydrogen carrier.” The plus sign on “NAD+” indicates the form of NAD that doesn’t have a hydrogen
atom attached. When it has a hydrogen atom attached, it is cal ed “NADH.”40. As NAD levels decline with age, so the body becomes more susceptible to disease, as two col aborators and I noted: “Restoration of NAD+ levels in old or diseased animals can
promote health and extend lifespan, prompting a search for safe and e cacious NAD-
boosting molecules that hold the promise of increasing the body’s resilience, not just to one
disease, but to many, thereby extending healthy human lifespan.” L. Rajman, K. Chwalek,
and D. A. Sinclair, “Therapeutic Potential of NAD-Boosting Molecules: The in Vivo
Evidence,” Cell Metabolism 27, no. 3 (March 6, 2018): 529–47,
https://www.ncbi.nlm.nih.gov/pubmed/29514064.
41. Y. A. R. White, D. C. Woods, Y. Takai, et al., “Oocyte Formation by Mitotical y Active Germ Cel s Puri ed from Ovaries of Reproductive Age Women,” Nature Medicine 18 (February
26, 2012): 413–21, https://www.nature.com/articles/nm.2669.
42. J. L. Til y and D. A. Sinclair, “Germline Energetics, Aging, and Female Infertility,” Cell Metabolism
17,
no.
6
(June
2013):
838–50,
https://www.sciencedirect.com/science/article/pi /S1550413113001976.
43. Our paper in which we showed that SIRT2 is a key player in regulating lifespan in a living organism came out in 2014. B. J. North, M. A. Rosenberg, K. B. Jeganathan, et al., “SIRT2
Induces the Checkpoint Kinase BubR1 to Increase Lifespan,” EMBO Journal 33, no. 13
(July 1, 2014): 1438–53, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4194088/.
44. The researchers frame their results within the epidemic of obesity in developing countries and its link to reproductive health issues, including not only polycystic brosis but also
gestational diabetes mel itus and endometrial cancer. They conclude that “Metformin may
be a valuable alternative to, or adjunct for, modifying the toxic e ects of obesity in these
populations.” V. N. Sivalingam, J. Myers, S. Nicholas, et al., “Metformin in Reproductive
Health, Pregnancy and Gynaecological Cancer: Established and Emerging Indications,”
Human
Reproduction
20,
no.
6
(November
2014):
853–68,
https://academic.oup.com/humupd/article/20/6/853/2952671.
45. “Chemotherapy-treated animals had signi cantly fewer o spring compared with al other treatment groups, whereas cotreatment with mTOR inhibitors preserved normal fertility.”
K. N. Goldman, D. Chenette, R. Arju, et al., “mTORC1/2 Inhibition Preserves Ovarian
Function and Fertility During Genotoxic Chemotherapy,” Proceedings of the National
Academy of Sciences of the United States of America 114, no. 2 (March 21, 2017): 3196–91,
http://www.pnas.org/content/114/12/3186.ful .
46. Mice de cient in mTORC1, the authors found, “present spermatozoa with decreased motility, suggesting that mTORC1, besides control ing glandular size and seminal vesicle
uid composition, also regulates sperm physiology during passage through the epididymis.”
P. F. Oliveira, C. Y. Cheng, and M. G. Alves, “Emerging Role for Mammalian Target of
Rapamycin in Male Fertility,” Trends in Endocrinology and Metabolism 28, no. 3 (March
2017): 165–67, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5499664/.
47. The term “aging in place” refers to a recently evolved philosophy in Western countries of encouraging the elderly to grow old in places that meet their needs and circumstances.Australia, like so many other countries, is facing a demographic explosion in the number of
its elderly, which has signi cant budgetary and societal implications. Australia’s 65- to 84-
year-old population is expected to double or more by 2050. H. Bartlett and M. Carrol ,
“Aging in Place Down Under,” Global Ageing: Issues & Action 7, no. 2 (2011): 25–34,
https://www.ifa- v.org/wp-content/uploads/global-ageing/7.2/7.2.bartlett.carrol .pdf.
SIX. BIG STEPS AHEAD
1. In a wide-ranging survey of interventions, the authors covered the health and life-prolonging bene ts of various small molecules, exercising, and fasting regimes. “The current epidemics
of obesity, diabetes, and related disorders constitute major impediments for healthy aging,”
they wrote. “It is only by extending the healthy human lifespan that we wil truly meet the
premise of the Roman poet Cicero: ‘No one is so old as to think that he may not live a year.’ ”
R. de Cabo, D. Carmona-Guttierez, M. Bernier, et al., “The Search for Antiaging
Interventions: From Elixirs to Fasting Regimens,” Cell 157, no. 7 (June 19, 2014): 1515–26,
https://www.cel .com/ful text/S0092-8674(14)00679-5.
2. J. Yost and J. E. Gudjonsson, “The Role of TNF Inhibitors in Psoriasis Therapy: New Implications for Associated Comorbidities,” F1000 Medicine Reports 1, no. 30 (May 8,
2009), https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2924720/.
3. Kil ing o senescent cel s in mice led to their having healthier lives, the author wrote in a story for Nature on Baker and van Deursen’s work. Their kidney function improved, and
their hearts were more resilient to stress, they tended to explore their cages more, and
developed cancers at a later age. E. Cal away, “Destroying Worn-out Cel s Makes Mice Live
Longer,” Nature, February 3, 2016, https://www.nature.com/news/destroying-worn-out-
cel s-makes-mice-live-longer-1.19287.
4. The impact of injected senescent cel s on young mice was also remarkable in their destructiveness. “As early as two weeks after transplantation, the SEN mice showed impaired
physical function as determined by maximum walking speed, muscle strength, physical
endurance, daily activity, food intake, and body weight,” according to the NIH press release.
“In addition, the researchers saw increased numbers of senescent cel s, beyond what was
injected, suggesting a propagation of the senescence e ect into neighboring cel s.” “Senolytic
Drugs Reverse Damage Caused by Senescent Cel s in Mice,” National Institutes of Health,
July 9, 2018, https://www.nih.gov/news-events/news-releases/senolytic-drugs-reverse-
damage-caused-senescent-cel s-mice.
5. R.-M. Laberge, Y. Sun, A. V. Orjalo, et al., “MTOR Regulates the Pro-tumorigenic Senescence-Associated Secretory Phenotype by Promoting IL1A Translation,” Nature Cell
Biology17,
no.
8
(July
6,
2015):
1049–61,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4691706/.
6. P. Oberdoer er, S. Michan, M. McVay, et al., “DNA Damage–Induced Alterations in Chromatin Contribute to Genomic Integrity and Age-Related Changes in Gene
Expression,”
Cell
135,
no.
5
(November
28,
2008):
907–18,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2853975/.
7. M. De Cecco, S. W. Criscione, E. J. Peckham, et al., “Genomes of Replicatively Senescent Cel s Undergo Global Epigenetic Changes Leading to Gene Silencing and Activation of
Transposable Elements,” Aging Cell 12, no. 2 (April 2013): 247–56,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3618682/.
8. “Adoptive transfer of T cel s isolated from vaccine-treated tumor-bearing mice inhibited tumor growth in unvaccinated recipients, indicating that the iPSC vaccine promotes an
antigen-speci c anti-tumor T cel response,” the researchers found. N. G. Kooreman, K.
Youngkyun, P. E. de Almeida, et al., “Autologous iPSC-Based Vaccines Elicit Anti-tumor
Responses in Vivo,” Cell Stem Cell 22, no. 4 (April 5, 2018), http://www.cel .com/cel -stem-
cel /ful text/S1934-5909(18)30016-X.
9. Cel s were taken from inside Streisand’s dog’s cheek and bel y skin and sent to a lab in Texas.
The cloning process resulted in four puppies, although one died shortly after birth. Streisand
wrote that the dogs’ resembling her beloved Samantha physical y was enough. “You can
clone the look of a dog, but you can’t clone the soul. Stil , every time I look at their faces, I
think of my Samantha . . . and smile.” B. Streisand, “Barbara Streisand Explains: Why I
Cloned
My
Dog,”
New
York
Times,
March
2,
2018,
https://www.nytimes.com/2018/03/02/style/barbra-streisand-cloned-her-dog.html.
10. It is one of the most interesting and important papers I’ve ever read. C. E. Shannon, “A Mathematical Theory of Communication,” Bell System Technical Journal 27, no. 3 (July
1948):
379–423
and
no.
4
(October
1948):
623–66,
http://math.harvard.edu/~ctm/home/text/others/shannon/entropy/entropy.pdf.
11. The results of their experiments were extremely promising when it came to slowing down aging by putting a halt to the molecular changes that cause it. “Molecular alterations induced
by in vivo reprogramming may potential y lead to a better maintenance of tissue homeostasis
and lifespan extension,” they wrote. A. Ocampo, P. Reddy, P. Martinez-Redondo, et al., “In
Vivo Amelioration of Age-Associated Hal marks by Partial Reprogramming,” Cell 167, no. 7
(December 15, 2016): 1719–33, https://www.cel .com/cel /pdf/S0092-8674(16)31664-
12. “I feel a strong responsibility that it’s not just to make a rst, but also make it an example,” he told the Associated Press. “Society wil decide what to do next” as to whether such
experiments should continue or be banned. M. Marchione, “Chinese Researcher Claims
First
Gene-Edited
Babies,”
Associated
Press,
November
26,
2018,
https://www.apnews.com/4997bb7aa36c45449b488e19ac83e86d.
SEVEN. THE AGE OF INNOVATION1. H. Singh, , A.N.D. Meyer, and E. J. Thomas, “The Frequency of Diagnostic Errors in Outpatient Care: Estimations from Three Large Observational Studies Involving US Adult
Populations,” BMJ Quality & Safety 23, no. 9 (August 12, 2014),
https://qualitysafety.bmj.com/content/23/9/727.
2. M. Jain, S. Koren, K. H. Miga, et al., “Nanopore Sequencing and Assembly of a Human
Genome with Ultra-long Reads,” Nature Biotechnology 36, no. 4 (2018): 338–45,
https://www.nature.com/articles/nbt.4060.
3. The evolution of such technology is tied by its inventors to bene ting the community, rather than corporations. That said, this particular company was also promoting the idea of a
“coin,” or digital currency, not for investment or as a security, according to the writer, but to
incentivize individuals to share their genomic data with scientists. “The underlying idea is to
incentivize users to make their personal genomic data available for biomedical and health-
related research for the greater good of medical discovery.” B. V. Bigelow, “Luna DNA Uses
Blockchain to Share Genomic Data as a ‘Public Bene t,’ ” Exome, January 22, 2018,
https://xconomy.com/san-diego/2018/01/22/luna-dna-uses-blockchain-to-share-genomic-
4. S. W. H. Lee, N. Chaiyakunapruk, and N. M. Lai, “What G6PD-De cient Individuals Should Real y Avoid,” British Journal of Clinical Pharmacology 83, no. 1 (January 2017):
211–12,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5338146/;
“Glucose-6-
Phosphate
Dehydrogenase
De ciency,”
MedlinePlus,
https://medlineplus.gov/ency/article/000528.htm.
5. J. A. Sparano, R. J. Gray, D. F. Makower, et al., “Adjuvant Chemotherapy Guided by a 21-Gene Expression Assay in Breast Cancer,” New England Journal of Medicine 379 (July 12,
2018): 111–21, https://www.nejm.org/doi/ful /10.1056/NEJMoa1804710.
6. K. A. Liu and N. A. D. Mager, “Women’s Involvement in Clinical Trials: Historical Perspective and Future Implications,” Pharmacy Practice 14, no. 1 (January–March 2016):
708–17, https://www.pharmacypractice.org/journal/index.php/pp/article/view/708/424.
7. Female mice that had received mTOR treatment lived 20 percent longer than the untreated mice in the control group. Leibniz Institute on Aging, Fritz Lipmann Institute, “Less Is
More? Gene Switch for Healthy Aging Found,” Medical Xpress, May 25, 2018,
https://medicalxpress.com/news/2018-05-gene-healthy-aging.html.
8. Swedish records showed that in every single year since 1800, women had lived longer than men. “This remarkably consistent survival advantage of women compared with men in early
life, in late life, and in total life is not con ned to Sweden but is seen in every country in every
year for which reliable birth and death records exist. There may be no more robust pattern in
human biology,” the authors noted. S. N. Austad and A. Bartke, “Sex Di erences in
Longevity and in Responses to Anti-aging Interventions: A Mini-review,” Gerontology 62, no. 2 (2015): 40–46, https://www.karger.com/Article/Ful Text/381472.9. E. J. Davis, I. Lobach, and D. B. Dubal, “Female XX Sex Chromosomes Increase Survival and Extend Lifespan in Aging Mice,” Aging Cell 18, no. 1 (February 2019), e12871,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6351820/.
10. One example where pharmacogenomics information is already used to inform drug prescription is in the treatment of HIV. Patients with HIV are tested for a speci c genetic
variant to see if they may have a bad reaction to an antiviral drug cal ed abacavir, according to
a fact sheet on the National Human Genome Research Institute’s website; see “Frequently
Asked Questions About Pharmacogenomics,” National Human Genome Research
Institute, May 2, 2016, https://www.genome.gov/27530645/.
11. The autopsy of a mummi ed corpse of a fourteenth-century Italian warlord lent credence to centuries-old rumors that days after his triumphant conquest of Treviso, 38-year-old
Cangrande I del a Scala had been poisoned with digitalis. H. Thompson, “Poison Hath Been
This Italian Mummy’s Untimely End,” Smithsonian.com, January 14, 2015,
https://www.smithsonianmag.com/science-nature/poison-hath-been-italian-mummys-
untimely-end-digitalis-foxglove-180953822/.
12. M. Vamos, J. W. Erath, and S. H. Hohnloser, “Digoxin-Associated Mortality: A Systematic Review and Meta-analysis of the Literature,” European Heart Journal 36, no. 28 (July 21,
2015): 1831–38, https://academic.oup.com/eurheartj/article/36/28/1831/2398087.
13. M. N. Miemeijer, M. E. van den Berg, J. W. Deckers, et al., “ABCB1 Gene Variants, Digoxin and Risk of Sudden Cardiac Death in a General Population,” BMJ Heart 101, no. 24
(December 2015), https://heart.bmj.com/content/101/24/1973?heartjnl-2014-307419v1=;
A. Oni-Orisan and D. Lanfear, “Pharmacogenomics in Heart Failure: Where Are We Now
and How Can We Reach Clinical Application?,” Cardiology in Review 22, no. 5 (September
1, 2015): 193–98, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4329642/.
14. Back in 2015, Johnson felt it would be only another ten years until our genome is de ned and stored for use while we’re alive. “When that happens, using genetic information to
inform decisions about the right drug and the right dose wil likely involve computerized
approaches that marry the genetic data with knowledge about drugs and genes, to lead to a
personalized treatment recommendation,” she wrote. J. A. Johnson, “How Your Genes
In uence What Medicines Are Right for You,” Conversation, November 20, 2015,
https://theconversation.com/how-your-genes-in uence-what-medicines-are-right-for-you-
15. That seems to be changing, though, according to the authors, as more of their col eagues publish papers on this area, ensuring “that the gut microbiota are moving out of the shadows
and are moving towards centre stage in drug safety studies and personalized health care.” I.
D. Wilson and J. K. Nicholson, “Gut Microbiome Interactions with Drug Metabolism,
E cacy and Toxicity,” Translational Research: The Journal of Laboratory and Clinical
Medicine179
(January
2017):
204–22,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5718288/; see also B. Das, T. S. Ghosh, S.
Kedia, et al., “Analysis of the Gut Microbiome of Rural and Urban Healthy Indians Living
in Sea Level and High-Altitude Areas,” Nature Scientific Reports 8 (July 4, 2018),
https://www.nature.com/articles/s41598-018-28550-3.
16. P. Lehouritis, J. Cummins, M. Stanton, et al., “Local Bacteria A ect the E cacy of Chemotherapeutic Drugs,” Nature Scientific Reports 5 (September 29, 2015),
https://www.nature.com/articles/srep14554.
17. The wait increased from 18.5 days in 2014 to 24 days in 2017, according to a study by MerrittHawkins. B. Japsen, “Doctor Wait Times Soar 30% in Major U.S. Cities,” Forbes,
March 19, 2017, https://www.forbes.com/sites/brucejapsen/2017/03/19/doctor-wait-
times-soar-amid-trumpcare-debate/#7ac0753b2e74.
18. The website of myDNAge o ers some encouragement: “You can’t change your genes, but
you can change how your genes behave through epigenetics,” runs the tagline. Al you have
to do is send in your bodily uids (blood or urine) and they’l determine your biological age
by measuring the epigenetic modi cations on your DNA. “Reveal Your Biological Age
Through Epigenetics,” myDNAge, 2017, https://www.mydnage.com/. TeloYears o ers to track your cel ular age based on your telomeres, which, it informs its website readers, are “the
caps on your DNA that, unlike your ancestry, you can actual y change.” TeloYears, 2018,
https://www.teloyears.com/home/.
19. M. W. Snyder, M. Kircher, A. J. Hil , et al., “Cel -free DNA Comprises an in Vivo Nucleosome Footprint That Informs Its Tissues-of-Origin,” Cell 164, nos. 1–2 (January 14,
2016): 57–68, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4715266/.
20. “Global Automotive Level Sensor Market Analysis, Trends, Drivers, Chal enges & Forecasts 2018–2022, with the Market Set to Grow at a CAGR of 4.13%—
Business
Wire,
May
2,
2018,
https://www.businesswire.com/news/home/20180502005988/en/Global-Automotive-
Level-Sensor-Market-Analysis-Trends.
21. University of Cincinnati senior scientist Jason Heikenfeld and his team worked with a US
Air Force Research Laboratory in Ohio on a simple way to keep track of how airmen
respond to everything from diet, stress, and injury to medication and disease. They came up
with patches that both stimulate and monitor sweat and then send data to a smartphone. J.
Heikenfeld, “Sweat Sensors Wil Change How Wearables Track Your Health,” IEEE
Spectrum, October 22, 2014, https://spectrum.ieee.org/biomedical/diagnostics/sweat-
sensors-wil -change-how-wearables-track-your-health.
22. Owlstone has already started lung cancer clinical trials in the United Kingdom, testing hundreds of patients for early signs. In the United Kingdom, it notes on its website, “only
14.5 percent of people are diagnosed with early stage, treatable lung cancer. If we are able to
increase this to 25% we’d save 10,000 lives in the U.K. alone.” D. Sfera, “Breath Test Detects
CancerMarkers,”
Medium,
August
2,
2018,
https://medium.com/@TheRealDanSfera/breath-test-detects-cancer-markers-
c57dcc86a583. With advancements in drug treatments, early detection, the company points out, is a more powerful tool to save lives than the development of new drugs. “A Breathalyzer
for Disease,” Owlstone Medical, https://www.owlstonemedical.com/.
23. Two examples are Öura Ring (https://ouraring.com/) and Motiv Ring (https://mymotiv.com/).
24. “A growing body of evidence suggests that an array of mental and physical conditions can make you slur your words, elongate sounds, or speak in a more nasal tone.” R. Robbins,
“The Sound of Your Voice May Diagnose Disease,” Scientific American, June 30, 2016,
https://www.scienti camerican.com/article/the-sound-of-your-voice-may-diagnose-disease/.
25. Researchers used the amount of time subjects took to press and release a key on the computer and converted it to a Parkinson’s disease motor index. L. Giancardo, A. Sánchez-Ferro, T. Arroyo-Gal ego, et al., “Computer Keyboard Interaction as an Indicator of Early
Parkinson’s Disease,” Nature Scientific Reports 6 (October 5, 2016): 34468,
https://www.nature.com/articles/srep34468.
26. For a more detailed account of what’s just around the corner, this book is wel worth the read: E. Topol, The Creative Destruction of Medicine: How the Digital Revolution Will
Create Better Health Care, Kindle edition (New York: Basic Books, 2011).
27. I am an investor in and former member of the board of InsideTracker, a Segterra company based in Massachusetts, http://www.insidetracker.com/. I have invested in and advise the company, and I am an inventor on a patent application led to calculate biological age based
on markers that are known to change with age.
28. The app is cal ed Clue. E. Avey, “ ‘The Clue App Saved My Life’: Early Detection Through
Cycle Tracking,” Clued In, September 24, 2017, https://medium.com/clued-in/the-clue-
app-saved-my-life-early-detection-through-cycle-tracking-91732dd29d25.
29. Over the past three decades, a new infectious disease has appeared every single year in some part of the world. In total, researchers put the number of unknown viruses in birds and
mammals that could infect humans between 631,000 and 827,000. Though there are
ongoing e orts to identify al these viruses, “we likely won’t ever be able to predict which
wil spil over next; even long-known viruses like Zika, which was discovered in 1947, can
suddenly develop into unforeseen epidemics.” E. Yong, “The Next Plague Is Coming. Is
America
Ready?,”
The
Atlantic,
July–August
2018,
https://www.theatlantic.com/magazine/archive/2018/07/when-the-next-plague-
30. L. M. Mobula, M. MacDermott, C. Hoggart, et al., “Clinical Manifestations and Modes of Death Among Patients with Ebola Virus Disease, Monrovia, Liberia, 2014,” American
Journal of Tropical Medicine and Hygiene 98, no. 4 (April 2018): 1186–93,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5928808/.
31. The measures to prepare for a future pandemic that Gates argues in an editorial should be put into play include building up public health systems in countries vulnerable to epidemicsand mimicking how the military preps for war with “germ games and other preparedness
exercises so we can better understand how diseases wil spread, how people wil respond in a
panic, and how to deal with things like overloaded highways and communications systems.”
B. Gates, “Bil Gates: A New Kind of Terrorism Could Wipe Out 30 Mil ion People in Less
than a Year—and We Are Not Prepared,” Business Insider, February 18, 2017,
http://www.businessinsider.com/bil -gates-op-ed-bio-terrorism-epidemic-world-threat-
32. It was only after a 2009 law was passed that companies had to inform the public and the government of any breaches. Since then, the volume of breaches at health care providers has
consistently climbed, from 150 in 2010 to 250 seven years later. Consumer Reports,
“Hackers Want Your Medical Records. Here’s How to Keep Your Info from Them,”
Washington Post, December 17, 2018, https://www.washingtonpost.com/national/health-
science/hackers-want-your-medical-records-heres-how-to-keep-your-info-from-
them/2018/12/14/4a9c9ab4-fc9c-11e8-ad40-cdfd0e0dd65a_story.html?
33. A. Sul eyman, “NHS Cyber Attack: Why Stolen Medical Information Is So Much More Valuable
than
Financial
Data,”
Independent,
May
12,
2017,
https://www.independent.co.uk/life-style/gadgets-and-tech/news/nhs-cyber-attack-medical-
data-records-stolen-why-so-valuable-to-sel - nancial-a7733171.html.
34. S. S. Dominy, C. Lynch, F. Ermini, et al., “Porphyromonas gingivalis in Alzheimer’s Disease Brains: Evidence for Disease Causation and Treatment with Smal -Molecule Inhibitors,”
Science
Advances
5,
no.
1
(January
23,
2019),
http://advances.sciencemag.org/content/advances/5/1/eaau3333.ful .pdf.
35. That rate of decline continued over the next few years, thanks to fewer older adults requiring hospitalization for pneumonia. “By 2009, more than half the nationwide decline in
pneumonia hospitalizations could be attributed to older adults, with some 70,000 fewer
annual hospitalizations for those age 85 and older.” “Infant Vaccine for Pneumonia Helps
Protect
Elderly,”
VUMC
Reporter,
July
11,
2013,
http://news.vumc.org/2013/07/11/infant-vaccine-for-pneumonia-helps-protect-elderly/.
36. M. R. Moore, R. Link-Gel es, W. Scha ner, et al., “Impact of 13-Valent Pneumococcal Conjugate Vaccine Used in Children on Invasive Pneumococcal Disease in Children and
Adults in the United States: Analysis of Multisite, Population-Based Surveil ance,” Lancet
Infectious
Diseases
15,
no.
3
(March
2015):
301–09,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4876855/.
37. If you have a companion animal, it can stil get the Lyme disease vaccine.
38. “ ‘The [research and development] model is broken,’ said Kate Elder, vaccines policy adviser at Médecins sans Frontières. ‘Priorities are chosen based on where the money is . . . diseases
predominantly in the developed world,’ she said.” H. Col is, “Vaccines Need a New BusinessModel,” Politico, April 27, 2016, https://www.politico.eu/article/special-report-vaccines-
39. “The analysis was conducted by Ronald Evens, adjunct research professor at Tufts CSDD
and Tufts University School of Medicine and adjunct professor in the Thomas J. Long
School of Pharmacy and Health Sciences at University of the Paci c, using data from
company reports, periodic biotechnology reports from the Pharmaceutical Research and
Manufacturers of America, IMS sales data, and FDA and Tufts CSDD databases.” M.
Powers, “Tufts: The Vaccine Pipeline Is Soaring and Global Sales Could Hit $40B by 2020,”
BioWorld, April 21, 2016, http://www.bioworld.com/content/tufts-vaccine-pipeline-
soaring-and-global-sales-could-hit-40b-2020.
40. Africa has borne the brunt of more than 90 percent of the globe’s malaria cases and deaths.
“Malaria,” World Health Organization, November 19, 2018, https://www.who.int/news-
room/fact-sheets/detail/malaria.
41. “Ghana, Kenya and Malawi to Take Part in WHO Malaria Vaccine Pilot Programme,” World Health
Organization,
Regional
O ce
for
Africa,
April
24,
2017,
http://www.afro.who.int/news/ghana-kenya-and-malawi-take-part-who-malaria-vaccine-
42. Crises such as an Ebola outbreak highlight a fundamental aw in medical research and drug development, researchers told a Boston Globe reporter. Unless there’s public concern,
researchers and pharmaceutical companies have “scant incentive to quickly develop vaccines
and drugs for little-seen diseases.” Y. Abutaleb, “Speeding Up the Fight Against Ebola, Other
Diseases,”
Boston
Globe,
August
22,
2014,
https://www.bostonglobe.com/metro/2014/08/21/faster-development-vaccines-and-drugs-
targeting-diseases-such-ebola-horizon/yrkrN56VgehrSzCtETPzzH/story.html.
43. An equal y sobering statistic is that each day twenty people die waiting for a transplant, while just one organ donor can save eight lives. “Transplant Trends,” United Network for Organ
Sharing, https://unos.org/data/.
44. That said, Crouch notes, in Cruise’s most recent Mission Impossible epic, “Mission: Impossible—Fallout,” the 56-year-old Cruise’s character, Ethan Hunt, appears to
acknowledge that with advancing years come limitations, as in, for example, needing a
younger associate to help him defeat a bad guy in a lengthy brawl or having an eye for ever-
younger girlfriends. I. Crouch, “The Wilford Brimley Meme That Helps Measure Tom
Cruise’s Agelessness,” “Rabbit Holes,” New Yorker, August 11, 2018,
https://www.newyorker.com/culture/rabbit-holes/the-wilford-brimley-meme-that-helps-
measure-tom-cruises-agelessness.
EIGHT. THE SHAPE OF THINGS TO COME
1. A. Jenkins, “Which 19th century physicist famously said that al that remained to be done in physics was compute e ects to another decimal place?,” Quora, June 26, 2016,https://www.quora.com/Which-19th-century-physicist-famously-said-that-al -that-
remained-to-be-done-in-physics-was-compute-e ects-to-another-decimal-place.
“The
Road
Ahead
(Bil
Gates
book),”
Wikipedia,
https://en.wikipedia.org/wiki/The_Road_Ahead_(Bil _Gates_book)#cite_note-Weiss06-3.
3. Kel y added a key point to that excel ent mantra: “It’s by use [that] we gure out what things are good for. Which is perhaps another way of saying “Go with the ow and see where it
takes you.” J. Altucher, “One Rule for Predicting What You Never Saw Coming . . . ,” The
Mission, July 15, 2016, https://medium.com/the-mission/kevin-kel y-one-rule-for-
predicting-what-you-never-saw-coming-1e9e4eeae1da.
4. L. Gratton and A. Scott, The 100 Year Life: Living and Working in an Age of Longevity (London and New York: Bloomsbury Publishing, 2018).
5. A phrase originating with the theologian Theodore Parker but made famous by Dr. Martin Luther King, Jr., and used several times by President Barack Obama.
6. It was a time of su cient population density that people began to take an interest in their appearance, notably changing how they looked with beads and pigments. E. Trinkaus, “Late
Pleistocene Adult Mortality Patterns and Modern Human Establishment,” Proceedings of the
National Academy of Sciences of the United States of America 108, no. 4 (January 25, 2011):
12267–71, https://www.ncbi.nlm.nih.gov/pubmed/21220336.
7. Up until 4,000 years ago, human numbers were minimal, according to a writer at the Global Environmental Alert Service. Since then, growth has climbed ever faster, the rate peaking in
the 1960s. In 2012, the United Nations estimated that by the end of the century world
population wil be 10.1 bil ion. “One Planet, How Many People? A Review of Earth’s
Carrying Capacity,” UNEP Global Environmental Alert Service, June 2012,
https://na.unep.net/geas/archive/pdfs/geas_jun_12_carrying_capacity.pdf.
8. Similar sentiments are held by the American public, according to a Pew Research Center survey, which found that 59 percent took a “pessimistic view about the e ect of population
growth saying it wil be a major problem because there wil not be enough food and
resources to go around.” “Attitudes and Beliefs on Science and Technology Topics,” Pew
Research
Center,
Science
&
Society,
January
29,
2015,
http://www.pewinternet.org/2015/01/29/chapter-3-attitudes-and-beliefs-on-science-and-
technology-topics/#population-growth-and-natural-resources-23-point-gap.
9. M. Blythe, “Professor Frank Fenner, Microbiologist and Virologist,” Australian Academy of
https://www.science.org.au/learning/general-
audience/history/interviews-australian-scientists/professor-frank-fenner.
10. Fenner contrasted the fate of humanity with that of the residents of Easter Island, who were decimated in the 1600s by their reliance on the forests they themselves had cut down.
Dwindling food sources, fol owed by civil war and the arrival of foreign sailors who brought
violence and disease, made its population plunge to 111 individuals by 1872. Though thenumbers have since rebounded, Fenner’s views on humanity’s future did not hold up such a
generous possibility, he told a reporter from the Australian. “As the population keeps
growing to seven, eight or nine bil ion, there wil be a lot more wars over food,” he said. “The
grandchildren of today’s generations wil face a much more di cult world.” C. Jones,
“Frank Fenner Sees No Hope for Humans,” Australian, June 16, 2010,
https://www.theaustralian.com.au/higher-education/frank-fenner-sees-no-hope-for-
humans/news-story/8d77f0806a8a3591d47013f7d75699b9?
nk=099645834c69c221f8ecf836d72b8e4b-1520269044.
11. “There are 925 mil ion people who go hungry every day, despite the amazing economic prosperity we’ve enjoyed over the past 60 years,” wrote Michael Schuman in a piece on
Malthus’s predictions for Time. “And twice in the past three years we’ve su ered through
destabilizing spikes in the cost of food that have trapped tens of mil ions in poverty. Today,
prices are nearly at historic highs.” M. Schuman, “Was Malthus Right?,” Time, July 15,
2011, http://business.time.com/2011/07/15/was-malthus-right/.
12. P. R. Ehrlich, The Population Bomb (New York: Bal antine Books, 1968), 1.
13. Ibid., 3.
14. Some of the statistics are simply mind-boggling. Not only is our global population growing by 74 mil ion a year, but “we have consumed more resources in the last 50 years than the
whole of humanity before us.” S. Dovers, “Population and Environment: A Global
Chal enge,”
Australian
Academy
of
Science,
August
7,
2015,
https://www.science.org.au/curious/earth-environment/population-environment.
15. “Municipal Solid Waste,” Environmental Protection Agency, March 29, 2016,
https://archive.epa.gov/epawaste/nonhaz/municipal/web/html/.
16. If you run your dryer two hundred times a year, according to a Guardian column on the carbon footprint of everyday items, that would generate approximately half a ton of CO2.
M. Berners-Lee and D. Clark, “What’s the Carbon Footprint of . . . a Load of Laundry?,”
Guardian, November 25, 2010, https://www.theguardian.com/environment/green-living-
blog/2010/nov/25/carbon-footprint-load-laundry.
17. MIT students estimated that “Whether you live in a cardboard box or a luxurious mansion, whether you subsist on homegrown vegetables or wolf down imported steaks, whether
you’re a jet-setter or a sedentary retiree, anyone who lives in the U.S. contributes more than
twice as much greenhouse gas to the atmosphere as the global average.” Massachusetts
Institute of Technology, “Carbon Footprint of Best Conserving Americans Is Stil Double
Global
Average,”
Science
Daily,
https://www.sciencedaily.com/releases/2008/04/080428120658.htm.
18. Residents of Luxembourg, Qatar, Australia, and Canada have greater average levels of consumption and waste, according to the Global Footprint Network,
https://www.footprintnetwork.org/.
19. “Country Overshoot Days,” Earth Overshoot Day, https://www.overshootday.org/about-earth-overshoot-day/country-overshoot-days/.
20. The Yale economist Wil iam D. Nordhaus has argued that although 2° Celsius is not obtainable, 2.5° might be possible, although it would take extreme global policy measures to
get there. W. D. Nordhaus, “Protections and Uncertainties about Climate Change in an Era
of Minimal Climate Policies,” Cowles Foundation for Research in Economics, Yale
University,
December
2016,
https://cowles.yale.edu/sites/default/ les/ les/pub/d20/d2057.pdf.
21. Pennsylvania State University professor David Titley conjures up a powerful metaphor for gradations in temperature limits over 2°C. Think of 2° as the 30-mph signposted speed for a
truck going down a hil . Then each fraction or whole degree beyond 2 increases the speed of
the descending truck and consequently the ever-shortening odds of disaster. D. Titley, “Why
Is Climate Change’s 2 Degrees Celsius of Warming Limit So Important?,” The
Conversation, August 23, 2017, https://theconversation.com/why-is-climate-changes-2-
degrees-celsius-of-warming-limit-so-important-82058.
22. The Great Barrier Reef isn’t only one of the most stunning and unique ecosystems in the world, it’s also a huge part of Australia’s tourism industry. It rakes in $4.5 bil ion annual y in
revenue from tourists and provides jobs for 70,000 people. B. Kahn, “Bleaching Hits 93
Percent of the Great Barrier Reef,” Scientific American, April 20, 2016,
https://www.scienti camerican.com/article/bleaching-hits-93-percent-of-the-great-barrier-
23. Unless the global temperature increase can be held down to 1.5°C, the reef, whose area is equivalent to the size of Italy, wil not survive, coral scientists have concluded. N. Hasham,
“Australian Governments Concede Great Barrier Reef Headed for ‘Col apse,’ ” Sydney
Morning Herald, July 20, 2018, https://www.smh.com.au/politics/federal/australian-
governments-concede-great-barrier-reef-headed-for-col apse-20180720-p4zsof.html.
24. By the end of the century, scientists predict, the sea level could rise by between .5 and 1.4
meters. A rise of 5 meters could swamp 3.2 mil ion square kilometers of coastlines,
impacting 670 mil ion people. As warming waters impact Greenland and Antarctic ice, they
wil speed up the pace of the sea level’s rising worldwide. “Study Says 1 Bil ion Threatened by
Sea
Level
Rise,”
Worldwatch
Institute,
January
27,
2019,
http://www.worldwatch.org/node/5056.
25. The WHO breaks down its estimate of 250,000 extra deaths due to climate change per year between 2030 and 2050 into these categories: heat exposure kil ing the elderly (38,000),
diarrhea (48,000), malaria (60,000), and childhood undernutrition (95,000). “Climate
Change and Health,” World Health Organization, February 1, 2018,
http://www.who.int/mediacentre/factsheets/fs266/en/.
26. Max Planck’s Wissenschaftliche Selbstbiographie was translated from German by Frank Gaynor and published as A Scientific Autobiography in 1949 by Greenwood Press Publishers,
Westport, Connecticut.27. Brexit is a good example of this, Onder noted. Whereas only a quarter of youths voted to leave the European Union, six out of ten people 65 or older voted to leave. H. Onder, “The
Age
Factor
and
Rising
Nationalism,”
Brookings,
July
18,
2016,
https://www.brookings.edu/blog/future-development/2016/07/18/the-age-factor-and-
28. Those who are 80 or older—the “oldest-old,” according to the United Nations—are increasing in number faster than are older people (those over 60) overal . In 2015, there were
125 mil ion 80-plus-year-olds; by 2050 there are expected to be close to 450 mil ion.
Department of Economic and Social A airs, Population Division, World Population Ageing
2015
(New
York:
United
Nations,
2015),
http://www.un.org/en/development/desa/population/publications/pdf/ageing/WPA2015_
29. “Strom Thurmond’s Voting Records,” Vote Smart, https://votesmart.org/candidate/key-
30. In an incisive piece in the Nation, UCLA and Columbia Law ’School professor Kimberlé Wil iams Crenshaw highlighted some of the abhorrent double standards surrounding
Thurmond. “For most critics of sexual racism, this is simply a textbook case of a white man
getting away with sexual behavior that would have sent an African-American man to his
death,” she wrote. Indeed, in 1942, then Judge Thurmond sent a black man to the electric
chair “based on an al eged rape victim’s cross-race identi cation, testimony now known to be
extremely unreliable.” K. W. Crenshaw, “Was Strom a Rapist?,” Nation, February 26, 2004,
https://www.thenation.com/article/was-strom-rapist/.
31. The elderly poor’s only alternatives were family, friends, or the poorhouse. B. Veghte, “Social Security, Past, Present and Future,” National Academy of Social Insurance, August 13, 2015,
https://www.nasi.org/discuss/2015/08/social-security%E2%80%99s-past-present-future.
32. Men who reached age 65 in 1940 lived on average for another 12.7 years. By 1990, that average had climbed to 15.3 years. Women’s average life expectancy for the same period
(assuming they, too, survived to 65), increased by almost 5 years, to 19.6 years. “Life
Expectancy
for
Social
Security,”
Social
Security,
https://www.ssa.gov/history/lifeexpect.html.
33. By 2015, around 8 percent of the elderly were below the poverty line. “Per Capita Social Security Expenditures and the Elderly Poverty Rate, 1959–2015,” The State of Working
America, September 26, 2014, http://www.stateofworkingamerica.org/chart/swa-poverty-
gure-7r-capita-social-security/.
“Actuarial
Life
Table,”
Social
Security,
2015,
https://www.ssa.gov/oact/STATS/table4c6.html.
35. Wil iam Sa re tracked down the source of this quote for the New York Times in 2007: it was Kirk O’Donnel , a top aide to Tip O’Neil . W. Sa re, “Third Rail,” New York Times,
February 18, 2007, http://www.nytimes.com/2007/02/18/magazine/18wwlnsa re.t.html.“Social
Security
Bene ciary
Statistics,”
Social
Security,
https://www.ssa.gov/oact/STATS/OASDIbenies.html.
“Quick
Facts:
United
States,”
United
States
Census
Bureau,
https://www.census.gov/quickfacts/fact/table/US/PST045217.
38. Older voters have notably more impact when it comes to the primaries, according to Harvard professor of government Stephen Ansolabehere. “Older people tend to vote more often in
primaries,” he says. “And since the primary turnout tends to be lower, that means that bloc
can be even more important.” D. Bunis, “The Immense Power of the Older Voter,” AARP
Bulletin, April 30, 2018, https://www.aarp.org/politics-society/government-elections/info-
39. The days of long summer vacations (leaving continental Europe’s capitals al but empty), early retirement, and blanket medical insurance seem to be becoming a thing of the past in
Europe, wrote the Washington Post’s Edward Cody. “In the new reality, workers have been
forced to accept salary freezes, decreased hours, postponed retirements and health-care
reductions.” E. Cody, “Europeans Shift Long-Held View That Social Bene ts Are
Untouchable,”
Washington
Post,
April
24,
2011,
https://www.washingtonpost.com/world/europeans-shift-long-held-view-that-social-
bene ts-are-untouchable/2011/02/09/AFLdYzdE_story.html?utm_term=.bcf29d628eea.
40. Part of the reason there’s such a stark di erence in life expectancy, according to public health researchers, is the disappearance of smoking from the lifestyles of the rich and educated. S.
Tavernise, “Disparity in Life Spans of the Rich and the Poor Is Growing,” New York Times,
February 12, 2016, https://www.nytimes.com/2016/02/13/health/disparity-in-life-spans-of-
the-rich-and-the-poor-is-growing.html.
41. Joint Committee on Taxation, U.S. Congress, “History, Present Law, and Analysis of the Federal Wealth Transfer Tax System,” JCX-52-15, March 16, 2015,
https://www.jct.gov/publications.html?func=startdown&id=4744.
“SOI
Tax
Stats—Historical
Table
17,”
IRS,
August
21,
2018,
https://www.irs.gov/statistics/soi-tax-stats-historical-table-17.
43. Horses pul ing carriages littered the streets with dung. Corpses rotted in over owing graveyards. Garbage piled up in the streets. L. Jackson, Dirty Old London: The Victorian
Fight Against Filth (New Haven, CT: Yale University Press, 2015).
44. W. Luckin, “The Final Catastrophe—Cholera in London, 1886,” Medical History 21, no. 1
(January 1977): 32–42, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1081893/?
45. H. G. Wel s highlighted the possibility of world destruction stemming from the splitting of the atom, wrote Smithsonian writer Brian Handwerk, as wel as the future threat of portable
devices capable of mass destruction. “Wel s also clearly saw the dangers of nuclear
proliferation, and the doomsday scenarios that might arise both when nations were capable
of ‘mutual y assured destruction’ and when non-state actors or terrorists got into the fray.”B. Handwerk, “The Many Futuristic Predictions of H. G. Wel s That Came True,”
Smithsonian.com, September 21, 2016, https://www.smithsonianmag.com/arts-
culture/many-futuristic-predictions-hg-wel s-came-true-180960546/.
46. According to the author and lm historian Mark Clark, Wel s’s sci- classic Things to Come and the subsequent 1936 lm, which Clark claims was made under the author’s creative
control, was his attempt “to save the world. Literal y.” It’s a story of a world racked by war,
only to be o ered salvation by the Airmen. “They are a society of scientists and engineers
who, hidden away from the rest of the world, have made great scienti c advances and are
now prepared to lead humanity to a brighter future—so long as it submits to their
benevolent rule.” M. Clark, “Common Thread: Wel s and Roddenberry,” Onstage and
Backstage, July 29, 2013, https://onstageandbackstage.wordpress.com/tag/gene-
47. Roddenberry’s work echoed Wel s’s utopian-themed writing, Clark has pointed out. Ibid.
48. As Wel s noted time and again in his writings, these are the only two options for humanity.
49. A. van Leeuwenhoek, “Letters 43–69,” Digitale Bibliotheek oor de Nederlandse, April 25, 1679, http://www.dbnl.org/tekst/leeu027al e03_01/leeu027al e03_01_0002.php#b0043.
50. We have long been losing the battle to balance technological developments in goods and services with the environmental impact of population growth. “One Planet, How Many
People?”’
51. Edward O. Wilson, The Future of Life (2002; repr., New York: Vintage Books), 33. In a review in the New Yorker (March 4, 2002): “Wilson, an eminent evolutionary biologist,
shows the extent to which human prosperity, even in the information age, rests on the
foundation of a diverse natural world, since the more species any ecosystem has, the more
stable and productive it wil be.”
52. Nowhere has the debate about a land’s carrying capacity been greater than in Australia. The Dutch may have been the rst Europeans to discover the great southern land Terra Australis,
but it was the British who permanently colonized the habitable southeastern coastal strip in
1788. One hundred years after the prisoners took their rst steps on the beaches of Sydney
and most of the original inhabitants had been pushed out or wiped out by guns and
smal pox, the British brimmed with optimism about the country’s future. That made sense:
the American colonies were thriving, albeit a little too wel for British tastes, and the
Australian continent was just as large as America. In 1888, a story in the Spectator sounded
like a proud mother talking about her child’s future, with only a hint of racism, sexism, and
disdain for Americans: “There is every reasonable probability that in 1988 Australia wil be a
Federal Republic, peopled by 50 mil ions of English speaking men, who, sprung from the
same races as the Americans of the Union, wil have developed a separate and recognisable
type. . . . The Australians, we conceive, with more genial and altogether warmer climate,
without Puritan traditions, with wealth among them from the rst . . . wil be a softer,
though not a weaker people, fonder of luxury, and better tted to enjoy art . . . The discontent which permeates the whole American character wil be absent and, if not exactlyhappier, they wil be more at ease. The typical Australian wil be a sunnier man.” “Topics of
the Day: The Next Centenary of Australia,” Spectator 61 (January 28, 1888): 112–13.
Though the prediction about Australian male sunniness and relative paucity of
Puritanism was spot on, it was o on its math. After 1888, Australia’s population grew less
than half as fast as predicted, in large part due to the absence of arable land. In 2018, the
country had a population of only 25 mil ion. But most Australians disagree with Sheridan,
even more so after a few beers. They believe that the country is already overcrowded and the
land is nearing its carrying capacity. Cal s to limit immigration have dominated
conversations, talk shows, and politics for three decades, long before it was fashionable in the
United States. Many are deeply angry about the ever-increasing housing costs and
commuting times. Some are simply racist. Others are professional scaremongers. Ted
Trainer, Australia’s answer to Paul Ehrlich, has been arguing his whole career that levels of
human consumption and resource use are already unsustainable. I know because I took his
university course in 1988. According to Trainer, gasoline would run out before the 2000s
and we were al supposed to be starving by now. Trainer’s version of utopia is his alternative-
lifestyle, unkempt educational farm an hour’s drive south of Sydney, cal ed Pigface Point. I
spent a day there, learning by example, for example, that to save the world we needed to start
living on three-acre farms, use solar-powered ovens to cook homegrown eggs, and commute
an hour each way in a rusty, smoke-belching car to give lectures on green living. Yes, we have
major issues to solve—climate change being the most threatening. But contrary to Trainer’s
teachings, technology is not the enemy. In the arc of human history, technology has
ultimately come to our rescue. For most of us, our daily life is improving, and it wil
continue to improve, just as London in 1840 and New York in 1900 did. There are more
people than ever in the cities of North America, Europe, and Australasia, but today the
impact of each human is rapidly declining and, contrary to what I was taught in the 1980s
about the future, cities are getting cleaner. We’re moving from petroleum to natural gas to
solar and electricity. A visit to Bangkok used to provoke respiratory distress. Now there’s
blue sky. When I arrived in Boston in 1995, a splash of water from the harbor could land you
in the hospital or the grave. Now it is safe for swimming.
53. E. C. El is, “Overpopulation Is Not the Problem,” New York Times, September 13, 2013,
https://www.nytimes.com/2013/09/14/opinion/overpopulation-is-not-the-problem.html.
54. “World Population Projections,” Worldometers, http://www.worldometers.info/world-population/world-population-projections/.
55. Ibid. and Population Division, Department of Economic and Social A airs, United Nations Secretariat,
“2017
Revision
of
World
Population
Prospects,”
https://population.un.org/wpp/.
56. Gates’s argument is simple enough: when you improve children’s health so they don’t die at an early age, families choose to have fewer children. B. Gates, “Does Saving More Lives Leadto Overpopulation?,” YouTube, February 13, 2018, https://www.youtube.com/watch?
57. The others were Denmark, Finland, Norway, Great Britain, Germany, and France. M. Roser,
“Share of the Population Who Think the World Is Getting Better,” Our World in Data,
https://ourworldindata.org/wp-content/uploads/2016/12/Optimistic-about-the-future-
58. The Guardian asked, “To what extent, if at al , do you feel that today’s youth wil have had a better or worse life than their parent’s [ sic] generation?” Though the Chinese pol ed were
optimistic for the future of their youths, only 20 percent in the United Kingdom thought
things wil be better for future young people, with 54 percent expecting them to get worse.
The pol was taken in the face of rising rents, house prices, and university fees in Great
Britain, together with a steep drop in wages, which had in turn driven austerity policies. S.
Malik, “Adults in Developing Nations More Optimistic than Those in Rich Countries,”
Guardian, April 14, 2014, https://www.theguardian.com/politics/2014/apr/14/developing-
nations-more-optimistic-richer-countries-survey.
59. In developing countries, which may wel stil have high child mortality rates, they nevertheless are also seeing declines in numbers. Our World in Data’s Max Roser pointed out
that in sub-Saharan Africa child mortality has dropped consistently over the past fty years;
whereas it was one in four in the 1960s, it’s now one in ten. M. Roser, “Child Mortality,”
Our World in Data, https://ourworldindata.org/child-mortality.
60. Steven Pinker, Enlightenment Now: The Case for Reason, Science, Humanism, and Progress (New York: Viking, 2018), 51.
61. Among her many charms, gifts, and abilities, there was also a dry, self-deprecating wit. At a luncheon for women executives shortly before her death, Thompson said about what was
her last marathon, “I didn’t get much attention, even though I was coming in rst—I was
the only one in my age group.” R. Sandomir, “Harriette Thompson, Marathon Runner into
Her
90s,
Dies
at
94,”
New
York
Times,
October
19,
2017,
https://www.nytimes.com/2017/10/19/obituaries/harriette-thompson-dead-ran-
62. “Old Age: Personal Crisis, U.S. Problem,” Life, July 13, 1959, pp. 14–25.
63. The price that older unemployed workers pay for this discrimination is harsh. AARP writer Nathaniel Reade laid out a few of the statistics: “Forty-four percent of jobless workers 55 or
older had been unemployed for over a year in 2012, a Pew study reported. And while older
workers have a lower unemployment rate overal , the ones who lose their jobs can nd the
long hunt for work unbearable.” Many are forced to tap into their Social Security, which
puts not only their bene ts but a nancial y secure retirement at risk. N. Reade, “The
Surprising Truth About Older Workers,” AARP The Magazine, September 2015,https://www.aarp.org/work/job-hunting/info-07-2013/older-workers-more-valuable.html.
64. That’s according to research by Fabrizio Carmignani, a professor of business at Gri th University in Australia. F. Carmignani, “Does Government Spending on Education
Promote
Economic
Growth?,”
The
Conversation,
June
2,
2016,
https://theconversation.com/does-government-spending-on-education-promote-economic-
65. M. Avendano, M. M. Glymour, J. Banks, and J. P. Mackenbach, “Health Disadvantage in US
Adults Aged 50 to 74 Years: A Comparison of the Health of Rich and Poor Americans with
That of Europeans,” American Journal of Public Health 99, no. 3 (March 2009): 540–48,
https://www.ncbi.nlm.nih.gov/pubmed/19150903.
66. Of al the European countries, the United Kingdom wil have the oldest working population, having set the retirement age to rise to 69 by 2046. “Retirement in Europe,” Wikipedia,
https://en.wikipedia.org/wiki/Retirement_in_Europe.
67. “Impact of Automation,” Life, July 19, 1963, 68–88.
68. A. Swift, “Most U.S. Employed Adults Plan to Work Past Retirement Age,” Gal up, May 8,
http://news.gal up.com/pol /210044/employed-adults-plan-work-past-retirement-
age.aspx?g_source=Economy&g_medium=lead&g_campaign=tiles.
69. Only 25 percent said they’d stop working altogether at retirement age, according to Gal up.
Those who planned to work part-time after retirement age comprised 63 percent of those
pol ed. Ibid.
70. In 2014, Massachusetts ranked fth nationwide for the number of patents issued, having had an 81.3 percent increase over the prior ten years in patents issued to state inventors. E.
Jensen-Roberts, “When It Comes to Patents, Massachusetts Is a Big Player,” Boston Globe,
August 9, 2015, https://www.bostonglobe.com/magazine/2015/08/08/when-comes-
patents-massachusetts-big-player/3AmNfmSE8xWzzNbUnDzvPK/story.html.
71. D. Goldman, “The Economic Promise of Delayed Aging,” Cold Spring Harbor Perspectives in
Medicine
6,
no.
2
(December
18,
2015):
a025072,
http://perspectivesinmedicine.cshlp.org/content/6/2/a025072.ful .
72. The authors contend that “the social, economic, and health bene ts that would result from such advances may,” in the same way a “peace dividend” enables countries to rise out of
poverty, “be thought of as ‘longevity dividends,’ and that they should be aggressively
pursued as the new approach to health promotion and disease prevention in the 21st
century.” S. J. Olshansky, D. Perry, R. A. Mil er, and R. N. Butler, “Pursuing the Longevity
Dividend: Scienti c Goals for an Aging World,” Annals of the New York Academy of Sciences
114 (October 2017): 11–13, https://www.ncbi.nlm.nih.gov/pubmed/17986572.
73. Though 0.1 percent might not sound like much of the global population, that’s stil 7.8
mil ion ful -time researchers. “Facts and Figures: Human Resources,” UNESCO,
https://en.unesco.org/node/252277.
74. If the experiment sounds vaguely familiar, perhaps it’s because its inspiration came from the 1964 murder of Kitty Genovese in Queens, New York. Her screams for help reached the earsof thirty-eight neighbors, but none tried to help her. I. Shenker, “Test of Samaritan Parable:
Who
Helps
the
Helpless?,”
New
York
Times,
April
10,
1971,
https://www.nytimes.com/1971/04/10/archives/test-of-samaritan-parable-who-helps-the-
75. Seneca, the philosopher who lived from c. 5 BC to AD 65, wrote about the brevity of life, the art of living, and the importance of morality and reason. Seneca, On the Shortness of Life:
Life Is Long if You Know How to Use It, trans. G.D.N. Costa, Penguin Books Great Ideas
(New York: Penguin Books, 2004).
NINE. A PATH FORWARD
1. J. M. Spaight, Aircraft in War (London: Macmil an, 1914), 3.
2. This was one of what has become known as three “laws” penned by Clarke, each famous in its own right. The other two were “The only way of discovering the limits of the possible is
to venture a little way past them into the impossible” and “Any su ciently advanced
technology is indistinguishable from magic.” A. C. Clarke, “Hazards of Prophecy: The
Failure of Imagination,” in Profiles of the Future: An Inquiry into the Limits of the Possible
(New York: Orion, 1962), 14, 21, 36
3. L. Gratton and A. Scott, The 100 Year Life: Living and Working in an Age of Longevity (London and New York: Bloomsbury Publishing, 2018).
4. “And the days of Isaac were an hundred and fourscore years. And Isaac gave up the ghost, and died, and was gathered unto his people, being old and ful of days: and his sons Esau and
Jacob buried him.” Genesis 35:28, King James Version.
5. It was initial y funded by a clerk from the Treasury Department garnishing twenty cents a month from each merchant seaman’s wages to pay for a number of contract hospitals. “A
Short History of the National Institutes of Health,” O ce of NIH History,
https://history.nih.gov/exhibits/history/index.html.
6. That’s according to the Buck Institute for Research on Aging, which also noted that “if we move outside academic research to money spent on commercialized research applications by
private companies, the pie changes quite a bit. In aggregate, drug companies outspend the
NIH on R&D every year by over $20 bil ion.” “Who funds basic aging research in the US?,”
Fight Aging!, March 25, 2015, https://www. ghtaging.org/archives/2015/03/who-funds-
basic-aging-research-in-the-us/.
7. The authors highlight the looming crisis the globe faces in terms of the increasing number of aged. Come 2050, they estimate, the number of those over 60 wil be just over 2 bil ion, ve
times the number they were a century before. And 1.5 bil ion wil be from developing
countries. L. Fontana, B. K. Kennedy, V. D. Longo, et al., “Medical Research: Treat Ageing,”
Nature 511, no. 750 (July 23, 2014): 405–7, July 24, 2014,https://www.nature.com/news/medical-research-treat-ageing-1.15585.
8. “Estimates of Funding for Various Research, Condition, and Disease Categories (RCDC),”
National
Institutes
of
Health,
May
18,
2018,
https://report.nih.gov/categorical_spending.aspx.
9. R. Brookmeyer, D. A. Evans, L. Hebert, et al., “National Estimates of the Prevalence of Alzheimer’s Disease in the United States,” Alzheimer’s & Dementia 7, no. 1 (January 2011):
61–73, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3052294/.
10. The average American spends $1,100 on co ee every year. “2017 Money matters report,”
Acorns, 2017, https://sqy7rm.media.zestyio.com/Acorns2017_MoneyMattersReport.pdf.
“Actuarial
Life
Table,”
Social
Security,
2015,
https://www.ssa.gov/OACT/STATS/table4c6.html.
12. Looking back over his life in a wide-ranging interview with Nautilus’s Jordana Cepelewicz, Hay ick noted that what money was invested in aging research did not go to where he felt it
should. Most studies of aging focus on longevity determinants or diseases linked to age, he
said. “Less than 3 percent of the budget of the National Institute on Aging in the past decade
or more has been spent on research on the fundamental biology of aging.” J. Cepelewicz,
“Ingenious:
Leonard
Hay ick,”
Nautilus,
November
24,
2016,
http://nautil.us/issue/42/fakes/ingenious-leonard-hay ick.
13. The lm Gattaca is about a future society driven by eugenics in which children are genetical y selected to ensure that they possess the best hereditary traits. A father asks the
geneticist, “We were wondering if we should leave some things to chance.” The geneticist
responds, “You want to give your child the best possible start. Believe me, we have enough
imperfection built in already. Your child doesn’t need any additional burdens.” A. Nicols,
director, Gattaca, 1997.
14. The authors calculated that “from 1970 to 2000, gains in life expectancy added about $3.2
tril ion per year to national wealth, with half of these gains due to progress against heart disease alone.” A cure for cancer “would be worth about $50 trillion.” K. M. Murphy and R.
H. Topel, “The Value of Health and Longevity,” Journal of Political Economy 114, no. 5
(October
2006):
871–904,
https://ucema.edu.ar/u/je49/capital_humano/Murphy_Topel_JPE.pdf.
15. D. Goldman, B. Shang, J. Bhattacharya, and A. M. Garber, “Consequences of Health Trends and Medical Innovation for the Future Elderly,” Health Affairs 24, suppl. 2 (February 2005):
W5R5–17,
https://www.researchgate.net/publication/7578563_Consequences_Of_Health_Trends_A
nd_Medical_Innovation_For_The_Future_Elderly.
16. For a good read, I recommend Bil Bryson’s books on this topic. Notes from a Big Country (UK)/ I’m a Stranger Here Myself (USA), 1999; and Down Under, 2000, are my favorites.
17. A phrase often used by US politicians, from the Parable of Salt and Light in Jesus’s Sermon on the Mount. In Matthew 5:14, Jesus tel s his listeners, “You are the light of the world. Acity that is set on a hil cannot be hidden.”
18. It’s clearly impacting the workforce. The trend to live longer “is one factor contributing to a steady rise in the workforce participation rate of older Australians, especial y women,” wrote
Matt Wade in the Sydney Morning Herald. “About one in ve Australian workers are now
aged over 55 years, compared with less than one in 10 in the 1980s and 1990s.” M. Wade,
“Trend for Australians to Live Longer Reshapes Economy,” Sydney Morning Herald,
August 12, 2018, https://www.smh.com.au/business/the-economy/trend-for-australians-to-
live-longer-reshapes-economy-20180810-p4zwuv.html?btis.
19. What does the phrase “Medicare for al ” mean? According to a CNBC article, Reuters de nes it as “a publicly nanced, privately delivered system with al Americans enrol ed and
al medical y necessary services covered.” Meanwhile, the cost of health care for US citizens
keeps on climbing. “The average annual deductible for employer-sponsored health care
plans, which make up most of the plans in the U.S., was $1,505 in 2017, compared to $303
in 2006,” according to the Kaiser Family Foundation, wrote Yoni Blumberg for CNBC
Make It. Y. Blumberg, “70% of Americans Now Support Medicare-for-Al —Here’s How
Single-Payer Could A ect You,” CNBC Make It, August 28, 2018,
https://www.cnbc.com/2018/08/28/most-americans-now-support-medicare-for-al -and-
20. “Australians Living Longer but Life Expectancy Dips in US and UK,” Guardian, August 16,
2018, https://www.theguardian.com/society/2018/aug/16/australians-living-longer-but-life-
21. Indeed, Americans who live in the top-income counties enjoy on average twenty years more of life than those living in the poorest counties, wrote Senator Bernie Sanders, and that is at
least partly due to what he termed “grossly unequal access to quality healthcare.” B. Sanders,
“Most Americans Want Universal Healthcare. What Are We Waiting For?,” Guardian,
August 14, 2017, https://www.theguardian.com/commentisfree/2017/aug/14/healthcare-a-
human-right-bernie-sanders-single-payer-system.
22. In fact, the Patient Factor lists at the top of the world’s health care systems (courtesy of the World Health Organization) the fol owing countries: (1) France, (2) Italy, (3) San Marino,
(4) Andorra, (5) Malta. “World Health Organization’s Ranking of the World’s Health
Systems,” The Patient Factor, http://thepatientfactor.com/canadian-health-care-
information/world-health-organizations-ranking-of-the-worlds-health-systems/.
23. “My father says that America has the best healthcare system in the world. What can I say to
prove him wrong?,” Quora, https://www.quora.com/My-father-says-that-America-has-the-
best-healthcare-system-in-the-world-What-can-I-say-to-prove-him-wrong.
24. N. Hanauer, “The Pitchforks Are Coming . . . For Us Plutocrats,” Politico, July/August
2014, https://www.politico.com/magazine/story/2014/06/the-pitchforks-are-coming-for-
us-plutocrats-108014.See
International
Journal
of
Astrobiology,
https://www.cambridge.org/core/journals/international-journal-of-astrobiology.
26. P. Dayal, C. Cockel , K. Rice, and A. Mazumdar, “The Quest for Cradles of Life: Using the Fundamental Metal icity Relation to Hunt for the Most Habitable Type of Galaxy,”
Astrophysical Journal Letters, July 15, 2015, https://arxiv.org/abs/1507.04346.
“List
of
Nearest
Terrestrial
Exoplanet
Candidates,”
Wikipedia,
https://en.wikipedia.org/wiki/List_of_nearest_terrestrial_exoplanet_candidates.
28. George Monbiot, “Cutting Consumption Is More Important Than Limiting Population,”
“George
Monbiot’s
Blog,”
Guardian,
February
25,
2009,
https://www.theguardian.com/environment/georgemonbiot/2009/feb/25/population-
29. S. Pinker, Enlightenment Now: The Case for Reason, Science, Humanism, and Progress (New York: Penguin Random House, 2018), 333.
30. One home builder told CNBC reporter Diana Olick that young adults are reluctant to leave rental apartments decked out by owners to be “resortlike,” given that they can’t a ord to buy
an apartment with similar amenities. Olick found that younger Americans “seem to be
drawn to smal er, simpler living,” citing the tiny-house trend, one underpinned by
technology equipping smal spaces with “big amenities.” D. Olick, “Why Houses in America
Are Getting Smal er,” CNBC, August 23, 2016, https://www.cnbc.com/2016/08/23/why-
houses-in-america-are-getting-smal er.html.
31. A $20 bil ion New York start-up cal ed WeWork focuses on o ering shared working environments rich with immediately accessible amenities. The eight-year-old business, at the
time of David Gel es’s New York Times business pro le, had “built a network of 212 shared
working spaces around the globe” and was in the process of putting up the fteen-story
Dock 72 on the East River. Along with a vast coworking space, “there wil be a juice bar, a
real bar, a gym with a boxing studio, an outdoor basketbal court and panoramic vistas of
Manhattan. There wil be restaurants and maybe even dry cleaning services and a
barbershop.” D. Gel es, “The WeWork Manifesto: First, O ce Space. Next, the World,” New
York Times, February 17, 2018, https://www.nytimes.com/2018/02/17/business/the-
wework-manifesto- rst-o ce-space-next-the-world.html.
32. And let us not forget the water we expend on producing crops and meat that we never eat.
2013 estimates put the amount of water being used for food production by 2050 at 10 to 13
tril ion cubic meters a year, which is 3.5 times as much as the amount of fresh water
currently consumed by the planet’s population. J. von Radowitz, “Half of the World’s Food
‘Is
Just
Thrown
Away,’ ”
Independent,
January
10,
2013,
https://www.independent.co.uk/environment/green-living/half-of-the-worlds-food-is-just-
33. Carl R. Woese Institute for Genomic Biology, University of Il inois at Urbana-Champaign,“Scientists Engineer Shortcut for Photosynthetic Glitch, Boost Crop Growth 40%,” Science
Daily,
January
3,
2019,
https://www.sciencedaily.com/releases/2019/01/190103142306.htm.
34. P. Mirocha, and A. Mirocha, “What the Ancestors Ate,” Edible Baja Arizona, September/October 2015, http://ediblebajaarizona.com/what-the-ancestors-ate.
35. J. Wenz, “The Mother of Al Apples Is Disappearing,” Discover, June 8, 2017,
http://blogs.discovermagazine.com/crux/2017/06/08/original-wild-apple-going-
36. “Vitamin A de ciency is the leading cause of preventable childhood blindness and increases the risk of death from common childhood il nesses such as diarrhoea,” according to a late-2017 UNICEF report. It stated that vitamin A had been shown to “reduce al -cause
mortality by 12 to 24 percent, and is therefore an important programme in support of e orts
to reduce child mortality.” “Vitamin A De ciency,” UNICEF, February 2019,
https://data.unicef.org/topic/nutrition/vitamin-a-de ciency/.
37. Luciano Marra ni and Erik Sontheimer at Northwestern University in Evanston, Il inois, were the rst to show how CRISPR protects bacteria from foreign DNA: the interference
machinery targets DNA directly. “From a practical standpoint, the ability to direct the
speci c, addressable destruction of DNA . . . could have considerable functional utility,
especial y if the system can function outside of its native bacterial or archaeal context,” they
wrote. L. A. Marra ni and E. J. Sontheimer, “CRISPR Interference Limits Horizontal
Gene Transfer in Staphylococci by Targeting DNA,” Science 322, no. 5909 (December 19,
2008): 1843–45, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2695655/; see also J.
Cohen, “How the Battle Lines over CRISPR Were Drawn,” Science, February 17, 2017,
https://www.sciencemag.org/news/2017/02/how-battle-lines-over-crispr-were-drawn.
38. M. R. O’Connel , B. L. Oakes, S. H. Sternberg, et al., “Programmable RNA Recognition and Cleavage by CRISPR/Cas9,” Nature 516, no. 7530 (December 11, 2014): 263–66,
https://www.ncbi.nlm.nih.gov/pubmed/25274302.
39. L. Cong, F. A. Ran, D. Cox, et al., “Multiplex Genome Engineering Using CRISPR/Cas
Systems,”
Science
339,
no.
6121
(February
15,
2013):
819–23,
https://www.ncbi.nlm.nih.gov/pubmed/23287718.
40. Court of Justice of the European Union, “Organisms Obtained by Mutagenesis Are GMOs
and Are, in Principle, Subject to the Obligations Laid Down by the GMO Directive,” July
https://curia.europa.eu/jcms/upload/docs/application/pdf/2018-
41. “Secretary Perdue Statement on ECJ Ruling on Genome Editing,” U.S. Department of
https://www.usda.gov/media/press-
releases/2018/07/27/secretary-perdue-statement-ecj-ruling-genome-editing.
42. LEDs are tiny, no larger than a eck of pepper, and mixing primary-colored LEDs, i.e., red, green, and blue, results in white light. “LED Lighting,” Energy Saver,https://www.energy.gov/energysaver/save-electricity-and-fuel/lighting-choices-save-you-
43. In 2016, the City of Angels achieved an 11 percent emissions cut (the same as 737,000 cars coming o the streets) via an improved public transport system and solar energy investments,
while creating “30,000 new green jobs,” wrote Matt Simon in Wired. M. Simon, “Emissions
Have Already Peaked in 27 Cities—and Keep Fal ing,” Wired, September 13, 2018,
https://www.wired.com/story/emissions-have-already-peaked-in-27-cities-and-keep-fal ing/.
44. It turned out that the source of the pol ution was “problematic connections between the pipes that carried sewage and those that were meant to carry clean rainwater out to the river,”
according to a Citylab story by Stephanie Garlock. When a rainstorm hit, “everything in the
pipes, sewage and al , would ush directly into the Charles and its tributaries through older
drainage pipes.” Renovations of the sewage systems have al but eradicated the problem. S.
Garlock, “After 50 Years, Boston’s Charles River Just Became Swimmable Again,” Citylab,
July 19, 2013, https://www.citylab.com/life/2013/07/after-50-years-bostons-charles-river-
just-became-swimmable-again/6216/.
45. The farm cost 200 mil ion Australian dol ars to construct and has a solar plant made up of 23,000 mirrors that re ects the heat of the sun to a solar tower. Rather than soil, the tomato
plants grow in “a watery solution fed by nutrient-rich coconut husks.” E. Bryce, “These
Farms Use Sun and Seawater to Grow Crops in the Arid Australian Desert,” Wired,
February 14, 2017, https://www.wired.co.uk/article/sundrop-farms-australian-desert. See also Sundrop Farms, http://www.sundropfarms.com.
Letter
from
Joseph
Wharton,
December
6,
1880,
https://giving.wharton.upenn.edu/wharton-fund/letter-joseph-wharton/.
47. P. Sopher, “Where the Five-Day Workweek Came From,” Atlantic, August 21, 2014,
https://www.theatlantic.com/business/archive/2014/08/where-the- ve-day-workweek-
CONCLUSION
1. E. Pesheva, “Rewinding the Clock,” Harvard Medical School, March 22, 2018,
https://hms.harvard.edu/news/rewinding-clock; see also A. Das, G. X. Huang, M. S.
Bonkowski, et al., “Impairment of an Endothelial NAD+-H2S Signaling Network Is a
Reversible Cause of Vascular Aging,” Cell 173, no. 1 (March 2018): 74–89,
https://www.sciencedirect.com/science/article/pi /S0092867418301521.
2. J. Li, M. S. Bonkowski, S. Moniot, et al., “A conserved NAD+ Binding Pocket That Regulates Protein-Protein Interactions During Aging,” Science 355, no. 6331 (March 24,
2017): 1312–17, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5456119/.
3. President’s Council on Bioethics, Beyond Therapy: Biotechnology and the Pursuit of Happiness (New York: HarperCol ins, 2003), 190.4. Ibid., 192.
5. Ibid., 200.
6. “ICD-11 for Mortality and Morbidity Statistics: MG2A Old Age,” World Health
https://icd.who.int/browse11/l-
m/en#/http://id.who.int/icd/entity/835503193.
7. Bravo Probiotic Yogurt, https://www.bravo-probiotic-yogurt.com/.
8. Y. Guan, S.-R. Wang, X.-Z. Huang, et al., “Nicotinamide Mononucleotide, an NAD+
Precursor, Rescues Age-Associated Susceptibility to AKI in a Sirtuin 1–Dependent
Manner,” Journal of the American Society of Nephrology 28, no. 8 (August 2017): 2337–52,
https://jasn.asnjournals.org/content/28/8/2337; see also S. Wakino, K. Hasegawa, and H.
Itoh, “Sirtuin and Metabolic Kidney Disease,” Kidney International 88, no. 4 (June 17,
2015): 691–98, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4593995/.
IndexA note about the index: The pages referenced in this index refer to the page numbers in the
print edition. Clicking on a page number wil take you to the ebook location that corresponds to
the beginning of that page in the print edition. For a comprehensive list of locations of any word
or phrase, use your reading system’s search function.
NOTE: Bold page numbers refer to picture captions.
AARP, 229
AAV (adeno-associated virus), 165
accidents, and death, 70
acetylated adenine ribose, 119
acetyls, 24, 34, 37, 45, 47, 118, 119, 134
actinobacterium, 120–21
aerospace, 270
age, tests for, 73
aged mind-set: experiment for, 72
aging
beginning of, 72
bene ts of delayed, 256–59
causes of, 8–18, 19, 20–23, 28, 35, 43, 44, 45–50, 52, 54, 57, 60
chaos as, 47
complexity of, 147
“cure” for, 145–46
as disease, 67–70, 80–84, 264, 268–69, 270, 299, 302–3
evolution of, 72–73
ghting, 80–84
hal marks/symptoms of, 15, 17–18, 19, 20, 28, 32, 38, 50, 52, 73–74, 77, 84, 147
as inevitable, 81, 89, 214and mapping human genome, 28
as natural process, 82
season analogy about, 147
stopping, 41–44
theories/hypotheses about, 13–18, 19, 20–23
as treatable condition, 81–82, 84, 127
universal regulators of, 147–48
universal/singular theory about, 8, 13, 14, 17, 19, 20–23
and why we age, 8–18, 19, 20–23, 38, 39, 62, 84
See also specific researcher or topic
Agouti gene, 37
agriculture. See consumption; food
Airbus A380 thought experiment, 157–58
airport scanners, 114
Al iance Assurance Company, 69, 246
al icin, 131
alpha-ketoglutarate, 173
alpha lipoic acid, 14
Alt, Frederick, 44–45
Alzheimer’s
and aging as disease, 67
cause of, 200
as characteristic of aging, 79
and death as a choice, 279
funding for research about, 267
ongoing research about, 297, 298
and senescence research, 67
and sirtuins, 24
and temperature, 108
treatment for, 18
volunteers for research about, 87–88
and yeast studies, 30
See also dementia; memory
American Association for the Advancement of Science, 285
American Medical Association, 285
amino acids, 25, 34, 99–102, 103, 117, 214
Amon, Angelika, 299
Amorim, Joao, 296AMPK gene, 26, 46, 56, 57, 103, 112, 125, 126, 129, 144–45, 148–49, 264, 296
analog information, 20–21, 22–23, 35, 60, 62, 127, 160, 161, 162
See also epigenome/epigenetics
Anderson, Rozalyn, 95
“antagonistic pleiotropy” (Medawar), 11, 152
antiretrovirals, 155
apes: senescence in, 152–53
apples, 285
arginine, 101
Aristotle, 10
Armstrong, David, 74
Aron, Jan, 124
arthritis, 18, 24, 52, 79, 200
arti cial intel igence, 178
asthma, 24
atherosclerosis, 24
athletes
and beginning of aging, 73
biosensors/trackers for, 188
Attia, Peter, 98
Australia
aging as disease in, 270
bushwalking in, 307, 308, 309–10
death in, 274
elderly in, 275
funding in, 274–75
health care in, 274–76
Nick (Sinclair) accident in, 204–5
optimism about world in, 247
physician-assisted suicide in, 280
problems in, 274–76
Sinclair lab research in, 50, 297
Sinclair’s youth/education in, 30–31, 32, 70, 105, 305, 309
Sundrop Farms in, 289treatment as right in, 274–76, 277
Austriaco, Nicanor, 42
azo dyes, 113
babies
aging of, 115
“designer,” 174–75
baby food, 94
Baker, Darren, 152
Bartke, Andrzej, 66
Barzilai, Nir, 66–67, 96, 126, 127, 299
Bates, Gil ian, 66
Baur, Joseph, 132
Bavarian, Roxanne, 297–98
Bayer, 184
Belmonte, Juan Carlos Izpisua, 166
beta-galactosidase, 150
betaine, 305
Beth Israel Deaconess Medical Center, 75, 76, 108
Beyond Therapy (President’s Council on Bioethics), 300–301
Biden, Beau, 9
Biden, Joe, 9
bilateral symmetry, 5
biological sciences
predictions about advances in, 263
See also specific researcher or topic
biosensors/trackers, 187–91, 192, 193–99, 214, 232, 273, 299
Biosphere 2 (research experiment), 92–93
birds: and why we age, 11–12
birth certi cate: mistake on, 83
birth rate, 244, 245, 247, 248
Bishop, Bronwyn, 274
bisphenol A, 37
Blackburn, Elizabeth, 149, 299Blasco, Maria, 66
blastocysts, 49
blindness. See vision
blocked carotid arteries, 191, 193
blood
monitoring of, 186, 188, 190–91, 304
and Sinclair’s personal regimen, 304
See also blood glucose; blood pressure; blood sugar
blood pressure, 93, 94, 95, 184
blood sugar, 93, 123–24, 132, 188, 190, 191
Bober, Eva, 43
Boguski, Mark, 176–78
bone loss, 52
bone marrow transplants, 17–18
Bonkowski, Michael, 62, 66, 295–96
Boston Children’s Hospital, 75, 200
Boston, Massachusetts
hospitals in, 75, 76, 108, 200
jobs in, 255
Sinclair arrival in, 105–6
Brady, Tom, 73
BRAF inhibitor, 10
brain, 12, 22, 78, 172, 297, 298
breathing
biosensors/trackers for, 189
See also lungs
Brenner, Charles, 135
Brigham and Women’s Hospital, 75
Brimley, Wilford, 208
bristlecone pine trees, 53–54, 56
“brown fat,” 108–9, 110–11, 214
Brunet, Anne, 299
Buck Institute for Research on Aging, 299
Budnik, Bogdan, 295
Buettner, Dan, 88
Bu ett, Warren, 252Bush, George W., 301
butter ies, 37
butylated hydroxytoluene, 15
Calment, Jeanne, 215
calorie restriction (CR), 91–99, 100, 105, 106, 111, 125, 130–31, 133, 158, 173
See also diet; fasting
Campbel , Keith, 16
cancer
and average lifespan, 77
and biosensors/trackers, 188, 194
change in thinking about, 10
as characteristic of aging, 67, 79, 80
cost of innovative medicine for, 273
and death as a choice, 279
diagnosis of, 177, 178–79, 186, 213
and elderly, 253
ght against, 9, 28, 42, 72, 79–80
funding for research about, 267, 268
and genetics, 71
and geroncogenesis, 79
immuno-oncological approaches to, 156
incidence of, 77
as inevitable and irreversible, 80
and metformin, 125
in mice, 155–56
and nitrates, 114
ongoing research about, 9–10, 66, 295, 298
and organization of modern medical culture, 76, 77
pharmaceuticals for, 9, 176, 179, 184
and precision medicine, 178, 180
and radiation, 114and resveratrol, 132
and senescence, 67, 151, 152, 153
and Sinclair’s mother, 70–71, 72, 79
and sirtuins, 24
survival rates for, 10
symptoms of, 138
and telomeres, 149
testing for, 49
vaccines for, 155–56
and why it happens, 9–10
and yeast studies, 30
Cannon Street Railway Bridge (London), 234–35, 236
Capel i, Peter, 251
capil aries: and reversing of aging, 63
carbon, 37
carcinogens, 114
cardiovascular disease, 77, 99, 101, 125
See also heart disease/problems
Carlson, Anton, 95–96
carnitine, 99
“carrying capacity” of planet, 220–25, 239–43, 283–90
Carter, Jimmy, 179
catalase, 118
caterpil ars, 37
Celexa, 183
celiac disease, 182
cel s
and attempts to explain life, 118
copies of, 5
di erentiation among, 36ex-di erentiation of, 60
and exercise, 104
and fasting, 96
“fate” of, 59
number of enzymes in, 118
resetting of age of, 54
and sirtuins, 43
cel ular scale: and exercise, 102–3
centenarian study, 96–97
Centers for Disease Control and Prevention, 102
Cepko, Connie, 294
cerebral vascular disease, 92
CETP, 126–27
chameleon lizard, 12
Charpentier, Emmanuel e, 286–87
checkpoint blockade therapy, 179
chemotherapy, 9, 48, 76, 139, 141, 154, 182, 183, 185, 273, 296–97
chickenpox, 202
children
and aging of babies, 115
genetical y altered, 174–75, 233
and great-grandparents, 292–93
healing of, 74
and senescence, 153
See also birth rate; childbirth
cholesterol, 93, 94, 124, 134, 191
chromosomes, 36, 149, 159, 182, 296
chronic il ness, 280
Chwalek, Karolina, 295Clarke, Arthur C., 262–63
climate
and broken DNA, 44
and concern for future, 261, 293
and consumption, 285, 286, 287, 289–90
and global carrying capacity, 240
and population growth, 220–25
research about, 258
as threat, 285
cloning, 15–16, 23, 159, 161, 162, 306
cnidarians, 297
coastal ooding, 224
Cobbett, Wil iam, 237
Cocoon (movie), 208
code of life, 27–28
Cohen, Haim, 45
communication, cel ular, 17, 22–23, 45, 161–62
Congress, U.S., 226, 265–66, 267
consciousness, 5
consumption
and “carrying capacity” of planet, 283–90
climate and, 285, 286, 287, 289–90
and concern about the future, 293
and death of “stu ,” 283–84
“dematerialization” and, 283
of food, 284–86
genetic modi cation and, 284–86, 287
and genome editing, 286–87
natural life cycle and, 301
and population growth, 283–90
and technology, 283–90
Cook, Tim, 252
COPD, 77
Copeland, Royal, 110
Copine2, 298
Coppotel i, Giuseppe, 298
Cornaro, Alvise (Luigi), 90
cosmic rays, 7Coumadin, 183
CR Society International, 93
Crimmins, Eileen, 78
crisis mode, evolution and, 16–18, 19, 20–23
Crouch, Ian, 208
Cruise, Tom, 208
cryotherapy, 110
CT scans, 44
Cutting, Windsor, 109
Cyteir Therapeutics, 42
cytoplasm, 155
DALY (disability-adjusted life year), 78–79
Das, Abhirup, 297
dasatinib, 154
Dawes, Ian, 30–31
Dayal, Pratika, 278
de Cabo, Rafael, 100, 124, 132–33
death
acceptance of, 10–13
average age of, 78
and biosensors/trackers, 191
causes/reasons for, 10–13, 62, 67–70, 72, 89
chance of, 69
as choice, 278–82
and deterioration, 69
and diet, 286
diseases and, 89
impact on population growth of, 245, 247
inevitability of, 119
internal clocks and, 69–70
and Law of Human Mortality, 69–70
lifestyle and, 89and mortality tables, 246
from motor vehicle accidents, 205
and NAD, 134
prediction of, 69–70
prevention of unnecessary, 180
scienti c mandate for, 264
of “stu ,” 283–84
universal model of life and, 41
See also specific disease
de bril ators, 78
delayed aging: bene ts of, 256–59
dementia, 52, 68, 77, 78, 80, 81, 125, 134, 150
See also Alzheimer’s; memory
“designer babies,” 174–75
DeStefano, Susan, 294–95
Deursen, Jan van, 152
diabetes
in Australia, 275
and biosensors/trackers, 188
cause of, 150
as cause of death, 68
as characteristic of aging, 79, 82
cost of innovative medicine for, 273
diet and, 95
and elderly, 253
and guanide/guanidine, 123–24
and healing, 74–75
incidence of, 77
as inevitable and irreversible, 80
insurance coverage for, 232
metformin and, 124–27
and NMN, 136
ongoing research about, 295
and organization of modern medical culture, 76
sirtuins and, 24
STACs and, 136
and sugar, 123–24and wounds, 74–75
diagnosis
in the future, 213
ongoing research about, 299
and precision medicine, 176–80
and technology, 181–86
See also specific disease
Dickinson, Richard, 31
diet
advice about, 88
“best,” 88
biosensors/trackers for, 188, 190, 191, 192
and death, 286
and disease, 91
DNA and, 91
epigenetics and, 91
impact on lifespan of, 88, 89–95, 97, 99, 101–2, 106, 111, 113–14
and self-knowledge, 182
and Sinclair’s personal regimen, 304, 307
sirtuins and, 25
and stress, 113–14
studies about, 91–95
survival circuit and, 24, 91, 98, 99, 102
and technology, 214
and universal regulators of aging, 148
vegetarian, 101–2
weight and, 95
See also calorie restriction; fasting; food
digital information, 20, 23, 60, 160
digoxin, 183–84
dimethyl biguanide, 124
dinitrophenol, 109–10discrimination, age, 251–53, 254, 258–59
disease
aging as, 67–70, 80–84, 264, 268–69, 270, 299, 302–3
Barzilai study of, 66–67
and beginning of aging, 70–75, 79
and bene ts of delayed aging, 257
biosensors/trackers and, 188, 192, 193–94
communicable, 273
cost of ghting/treating, 258, 273
and death as a choice, 279–80
and diet, 91
focus on individual, 75–80
and FOXO gene, 57
funding for research about, 268–69, 273, 299
increase in, 77
and Law of Human Mortality, 67–70
and NAD, 24
ongoing research about, 298
and organization of modern medical culture, 75–80, 77
prediction about surviving, 71
and shift from miasmatic theory to germ theory, 237–38
sirtuins and, 24
and technology, 243
and universal model of life and death, 41
and universal regulators of aging, 148
See also diagnosis; treatment; specific disease
“Disposable Soma Hypothesis” (Kirkwood), 11–12
divisions
and health care as a right, 277–78
DNA
and attempts to explain life, 118, 119
biosensors/trackers and, 189, 191, 194
characteristics of, 20
complexity of, 159
consumption and, 287and diagnosing disease, 201
and diet, 91
as digital information, 20
and economic divisions, 232
and epigenetic landscape, 58
and epigenome, 36
and funding for aging research, 271
helicases, 33
and mapping human genome, 27–28
metformin and, 126
methylated, 169, 170, 171, 181, 306
ongoing research about, 294
packaging of, 24, 37, 41, 47, 119
and piano analogy, 37
and precision medicine, 178, 179, 180
and reprogramming, 159, 169, 170, 171
and self-knowledge, 181
sequencing of, 178, 179, 181, 194, 197, 201, 232, 294
and sirtuins, 24
as storing and copying information, 14, 20, 44, 47, 114, 160
and telomeres, 149
and threats to humans, 197
and understanding aging, 117
and universal model of life and death, 41
and why we age, 14, 15, 16, 20
and yeast studies, 30–35, 38, 39, 40–44, 152
See also DNA, broken/damaged; DNA, repair of
DNA, broken/damaged
as cause of aging, 13–14
and epigenetics, 38, 59, 60, 61, 137
and evolution of aging, 5, 6, 7, 26and nitrates, 114
number of, 44
ongoing research about, 297
reprogramming and, 162, 171, 174
and resveratrol, 132
retrotransposons and, 154, 155
and smoking, 79
sugar and, 44
and survival circuit, 44, 45, 46–47, 48, 50, 52, 57, 162
and telomeres, 149
and universal model of life and death, 41
and why we age, 17
and yeast studies, 35, 42, 43, 152
DNA methyl-transferases (DNMTs), 59
DNA, repair of
and evolution of aging, 5, 6, 7
ongoing research about, 296, 298
and retrotransposons, 155
and sirtuins, 24, 25, 43, 44, 45, 46, 48, 137
smoking and, 79
and stress, 113
survival circuit and, 44, 45, 47
and telomeres, 149
and TOR gene, 25
DNP (2, 4-dinitrophenol), 109–10
doctors
and biosensors/trackers, 192
and funding in aging research, 269
lack of aging knowledge of, 88
misdiagnosis by, 177, 178, 179–80, 213
as only conduit for diagnosis, 185
and patients at center of their own care, 180
and physician-assisted suicide, 280
and technology, 176–77
and treatment as a right, 272
video home visits by, 186wait time for, 185–86
Dol y (cloned sheep), 16, 159, 161
Dongsheng Cai, 105
double helix, 28
Doudna, Jennifer, 286–87
drug overdoses, 78
drugs. See pharmaceuticals
Duesberg, Peter, 9
DVD analogy, 20, 22, 23, 35, 52, 158, 159, 160, 161, 164, 171
Ebola virus, 196
economics/economy
and betterment of individual lives, 247
and elderly, 255–59
and expansion of lifespan, 219, 230–31
in the future, 261
haves and have-nots and, 231–34
and health care costs, 256–59
and retirement, 255–56
social insecurity and, 230–31
and treatment as a right, 272
education, 250, 255, 258, 259, 290–91
Ehrlich, Anne, 222
Ehrlich, Paul, 222
elderly
in Australia, 275
as burden/problem, 250–51
and cost of medical care, 256–59
diseases of, 253
and economy, 255–59
in the future, 265
poverty among, 227–31
as revered, 250
role of, 249–56
in workforce, 229, 251–56See also great-grandparents
Eli Lil y, 42
El is, Erle C., 241–42
El ison, Martin, 230
embryonic development, 21
embryos: aging of, 115
emphysema, 71
environment. See climate
epigal ocatechin gal ate, 131
epigenetic landscape, 58–60, 61, 62, 112–15, 137, 145, 158, 164, 166
epigenetic noise, 137–41, 145–46, 150, 160, 161, 162, 214, 297, 379
epigenome/epigenetics
and analog information, 60
and blood sugar, 123
as cause of aging, 35, 44, 48–50
and chaos, 38
complexity of, 36
and economic divisions, 232
evolution of, 60
and expansion of lifespan, 145–46
and how it works, 137–41
and information storage, 160
and Information Theory of Aging, 36, 48–50
and life expectancy, 245
and lifespan in future, 214, 218
and longevity in mammals, 55
meaning of, 21
and metformin, 126
in mice, 164
ongoing research about, 295, 296, 297
and piano analogy, 36–38
and pine trees, 54
power of, 122and rapamycin, 122
and reprogramming, 161, 162, 164, 169, 170, 214, 218
and reverse aging, 63
and Shannon communication theory, 162
smoking and, 79
and storage of information, 160
stress and, 112–15
and survival circuit, 47, 48–50, 52, 57
and telomeres, 149
and universal model of life and death, 41
Waddington research on, 21
See also analog information; epigenetic landscape; epigenetic noise; Waddington, Conrad H.
Erbitux, 183
ERCs (extrachromosomal ribosomal DNA circles), 40–41, 42, 43, 44, 47, 91
“Error Catastrophe Hypothesis” (Orgel), 14
ethics
and aging as disease, 303
and Airbus A380 thought experiment, 157–58
and expansion of lifespan, 218–19
and funding for aging research, 270–71
of reprogramming, 173–75
euchromatin genes, 36
European Union, 287
Evagrius Ponticus, 90
evolution, 3–5, 6, 7–8, 12–13, 16, 119, 152–53, 220
exercise
and beginning of aging, 73
and biosensors/trackers, 190
diet and, 104–5
expansion of lifespan and, 145
and longevity, 26, 104, 129mimic of, 26
and NAD, 136
and reverse aging, 62–63
and Sinclair’s personal regimen, 304, 307
sirtuins and, 25
and technology, 214
and universal regulators of aging, 148
See also physical tness
experimentation: importance of, 274
eyes. See glaucoma; macular degeneration; vision
Farr, Wil iam, 237
fasting
and exercise, 105
and expansion of lifespan, 145
impact on lifespan of, 89–99
intermittent, 95–96, 98, 133, 145, 304
and longevity genes, 26
and NAD, 136
and resveratrol, 133
and temperature, 112
See also calorie restriction; diet
Federal Food, Drug, and Cosmetic Act, U.S. (1938), 109–10
Feinstein, Dianne, 226
Feng Zhang, 287
Ferry, Edna, 91
fertility, 34, 47, 138–39, 140–41, 149, 258, 297
See also infertility; reproduction; sterility
Feynman, Richard, 119
tness. See exercise; physical tness
ies
death of, 69
See also fruit ies
folic acid, 37“The Fol ower” (Heaney poem), 250–51
food
consumption of, 284–86
genetical y modi ed, 284–86
and population growth, 222, 223, 224, 244
and Sinclair’s personal regimen, 304
wasting of, 284
See also diet
Food and Drug Administration, U.S., 127, 184, 188
Foot, Philippa, 158
FOXO genes. See DAF-16 gene
FOXO genes, 56
FOXO3 genes, 56, 57, 118, 119, 126–27
frailty, 52, 62, 63, 75, 82, 125, 298, 301
“Free Radical Theory of Aging” (Harman), 14–15
free radicals, 14–15
“French paradox,” 130
Friedreich’s ataxia, 136
frogs: reprogramming of, 159, 164
fruit ies, 43, 91, 108, 122, 132
Ful er, Ida May, 228
funding
for aging research, 81, 258, 265–71, 293, 294, 299, 301, 303
in Australia, 274–75
and concern for future, 293
and consumption, 290
and de nition of disease, 266
federal, 265–67
and opposition to aging research, 301
for vaccines, 203
future
description of world in, 264–65great-grandparents in, 292–93
imagining the, 207–9
and London example, 234–39
making decisions about, 261
math concerning the, 213–20
planning for the, 293
population concerns about, 220–25
prolonged youthfulness in, 265
questions to ask about, 207
See also optimism/pessimism
G. officinalis, 145
Gasser, Susan, 41–42
gastrointestinal conditions, 89
Gates, Bil , 197, 203–4, 216, 245, 252
Gates, Melinda, 203–4
Gattaca (movie), 233
Gencaro, 184
gender, 182–83
See also women
See also “mating-type” genes
gene B, 5, 6, 7, 24, 25, 34, 39, 45
gene therapy, 164, 173–75, 294, 296
See also reprogramming
genes
and attempts to explain life, 118
and cause of aging, 28
and evolution of aging, 4, 5, 6, 7–8
funding for research about, 299
number of, 36
and piano analogy, 36–38
and purpose of gene circuit, 7–8
sequencing of, 209
silencer, 5switching/swapping of, 33
transfer of, 5
and yeast studies, 30
See also epigentics; gene therapy; genetics; reprogramming; specific gene
genetic modi cation, 174–75, 214, 233, 236, 284–86, 287
See also cloning; reprogramming
genetics
and biosensors/trackers, 191
and cancer, 71
and cause of aging, 13–14
and evolution of aging, 4
outlawing of improvements in, 271
and technology, 186
See also cloning; genetic modi cation; genome/genomics; reprogramming
genistein, 37
genome/genomics
biosensors/trackers and, 191
and diagnosis of disease, 201
evolution of, 60
and evolution of aging, 7
functions of, 37
letters in human, 181
mapping of human, 27–28
nuclear, 15
ongoing research about, 297
pharmaceuticals and, 184–85
and piano analogy, 36–38
precision medicine and, 177–78, 179
and reprogramming, 172
retrotransposons and, 155
and self-knowledge, 182, 183, 184
and sequencing, 177–78
sirtuins and, 128
and survival circuit, 48
and universal model of life and death, 41
and why we age, 14, 15, 39, 40and yeast studies, 35, 39, 40, 41, 42
germ theory: shift to, 237–38
German Center for Neurodegenerative Diseases, 122
geroncogenesis, 79
glaucoma, 152, 154, 164–65, 167, 169
GlaxoSmithKline, 203–4
Glenn Center for the Biology of Aging (Harvard), 298
Glenn Center for the Biology of Aging (MIT), 299
Global Climate Action Summit (2018), 289
Global Foodprint Network, 223
global warming. See climate
glucokinase, 117
See also blood glucose
gluten, 182
Goldwater, Barry, 229
Good Samaritan, 265
Goodal , David, 280
Gorbunova, Vera, 155
Gotta, Monica, 41–42
Grassley, Chuck, 226
Graunt, John, 68
gray hair, 32, 38, 50, 73, 79, 82, 165, 166, 300
Great Barrier Reef, 224
great-grandparents, 292–93
Great Mouse Treadmil Failure (2017), 62–63, 136, 297
Greek Orthodox Church: and fasting, 97
Gregory-Ksander, Meredith, 169
Greider, Carol, 149
Gri n, Marie, 202
Gri n, Patrick, 297
“group selection,” 10–11
growth: and TOR gene, 25
guanidine, 123–24
guanine, 79
Guarente, Leonard, 32–33, 35, 38, 41, 42, 127, 134, 299, 310
Gueniot, Alexandre, 90Gutenberg, Johannes, 250
H1N1 virus, 194
Haigis, Marcia, 298
hair loss, 32
Haldane, J.B.S., 11
Hal , Michael, 122
hal marks/symptoms of aging
and beginning of aging, 73–74
complexity of, 147
discovery of, 28
epigenetics and, 52
and organization of modern medical culture, 77–78, 77
and singular theory of aging, 19
and Werner syndrome, 32
and why we age, 15, 17–18, 20, 38, 84
See also specific hallmark or symptom
Harman, Denham, 14–15
Harry Potter and the Goblet of Fire, 50
Hartley, Ralph, 161
Harvey, Wil iam, 102
haves and have-nots, 231–34
Hawking, Stephen, 222–23
HDAC (histone deacetylase), 47
He Jiankui, 174
health care. See medical care
healthspan
and cost of medical care, 256–59
of elderly, 243
and lifespan, 282
and organization of modern medical culture, 78pharmaceuticals for extension of, 273
Heaney, Seamus, 250–51
heart disease/problems
biosensors/trackers for, 189, 193
as cause of death, 68
as characteristic of aging, 67, 79, 80
and death as a choice, 279
and elderly, 253
and “French paradox,” 130
funding for research about, 267
importance of, 205
incidence of, 77
and Lyme disease, 200
and organization of modern medical culture, 76, 78
protein and, 99
and resveratrol, 132
and sirtuins, 24
and temperature, 112
and Werner syndrome, 32
See also cardiovascular disease
Heilbronn, Leonie, 93
Heitman, Joseph, 122
Helen Hay Whitney Foundation Fel owship, 32
helicases, 33
hepatitis, 75
heterochromatin genes, 36
hip fractures, 73–74
Hippocrates, 90
histone methyltransferases (HMTs), 59
histones, 21, 24, 34, 36, 45, 47, 118, 119, 134, 149, 171
HIV, 155, 157, 174, 188Hoelzel, Frederick, 96
Holonyak, Nick, 288
homeostasis, 106
Hooke, Robert, 116
Hopper, Grace, 58
hormone replacement therapy, 267
Horvath, Steve, 52
See also specific hospital
Human Capital Index, 275
Human Genome Project, 177, 181
humans
di culties of testing on, 91–92, 93
mammals compared with, 56
and mapping of human genome, 27–28
survival circuit in, 8
and why we age, 8–13
See also specific topic
Hundred Years Warning, 220–25
hunger. See calorie restriction; diet; fasting; food
Hunt, Greg, 270
Hurricane Katrina analogy, 45–46, 47
Huxley, Thomas Henry, 239
Hydra vulgaris polyp, 54
hypothermia, 111
hypoxic response, 104
Ideker, Trey, 52
IGF-1 (insulin-like growth factor 1), 66, 96–97
Ikaria, Greece, 97
Imai, Shin-ichiro, 42, 45, 134, 136
immortality: why don’t we have, 11–13
immune system, 10, 22, 42, 113, 121, 150, 156, 165, 178, 179, 213implants, 192
See also transplants
income
and concern about the future, 293
divisions/inequality in, 231–34, 277–78
and life expectancy, 231–34
and treatment as a right, 277
See also poverty; standard of living; wealth
infections
and biosensors/trackers, 188, 192
and death, 70
funding for study of, 265
and senescence, 153
spleen, 205
symptoms of, 138
as threat to life expectancy, 195–98
See also fertility; reproduction; sterility
in ammation
biosensors/trackers for, 189, 191
and NAD, 134
ongoing research about, 296
and resveratrol, 132
and retrotransposons, 155
and TOR gene, 25
in uenza, 89, 122, 194–95, 195, 202, 204, 276
information
aging as loss of, 20–23
and attempts to explain life, 119
availability of patient, 213
consolidation of biotracking/sensor, 197
and DVD analogy, 159, 160, 161
loss of, 26, 159, 160, 161, 162and reprogramming, 159, 160, 161–62
and reverse aging, 23
storage/backup of, 14, 20, 22, 44, 47, 114, 160, 161–62, 169, 297
types of, 20–21
See also type of information
Information Theory of Aging
attempts to disprove, 38, 40–44
basis for, 23–26
and causes of aging, 48–50, 57
epigenetic noise as central to, 47, 57
epigenome and, 36
and ICE mice research, 49–50, 51, 52
importance of, 84
and lifespan in future, 218
and loss of information, 160
ongoing research about, 295
and preservation of information, 22
reprogramming and, 170
reverse aging and, 63
and Shannon communication studies, 162
survival circuit and, 23–26, 57
and why we age, 38
yeast studies and, 29–35
innovation: optimism about future, 213–20
inoculations. See vaccines
InsideTracker, 191
Institute of International Law, 262
See also diabetes
insurance companies, 69, 246, 303
internal clocks: and death, 69–70
International Classification of Diseases (WHO), 68, 302–3
investments, people as, 252–53, 256
iPSC cel s, 163–64
Ipsos MORI, 248
isoleucine, 101
Japan, 88, 92, 206, 217, 275jel y sh, 54, 56, 57, 158, 297
Jenner, Edward, 148
Johnson, Bradley, 42
Johnson, Julie, 184–85
Johnson, Lyndon, 229
Johnston, John, 29–30
Jun Li, 298
Kagawa, Yassuo, 92
Kane, Alice, 298
Karcher, Jean-Frederic, 198
Kel y, Kevin, 216–17
kidneys, 75, 79, 124, 136, 150, 166, 172, 205, 207
Kirkland, James, 154
Kirkwood, Thomas, 11–12
Ksander, Bruce, 169
Kuhn, Thomas, 16
laboratories
medical testing in, 199–201, 258
tour of Sinclair, 294–98
See also specific laboratory or researcher
Lamming, Dudley, 100
landscape
See also epigenetic landscape
larotrectinib, 184
Lassa fever, 204
Law of Human Mortality, 69–70, 76, 246
Lawan Kuhn, 176, 177, 178, 180, 182
Lecomte du Nouy, Pierre, 74
Leeuwenhoek, Antonie van, 148, 239–40
leucine, 101Levine, Benjamin, 62
Li-Huei Tsai, 299
life
attempts to explain, 116–20
quality of, 273
life expectancy
100 years as, 245
120 years as potential, 245
150 years as, 245
average, 195
decline in, 276
and evolution, 153
and u epidemic (1918), 195
gains in, 195
impact of expansion of, 217–20
and income, 231–34
increase in, 70
math about, 213–20
reprogramming and, 245
and technology, 245
threats to, 194–99
See also lifespan; specific nation
lifespan
and death as a choice, 281
and diet, 102
ethics about expansion of, 218–19
and evolution of humans, 220
expansion of, 116–20, 144–46, 213–31, 264, 277
in the future, 213–20
and healthspan, 282
lack of knowledge about, 116–17
predictions about, 263
skeptics about, 263–64
technology impact on, 213–20, 264
universal model of, 41
See also life expectancy; specific topic
lifestyle, 11–12, 89, 93, 110–11, 145, 190, 239lighting, 288
LINE-1 retrotransposons, 154–55
LINE-1 virus ( ctional virus), 81–82
List of Essential Medicines (WHO), 124
Lombard, David, 42
longevity
and gender, 182–83
as inevitable, 219
longevity genes
and evolution of aging, 23–26
and expansion of lifespan, 264
mapping human genome and, 28
ongoing research about, 296, 298
survival circuit and, 57
and treatment as a right, 278
underutilization of, 102
See also sirtuin genes; specific gene or sirtuin
Longo, Valter, 95
See also breathing
macrophages, 150
macular degeneration, 25, 164–65
See also vision
Ma ei 1 galaxy, life on, 278–79
Magna superstes, 4–5, 7, 12, 23, 24, 38, 45, 60, 106, 119
malaria, 203–4
mammals
humans compared with, 56
See also specific mammal
Manhattan Project, 14
maple syrup urine disease, 31marathons: elderly as running in, 249–50
Martin, George, 32
A Mathematical Theory of Communication (Shannon), 22–23, 162, 163
“mating-type” genes, 33, 34, 39, 43, 45, 47, 48, 69, 128, 149
Mayo Clinic, 104
McBurney, Michael, 44
McCay, Clive, 91
McGlothin, Paul, 93–94
McNabb, David, 42
measles, 203
meat, 88, 99, 100, 101, 114, 286, 304
MECP2 gene, 306
medical care
availability of, 248
equitable access to, 271–78
optimism/pessimism about, 215–16, 248
organization of modern, 75–80, 81
and precision medicine, 177–78
quality of, 271–78
and quality of life, 273
refocusing/transformation of, 26, 177–78
and treatment as a right, 271–78
U.S. as best, 277
See also doctors; hospitals
medical records: trust with, 197–98
medicines. See pharmaceuticals
Melton, Douglas, 32
See also Alzheimer’s; dementia
Mendel, Lafayette, 91
meningitis, 203
menopause, 138–39, 194menstruation, 138–39, 141, 194
The Merck Manual of Geriatrics, 80–81
metabolism, 4, 17, 24, 25, 43, 45, 101, 109, 126
metformin, 124–27, 129, 131, 137, 141, 143, 144, 232, 264, 269, 304
methionine, 100–101
methylation, 52
methylfolate, 305
methyls, 37, 169, 170, 171, 172, 305
miasmatic theory, 237–38
mice
Bramble Cay melomys, 224
cancer in, 155–56
and cholesterol, 124
death of, 69
diabetes in, 136
diet of, 91, 92–93, 94, 99, 100, 101
and DNA damage, 114
and epigenetics, 37, 48–50, 52, 60, 164
exercise/treadmil tracking program for, 62–63, 136, 297
gender of, 182
genetical y modi ed, 214
ICE, 49–50, 51, 52, 60, 114, 166–67, 295
in ammation in, 153
and Kirkwood theory, 11–12
LMNA, 166
metformin in, 124–25
old versus sick, 50
ongoing research about, 296, 297, 298
premature aging in, 166
and radiation exposure, 114
and rapamycin, 122
and reprogramming, 164, 165, 166–69, 170, 172
and resveratrol, 132–33and retrotransposons, 155
reverse aging in, 62–64, 167–68
Richardson–Van Remmen experiments with, 15
senescence in, 150, 152, 153, 154, 155
SIRT2 and, 140
SIRT6 in, 155
survival circuit studies and, 49–50, 51, 52
tumors in, 172
universal regulators of aging in, 147
and weight, 124
See also rats; rodents
Michelson, Albert, 216
Micrographia (Hooke), 116
Mil s, Kevin, 42
MinION, 181
MIT (Massachusetts Institute of Technology)
biosensor/trackers research at, 188
Glenn Center at, 299
See also specific researcher
Mitchel , Jay, 100
mitochondria
and cause of aging, 14
exercise and, 104
and long-term wel -being, 84
metformin and, 125
ongoing research about, 298
resveratrol and, 132
STACs and, 136
and survival circuit, 52
and temperature, 108, 109, 110
and why we age, 15, 17, 38Mohammed, Parvez, 298
Monbiot, George, 283
Morris, Margaret, 136
Mortimer, Robert, 29–31
Mosquirix, 204
Mostovslavsky, Raul, 44–45
mother, Sinclair’s See Sinclair, Diana (mother)
mother, student’s: and NMN, 138–39
Movva, Rao, 122
MSN2 gene, 56
mTOR enzyme
and emergency situations, 46
exercise and, 103
identi cation of, 122
as longevity pathway, 129
prevalence of, 25
and reproduction, 141
and self-knowledge, 183
and stress, 112
in yeast, 122
mumps, 203
Murthy, Mala, 42
muscles, 24, 52, 63, 101, 102, 103, 137, 150, 191
mutations
cause of, 15
of DAF-16 genes, 56
deleterious, 54
ongoing research about, 297, 298
in pine trees, 54
and precision medicine, 179
reprogramming and, 164, 170and self-knowledge, 182
senescence and, 152
and smoking, 79
survival circuit and, 52
and why we age, 11, 14, 15, 16, 23
N-nitroso compounds, 114
NAD (nicotinamide adenine dinucleotide)
and attempts to explain life, 118, 119
discovery of, 134
and epigenetic landscape, 137
exercise and, 103
and expansion of lifespan, 145, 264
fertility and, 138–39
functions of, 24, 129, 134, 137, 140
Imai research about, 42
metformin and, 126
rapamycin and, 121
and retrotransposons, 155
senescence and, 155
and Sinclair’s personal regimen, 305
as STAC, 134–37
and yeast, 155
nanoworld: and understanding aging, 117–20
National Academy of Sciences, 285
National Aeronautical and Space Agency (NASA), 296
National Institute on Aging, 268
National Institutes of Health (NIH), 16, 79, 182, 265–66, 267, 268, 288
National Oceanic and Atmospheric Association (NOAA), 224
national security, 263
nationalism, 226
natural: what is, 242–43
natural life cycle, 300–302natural selection, 11, 12, 152, 243
Navratilova, Martina, 73
nerves/nervous system, 137, 150, 167, 168, 171, 200, 296–97
neurons
degeneration of, 132, 136, 190
and epigenetics, 22
ongoing research about, 297
Newton, Isaac, 239
nicotinamide, 305
nicotinamide mononucleotide. See NMN
Nicoya, Costa Rica: as centenarian-heavy place, 88
NMN (nicotinamide mononucleotide), 135–37, 138, 139, 140, 141–44, 148, 297, 298, 305, 306
Nobel Prizes, 30, 122, 149, 164, 216, 286
“noisy-channel coding theorem” (Shannon), 161–62
Noori, Indra, 252
Notch genes, 167
NR (nicotinamide riboside), 135, 136–37, 305
nucleolar explosion, 91
Nyquist, Harry, 161
Nystrom, Thomas, 66
Obama, Barack, 9
obesity, 78, 104, 108, 109–10, 267, 275
See also weight
Okinawa, Japan: as centenarian-heavy place, 88, 92
Olshansky, S. Jay, 76–77
Oncotype DX, 182
Onder, Harun, 226
optic nerves. See vision
optimism/pessimism
and “carrying capacity” of planet, 243
and consumption, 283
about expansion of lifespan, 213–20
about future, 172–73, 248and medical care availability, 248
of medical professors, 215–16
and population growth, 220–25, 243, 247, 249
and reprogramming, 172–73
of Sinclair, 213–20, 234, 243, 299–300
and standard of living, 247, 248
about technology, 209
organ donors, 205–7
organic food, 131
organohalides, 113
Orgel, Leslie, 14
Osborne, Thomas, 91
oxidation: and cause of aging, 14
pain: cause of, 62
Parable of the Good Samaritan, 259–60
Partridge, Linda, 66
Pasteur, Louis, 237–38
Patrick, Rhonda, 188
PD-1 inhibitors, 155
PD-L1 inhibitors, 156
Pearce, Christie, 73
Pelosi, Nancy, 226
Perdue, Sonny, 287
pets: on NMN, 306
Pew Research Center, 220–21
pharmaceuticals
adverse reactions to, 138
and aging as disease, 270, 303
and attempts to explain life, 120
and Barzilai study of diseases, 67
bene ts of, 149contribution to lifespan of, 89
development of, 276
and economic divisions in U.S., 232
and expansion of lifespan, 146
for extension of healthspans, 273
funding for development of, 269, 270, 299
and the future, 214
impact on aging of, 120–23
insurance coverage for, 232
and knowledge about aging, 18
for longevity, 273
and mapping human genome, 28
metabolizing of, 183
ongoing research about, 298
and precision medicine, 179
as prolonging life, 273
purpose of, 120
retirement age and, 229
and self-knowledge, 181–82, 183
and sirtuins, 24
survival mechanisms and, 146
and WHO List of Essential Medicines, 124
See also specific pharmaceutical or disease
phosphorus, 117
See also exercise
piano analogy, 36–38
Pichardo-Casas, Israel, 295
pigs: as organ donors, 206–7
pine trees, bristlecone, 53–54, 56
Planck, Max, 225
plants
protein from, 99–102
and Sinclair’s personal regimen, 304
stressed, 131survival circuits of, 131
plasmids, 49
Plavix, 183
politics/politicians
and concern about the future, 293
consumption and, 283
and divisions in American society, 232
and expansion of lifespan, 219, 229–30
health care system and, 277
and hundred-year politician, 225–27
social insecurity and, 229–30
polycystic ovary syndrome, 141
polyphenols, 101
population
and “carrying capacity” of planet, 220–25, 239–43, 283–90
child mortality and, 248
and consumption, 283–90
decline in annual rate of, 244–45
and expansion of lifespan, 226, 244–45, 247–49
and food supply, 222, 223, 224, 244
growth of, 283–90
Leeuwenhoek views about, 239–40
optimism/pessimism about growth in, 220–25, 247, 249
and standard of living, 247, 248, 249
See also birth rate
poverty, 227–31, 248, 264, 277, 293
See also income; standard of living
Poznan University of Medical Sciences, 130
precision medicine, 177–80
predictions
of Clarke, 262–63
about death, 69–70
economic, 230–31of Hawking, 222–23
about IGF-1, 96–97
about lifespan, 263
model for future, 230
and population in the future, 222–23, 224
about surviving disease, 71
about technology, 263
of Wel s, 236
premature aging, 16, 43, 153, 166, 296
President’s Council on Bioethics, 300–301
Prevnar vaccine, 202
Princeton Theological Seminary: good samaritans at, 259–60
printing living tissue, 207, 214, 294
privacy issues, 197–98, 270–71
Program for Appropriate Technology in Health (PATH), 203–4
protein
and amino acids, 99
animal, 99
and attempts to explain life, 117, 118, 119
bene ts of, 99
and consumption, 286
epigentics and, 21
“good,” 101
and guanidine, 123
and metformin, 125
ongoing research about, 295, 297
plant, 99–102
and precision medicine, 178
and self-knowledge, 185
senescence and, 150
and Sir2, 66
sources of, 99–102sugar and, 123
and survival circuit, 23
TOR gene and, 25
and why we age, 17
and yeast studies, 34
See also specific protein
public health
in London, 234–36
as threat, 194–99
pulmonary disease, 71
radiation, 9, 14, 44, 46–47, 114–15, 156, 179, 287, 296
Rajman, Luis, 295
Rando, Thomas, 299
rapalogs, 264
rapamycin, 120–23, 129, 137, 141, 154
rats
and exercise, 104
and reprogramming, 164
and temperature, 108
See also mice
Ravussin, Eric, 93
rDNA. See ribosomal DNA
Rebelo, Bernard, 110
Reich, David, 294
religion, 294–95
reproduction
and epigenetic landscape, 59
and evolution of aging, 5, 6, 7, 8
survival circuit and, 24, 45, 46, 47
and why we age, 11–12
See also fertility; infertility; sterilityreprogramming
and chromosomes, 159
and DNA, 159, 162, 169, 170, 171, 174
and epigenetics, 158, 161, 162, 164, 166, 169, 170
ethics of, 173–75
and expansion of lifespan, 264
and genome, 172
histones and, 171
immune system and, 165
and information, 159, 160, 161–62
and Information Theory of Aging, 170
life expectancy and, 245
and lifespan in future, 218
methyls and, 169, 170, 171, 172
and mice, 164, 165, 166–69, 170, 172
on-going research about, 296–97
and optimism about future, 172–73
purposes of, 205–7
and retirement, 255
reverse aging and, 163, 164, 167–68
and RNA, 169
sirtuins and, 164
and stem cel s, 163–64
and survival circuit, 162, 171
and transplants, 207
unanswered questions about, 172
See also gene therapy; specific disease or injury
research, aging
bene ts of, 299–300
cancer research compared with, 10
expansion of ongoing, 294–300
funding for, 81, 258, 265–71, 276, 293, 294, 299, 301, 303
opposition to, 300–302
and scienti c interest in aging, 269
and tour of Sinclair labs, 294–98
volunteers for, 87–88, 93, 173See also specific researcher or research topic
research centers, medical: organization of, 76, 81
resilience: acceptance of loss of, 75
resveratrol, 129, 130–33, 134, 296, 304
retirement, 229, 230, 253, 255–56, 291, 293
See also work/workforce
reverse aging
and bene ts of delayed aging, 257
and digital information, 23
and fertility, 140–41
and information storage and recovery, 23
ongoing research about, 298
reprogramming and, 163, 164, 167–68
and Shannon’s studies, 163
should we do?, 64
Sinclair father’s story about, 141–44
revolution: and health care as a right, 277–78
ribosomal DNA (rDNA), 34, 39, 40–41, 43, 44, 69, 128
See also ERCs
ribosomes: and attempts to explain life, 117
Richardson, Arlan, 15
right: treatment as a, 303
Rinaldi, Conrad, 298
robots, 18
Roddenberry, Gene, 236
rodents
and cancer, 125
death of, 280
Harman experiments with, 14–15
and metformin, 125
and NAD, 141
as precursors for human experiments, 141
See also mice; rats
Rogan, Joe, 110Ross, Jaime, 297
Rothschild, Nathan Mayer, 69, 246
roundworms, 56, 66, 69, 132, 147
Royal Society of London for Improving Natural Knowledge, 65–67, 84, 238–39, 246, 310
RPE65 mutation, 164
rubel a, 203
running out of money, 255–56
Ryan/Vitéz, Vera (grandmother), 74, 76, 275, 307, 308, 310
Sabatini, David, 122
Saccharomyces cerevisiae (yeast), 29, 30–31, 111, 121
Samuelson, Paul, 225
sanitation systems, 234–39
Sardinia, Italy: as centenarian-heavy place, 88
“sauna bathing,” 111–12
scanners, 188
See also biosensors/trackers
Schrodinger, Erwin, 117
science
and attempts to explain life, 116–20
as boring, 118
and elderly in workforce, 253
and evolution of aging, 13
and evolution of scienti c discovery, 16–18, 19, 20–23, 119
and mapping human genome, 28
predictions about advances in biological, 263
See also technology; specific scientist or research topic
scienti c method, 243
scientists
age of, 262–63
and immortality, 281
interest in aging research of, 269
need for visionary, 289–90
washed-up, 264
See also specific scientistScott, Andrew, 230
Scripps Research Institute, 106, 108
seasons analogy, 147
sedentary lifestyle, 78, 103, 141
Sedivy, John, 155
Sehgal, Suren, 120–21
Selesniemi, Kaisa, 298
self-knowledge, 181–86
sel sh genes, 11, 12, 49, 82, 154–55
senescence
causes of, 149
deleting/kil ing of, 151, 153–54, 156, 214
epigenomes/epigenetics and, 61, 150, 152, 155
and evolution, 152–53
function of, 152
Gems research about, 67
and Hydra vulgaris polyp, 54
and long-term wel -being, 84
in mice, 150, 152, 153, 154, 155
mutations and, 152
and NAD, 155
“negligible,” 53
and pharmaceuticals, 151, 153–54
premature aging and, 153
protein and, 150
STAC and, 128
and stress, 152
and survival circuit, 151, 152
and technology, 153
TOR gene and, 25
and universal model of life and death, 41
and why we age, 17, 18, 20, 38
See also specific disease
“senomorphic” molecules, 154sequencing
of DNA, 178, 179, 181, 194, 197, 201, 232, 294
and technology, 181
sequencing machines, 18
SERPINA1 gene, 71
SGS1 (Slow Growth Suppressor 1), 33, 34, 42–43
Shannon, Claude, 22–23, 161–63, 163, 170, 171, 297
sharks, Greenland, 54–55
Sharpe, Arlene, 179
sheep: reprogramming of, 16, 159, 161
“silencing” protein, 5
Sinclair, Alex (son), 57, 144, 189, 218–19, 310
Sinclair, Andrew (father), 141–44, 183–84, 250, 259, 275, 305, 307, 308, 309–10
Sinclair, Ben (son), 304, 307, 309–10
Sinclair, David
accomplishments of, 42–43
and death as a choice, 281–82
family of, 305–6
letters to, 87–88
lifespan for, 307
optimism of, 213–20, 234, 243, 299–300
personal regimen of, 303–7
retirement of, 255
thoughts about life of, 305–6
tour of lab of, 294–98
Sinclair, Diana (mother), 70–71, 72, 79, 141, 183–84, 279
Sinclair, Natalie (daughter), 57, 144, 200–201, 310
Sinclair, Nick (brother), 205, 206, 306, 307
Sinclair/Luikenhuis, Sandra (wife), 111, 133, 143, 144, 189, 200, 281, 300, 302, 305–6, 307, 310
Sir2
and cause of aging, 127–28
functions of, 34, 35, 39, 42, 43, 47–48, 66, 140
and Law of Human Mortality, 69
and mating-type genes, 149and STACs, 135
and survival circuit, 47–48
and temperature, 111
and why we age, 39
and yeast studies, 34, 35, 39, 149
SIR2 gene
and epigenetic noise, 137
extra copy of, 48
functions of, 34, 41, 43, 45, 48
and naming of sirtuins, 24
and resveratrol, 130
and STACs, 135
See also Sir2
SIRT
functions of, 45
and survival circuit, 45
See also specific enzyme/gene
SIRT1
and attempts to explain life, 118–19
and exercise, 103
functions of, 43
Howitz study of, 128
longevity of, 44
and metformin, 125
ongoing research about, 296
and reversing of aging, 63
See also STAC
SIRT1-6 genes, 43, 45, 103, 108, 126–27, 140, 155
sirtuin genes
and cause of aging, 51
and DAF-16 gene, 56
and diet, 91
discovery of, 127–28
and DNA breaks, 128
and epigenetics, 59, 60, 137–38exercise and, 103
and expansion of lifespan, 264
functions of, 24–25, 36, 45–47, 129
and genomes, 128
and ICE mice research, 51
as longevity pathway, 129
manipulation of, 25
and metformin, 126
ongoing research about, 298
reprogramming and, 164
resveratrol and, 132
and retrotransposons, 155
and senescence, 155
stress and, 112
and survival circuit, 25, 43, 44, 45–47, 48, 49, 50, 51, 52, 57, 135
and telomeres, 149
and yeast studies, 34–35, 38, 39, 40–41, 42, 43
See also specific researcher
sitting-rising test (SRT), 73
Skerritt, John, 270
skil baticals, 291
skin cel s, 298
skin patches, 189
Smeal, Tod, 42
smoking, 37, 70–72, 79–80, 97, 104, 113, 114, 304
Snow, John, 237
social change: and expansion of lifespan, 219
Social Security, 228–29, 254, 256, 291, 293
sodium nitrite, 114
Sohn, Joel, 297
Southern blot technique, 38, 40
spinal cord injuries, 167, 172, 173spleen, 205
SRT1720/SRT2104 (STAC), 133
STAC (SIRT1-activating compound), 128, 130–37, 145, 148–49, 296
See also specific STAC
standard of living, 247, 248, 249
See also income; poverty; wealth
Stanford University: cancer research at, 155–56
statins, 78
stem cel s
access to, 231
and cause of aging, 51
and epigenetic landscape, 58–59
fertility and, 140
and ICE mice research, 51
ongoing research about, 298
and printing living tissue, 207
reprogramming and, 163–64
and telomeres, 149
and vaccines, 156
sterility, 41, 42, 43, 44, 45, 69, 91
See also infertility; reproduction
Sterne, Jean, 124
Streisand, Barbra, 161
Streptococcus pneumoniae, 202
Streptomyces hygroscopicus, 120–21, 145
stress
and biosensors/trackers, 188, 190
diet and, 113–14
and epigenetic landscape, 137
and evolution of aging, 26
examples of, 26
NAD and, 137
pharmaceuticals for, 26
and senescence, 152
and survival circuit, 57, 100temperature and, 111
stroke, 77, 95, 124, 132, 193, 279
sugar, 44, 48, 88, 123–24, 188, 190, 304
See also blood sugar
Suh, Yousin, 96–97
suicide, physician-assisted, 280
Sundrop Farms (Australia), 289
supplements, 286, 303, 304–5, 307
See also specific supplement
survival
and “carrying capacity” of planet, 242–43
and consumption, 288
epigenetics importance to, 21–22
and evolution of humans, 220
and longevity pathways, 129
pharmaceuticals and, 146
sirtuins and, 24
See also survival circuit
survival circuit
and amino acids, 101
and “carrying capacity” of planet, 242–43
as cause of aging, 8–13, 45–50, 52
components of, 57
de nition of, 44
and DNA damage, 162
and epigenetics, 38, 48–50, 60, 61
evolution of, 23
and evolution of aging, 3–5, 6, 7–8
exercise and, 103
and the future, 214
Information Theory of Aging and, 23–26
metformin and, 126
as overworked, 57
of plants, 131
reverse aging and, 63
role in aging of, 35, 48–50sirtuins and, 25, 43, 45–47, 135
and STACs, 133
stress and, 100
and telomeres, 149
universality of, 57
and yeast studies, 35
Suzanne (patient): tracking of, 194
Suzuki, Ichiro, 73
Szilard, Leo, 13–14, 15–16, 20, 23
Tainter, Maurice, 109
tamoxifen, 49–50
Targeting Aging with Metformin (TAME) study, 127
Tatar, Marc, 132
taxes, 232
technology
and “carrying capacity” of planet, 241, 242
and consumption, 283–90
contribution to lifespan of, 89
and diagnosis of disease, 181–86, 199–201, 243
and DNA methyl, 181
doctors and, 176–77
and economic divisions, 233
and elderly in workforce, 251, 253
and evolution of aging, 13
and evolution of scienti c discovery, 16–18, 19, 20–23
and expansion of lifespan, 264
funding for development of, 266, 269
genomics and, 186
and life expectancy, 245
and mapping human genome, 28
as natural, 243optimism about, 209
predictions about, 263
and self-knowledge, 181–86
senescence and, 153
sequencing and, 181
and treatment decisions, 178–80, 181–86
universal access to, 278
See also biosensors/trackers; science; specific technology
telomerase enzyme, 149
telomeres, 17, 18, 30, 38, 52, 84, 102–3, 104, 149
temperature, 26, 105–6, 107, 108–12, 189, 214, 223–24, 304
Thompson, Harriet, 249–50
Thompson, Leonard, 123
Thornton, Janet, 66
Thurmond, Strom, 226–27
Til y, Jonathan, 140
Toles, Tom, 133
TOR (target of rapamycin) genes, 25, 122–23, 125, 145, 264
See also mTOR enzyme
tracking devices/systems. See biosensors/trackers
Transmission Control Protocol/Internet Protocol (TCP/IP), 162
transplants, 17–18, 78, 121, 164, 205–7, 214, 233
travel: and spread of pathogens, 197
treadmil tracking program, 62–63
treatment
and death as a choice, 278–82
making better decisions about, 176–80
refusing, 282
and technology, 176–80, 181–86
trees: longevity of, 53–54, 56, 57
triglycerides, 191
trimethylamine N-oxide (TMAO), 99
trimethylglycine, 305
triple-bypass surgery, 78
trust: with medical records, 197–98tuberculosis, 89
tumors
biosensors/trackers and, 194
and evolution of aging, 5
precision medicine and, 178, 179
and reprogramming, 166, 167, 172, 205–7
See also cancer
twins, 37
ulcerative colitis, 24
UNICEF, 286
United Kingdom
“best way” in, 274
See also London, England
United Nations, 226, 240, 244, 271, 284
United States
average lifespan in, 277
“best way” in, 274
funding for health care in, 276
and Human Capital Index, 275
inadequacy of health care in, 276–77
and treatment as a right, 276–77
See also specific topic
Université Laval, 108
University of Cincinnati: biosensors/trackers studies at, 188
University of Pennsylvania: Wharton School at, 290, 291
vaccines, 82, 122, 148, 155–58, 173–74, 202–4, 214, 238, 300, 301
valine, 101
Van Remmen, Hol y, 15
Vecitibix, 183
vegetarians, 101–2
Vera, Daniel, 295
video home visits by doctors, 186
viruses, 135, 155, 166, 196, 296vision
and blood sugar, 124
and corneal transplants, 206
and diet, 94
ongoing research about, 294, 297
and reprogramming, 164–65, 167, 168, 169, 170, 171–72, 173
treatment for, 18
See also cataracts; macular degeneration
“vitality genes,” 23–26
vitamin A, 286
vitamin B, 119
vitamin B12, 37
vitamin C, 173
vitamin D, 304
vitamin K2, 304
Vitrakvi, 184
Vogt, Peter, 9
volunteers
community, 259
for experiments, 173
See also organ donors
Wada, Juro, 206
Waddington, Conrad H., 21, 58–59, 61, 63, 138, 165, 166
Wagers, Amy, 298
wait time: for doctors, 185–86
Walford, Roy, 92–93
See also consumption
water, consumption of, 284, 288
See also income; poverty; standard of living
See also obesity
Werner, Otto, 83
Werner syndrome, 32–34, 42–43, 83WeWork, 284
Wharton School (University of Pennsylvania), 290, 291
whooping cough, 203
why we age, 12–13
Wilson, Edward O., 241
Wnt, 167
work/workforce
age discrimination in, 251–53, 254, 258–59
automation and, 254
and concern about the future, 293
and ve-day work week, 290
and funding for aging, 269
reentry into, 258–59
rethinking about, 290–92
and skil baticals, 291
See also retirement
World Health Organization, 68, 124, 196, 221, 224, 277, 285–86, 302–3
worms
and as precursors of human experiments, 141
and reprogramming, 173
See also roundworms
wounds, 74–75, 124, 132, 165, 297
wrinkles, 32, 37, 38, 73, 79, 82, 94, 165, 300
Wu, Lindsay, 297
Wyss-Coray, Tony, 299
X chromosome, 183
X factors, 307
xenohormesis, 131
xenotransplantation, 206–7
Xiao Tian, 296–97Yamanaka factors, 164, 165, 166, 167, 169, 172
Yamanaka, Shinya, 163–64
Yang, Jae-Hyun, 296
Yang, Luhan, 206–7
Yankner, Bruce, 298
yeast
aging in, 29–35, 38, 39, 40–44
calorie-restricted, 130–31
cause of aging in, 127–28
DAF-16 genes in, 56
death of, 69
and diet, 91
DNA and, 30–35, 38, 39, 40–44, 152
early studies about, 24, 29–35, 38, 40–44, 91, 111, 127–28
and epigenetics, 38
and hal marks of aging, 43
lessons from studies of, 39
measuring aging in, 130
as precursors for human experiments, 141
rapamycin and, 121
and retrotransposons, 155
and sequencing of yeast genome, 32
and sirtuins, 24, 34–35, 38, 39, 40–41, 43
and sugar, 44
survival circuit and, 35, 45, 47, 48
universal regulators of aging in, 147
and why we age, 39
See also specific researcher or topic
YouGov, 247
youthfulness: prolonged, 265
Yuancheng Lu, 167–69, 171, 296
Zhang, Kang, 52Zhigang He, 169
Zika virus, 196
zombie cel s. See senescence

This publication contains the opinions and ideas of its author. It is sold with the understanding
that the author and publisher are not engaged in rendering health services in the book. The
reader should consult his or her own medical and health providers as appropriate before
adopting any of the suggestions in this book or drawing inferences from it.
The author and publisher speci cal y disclaim al responsibility for any liability, loss, or risk,
personal or otherwise, which is incurred as a consequence, directly or indirectly, of the use and
application of any of the contents of this book.
An Imprint of Simon & Schuster, Inc.
1230 Avenue of the Americas
New York, NY 10020
Copyright © 2019 by David A. Sinclair, PhD
Al rights reserved, including the right to reproduce this book or portions thereof in any form
whatsoever. For information, address Atria Books Subsidiary Rights Department, 1230 Avenue
of the Americas, New York, NY 10020.
First Atria Books hardcover edition September 2019
and colophon are trademarks of Simon & Schuster, Inc.
For information about special discounts for bulk purchases, please contact Simon & Schuster
Special Sales at 1-866-506-1949 or business@simonandschuster.com.
The Simon & Schuster Speakers Bureau can bring authors to your live event. For more
information, or to book an event, contact the Simon & Schuster Speakers Bureau at 1-866-248-
3049 or visit our website at www.simonspeakers.com.
Il ustrations and Sinclairfont by Catherine L. Delphia. Il ustrations in Cast of Characters by
David A. Sinclair, PhD. For reproductions, contact David A. Sinclair, PhD.
Interior design by Ruth Lee-Mui
Cover design by Emma A. Van Deun
Cover il ustration by Grazia Bel abarba
Author photograph © Brigitte LacombeLibrary of Congress Cataloging-in-Publication Data
Names: Sinclair, David A., 1969– author. | LaPlante, Matthew D., author.
Title: Lifespan : why we age—and why we don’t have to / David A. Sinclair, Ph.D., A.O. with
Matthew D. LaPlante ; il ustrations and Sinclairfont by Catherine L. Delphia.
Description: First Atria Books hardcover edition. | New York : Atria Books, 2019. | Includes
bibliographical references and index.
Identi ers: LCCN 2019007196 (print) | LCCN 2019009229 (ebook) | ISBN 9781501191992
(eBook) | ISBN 9781501191978 (hardback)
Subjects: LCSH: Life spans (Biology) | Longevity. | BISAC: SCIENCE / Life Sciences / Genetics
& Genomics. | HEALTH & FITNESS / Diseases / Genetic.
Classi cation: LCC QH528.5 (ebook) | LCC QH528.5 .S56 2019 (print) | DDC 570—dc23
LC record available at https://lccn.loc.gov/2019007196
ISBN 978-1-5011-9197-8
ISBN 978-1-5011-9199-2 (ebook)
Thanks to Christine Liu and her team at the Innovative Genomics Institute (IGI) for permission
to use the Glossary icons. Thanks to Wikipedia for biographical facts in the cast of characters.
Document Outline
Table of Contents
Settings
Theme
Ask about this document
Ask anything about this document.